

Background: Enthesitis and dactylitis are hallmark manifestations of PsA and are reported in 35-50% and 40% of patients with PsA, respectively. [1-2] Patients with enthesitis have greater disease activity, erosive damage and poorer quality of life compared to patients without enthesitis. Similarly, dactylitis is associated with greater disease activity, impaired physical function, erosive disease, and lower likelihood of achieving minimal disease activity. [3] The phase 3 KEEPsAKE 1 and KEEPsAKE 2 trials demonstrate that risankizumab (RZB) provides a high level of durable improvement in musculoskeletal manifestations, including enthesitis and dactylitis. Safety data that was previously reported for the two trials show that RZB is well tolerated over 100 weeks. [4]
Objectives: To evaluate the efficacy of risankizumab on enthesitis and dactylitis by location and severity at baseline in adult patients with PsA.
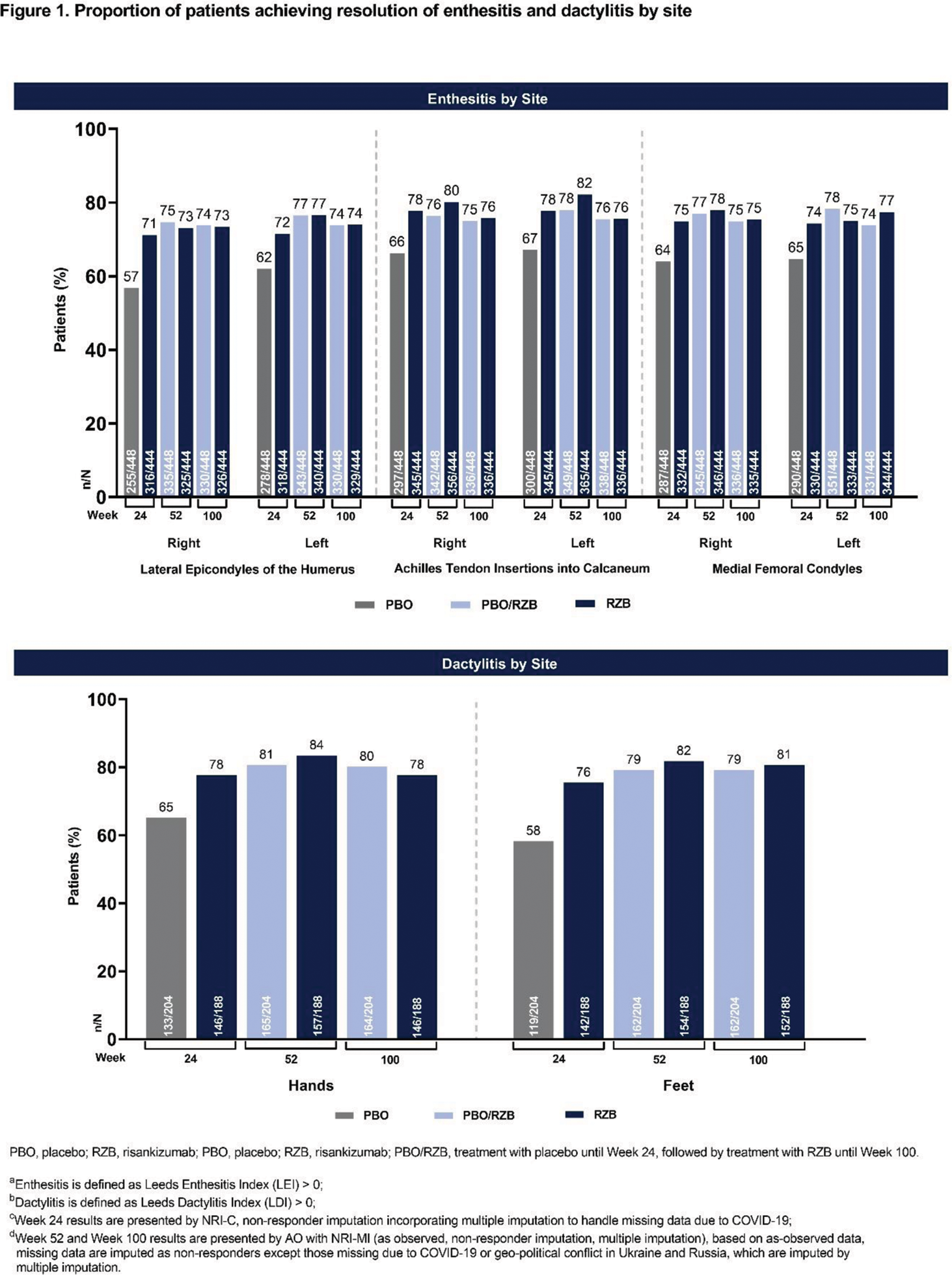
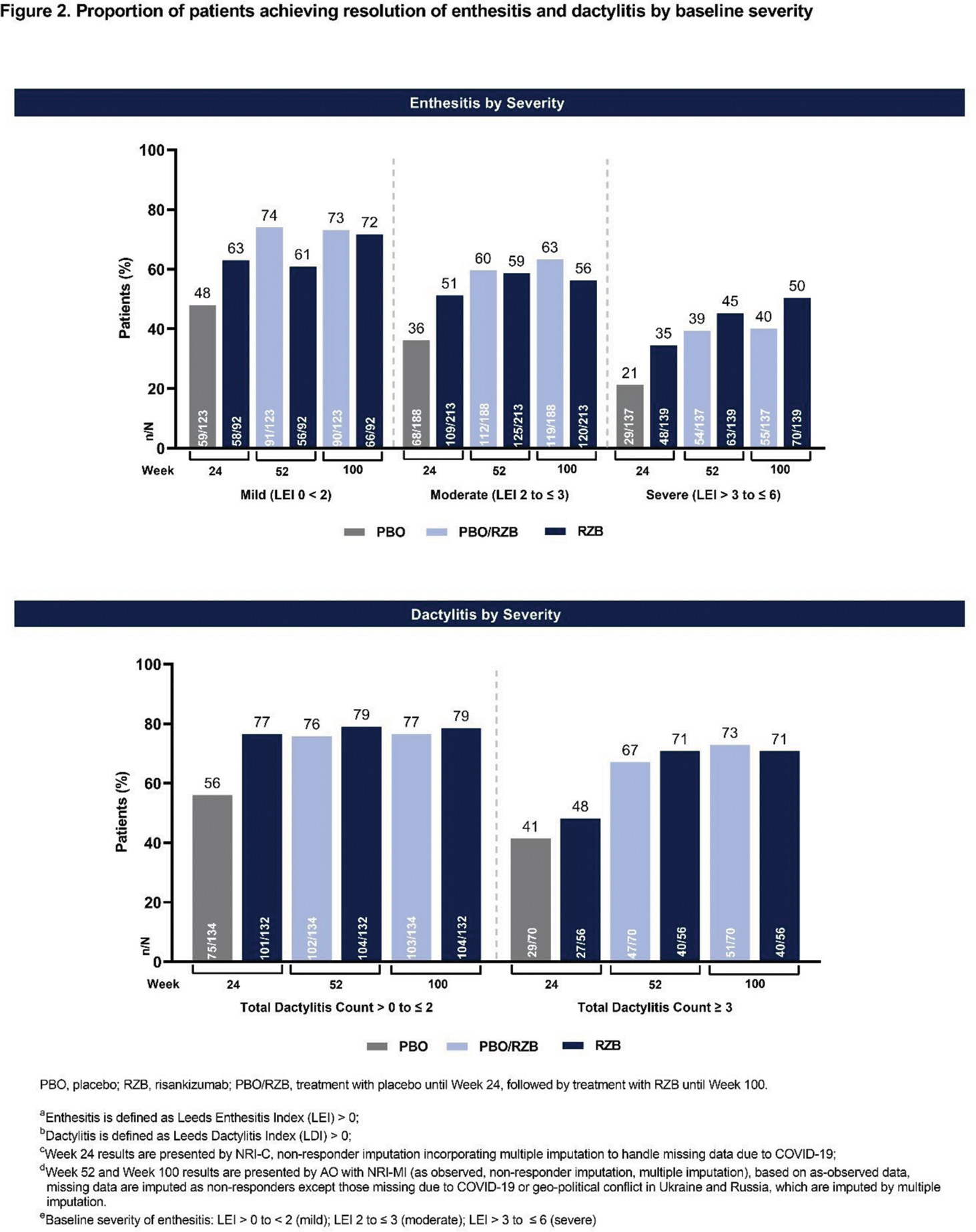
Methods: This post hoc analysis included pooled data from the KEEPsAKE 1 and 2 studies of RZB in the treatment of adult PsA patients with an inadequate response, intolerance, or contraindication to ≥ 1 conventional synthetic DMARDs and/or 1 or 2 biologic therapies (KEEPsAKE 2 only). Patients were randomized 1:1 to receive subcutaneous RZB 150 mg or placebo (PBO). Patients initially randomized to PBO were switched to receive open-label RZB at week 24. For patients with baseline Leeds Enthesitis Index (LEI) > 0, the proportion of patients achieving resolution of enthesitis at the following LEI sites was evaluated: right and left lateral epicondyles of the humerus; right and left Achilles tendon insertions into calcaneum; right and left medial femoral condyles. For patients with baseline Leeds Dactylitis Index (LDI) > 0, the proportion of patients achieving resolution of dactylitis was evaluated. Resolution of enthesitis based on baseline severity was assessed by evaluating the proportion of patients achieving LEI = 0 in the following subgroups: LEI 0 to < 2 (mild); LEI 2 to ≤ 3 (moderate) LEI > 3 to ≤ 6 (severe). Similarly, resolution of dactylitis based on baseline severity of dactylitis was evaluated by assessing the proportion of patients with a total dactylitis count > 0 to ≤ 2 and ≥ 3 at baseline. The presence of enthesitis and dactylitis by sites and baseline severity were reported at week 24, week 52 and week 100.
Results: In the pooled population from the KEEPsAKE 1 and 2 studies, 892 patients (RZB, n = 444; PBO, n = 448) had baseline enthesitis and 392 patients (RZB, n = 188; PBO, n = 204) had baseline dactylitis. Across all sites that were evaluated, treatment with RZB resulted in greater resolution of enthesitis and dactylitis compared to PBO at week 24 (Figure 1). The proportion of patients achieving resolution of enthesitis and dactylitis with treatment with RZB was consistent across all sites evaluated. Furthermore, patients who switched from PBO to RZB 150 mg at week 24 were able to achieve similar resolution of enthesitis and dactylitis as the continuous RZB group by week 52. When evaluating resolution of enthesitis and dactylitis stratified by baseline severity, patients with milder disease at baseline showed consistent responses at all timepoints (Figure 2). In patients with more severe disease at baseline, continued improvement in resolution of enthesitis and dactylitis was seen from week 24 to 52 with RZB treatment, and the resolution was maintained to week 100.
Conclusion: The results from this post hoc analysis show that treatment with RZB 150 mg demonstrates efficacy in the resolution of enthesitis and dactylitis at all sites that were evaluated and across all assessed levels of severity at baseline.
REFERENCES: [1] Kaeley GS, et. al. Semin Arthritis Rheum . 2018; 48: 35 - 43
[2] Kaeley GS, et. al. Semin. Arthritis Rheum. 2018; 48:263 - 273
[3] Mease PJ, et. al. Arthritis Care Res (Hoboken ). 2017; 69:1692 - 9
[4] Erik L, et. al. Arthritis Rheumatol. 2022; 74 (suppl 9) [abstract]
Acknowledgements: NIL.
Disclosure of Interests: Dennis McGonagle AbbVie, Janssen, Novartis, and Pfizer., AbbVie, Janssen, Novartis, and Pfizer, UCB, BMS, Celgene., Oliver FitzGerald AbbVie, Amgen, Bristol Myers Squibb, Celgene, Eli Lilly, Janssen, Novartis, Pfizer Inc, and UCB., AbbVie, Amgen, Bristol Myers Squibb, Celgene, Eli Lilly, Janssen, Novartis, Pfizer Inc, and UCB., Maria-Antonietta D’Agostino Amgen, Galapagos, Novartis, BMS, Janssen, Celgene, AbbVie, UCB pharma, and Eli Lilly., Amgen, Galapagos, Novartis, BMS, Janssen, Celgene, AbbVie, UCB pharma, and Eli Lilly., Yael Klionsky MedIQ, Amgen, AstraZeneca, and Lilly., Cuiyong Yue AbbVie, Jamie Vora AbbVie, Bhumik Parikh AbbVie, Catherine Bakewell AbbVie, Novartis, Pfizer, UCB, Lilly, Janssen, and Sanofi., AbbVie, Novartis, Pfizer, UCB, Lilly, Janssen, and Sanofi.