

Background: It is reported that up to 90.4% of individuals diagnosed with Rheumatoid Arthritis (RA) seek medical assistance due to severe pain. The underlying causes of this pain are multifaceted, involving factors such as inflammation, secondary osteoarthritis, as well as central and peripheral sensitization. CGRP (calcitonin gene-related peptide) is a peptide exerting nociceptive and vasodilatory effects and a debated immunomodulatory effect; inflammatory arthropathies have been associated a local increase of CGRP release and CGRP seems to increase IL6 and IL8 secretion from fibroblast-like synoviocytes isolated from RA patients.
Objectives: The aim of this prospective pilot study was to determine whether patients with RA have detectable levels of circulating CGRP, to investigate the correlation between CGRP and with pain levels reported by the patients and to assess the effect of baricitinib on pain and CGRP levels.
Methods: We enrolled RA patients starting treatment with baricitinib for high-to-moderate active RA. At baseline and after 4 and 12 weeks of treatment we collected clinical data (number of tender and swollen joints), Erytrhosedimentation Rate (ESR), C Reactive Protein (CRP) levels, and patients reported outcomes [Patients Global Assessment (PGA) and pain on a 0-10 cm Visual Analogic Scale (VAS)]. CGRP serum levels were assessed in serum from RA patients at baseline and after 4 and 12 weeks of treatment with baricitinib using an ELISA kit. Data were expressed as mean ± standard deviation or median (IQR) according to distribution. Mann-Whitney and Spearman tests were performed for comparisons and p values < 0.05 were considered statistically significant.
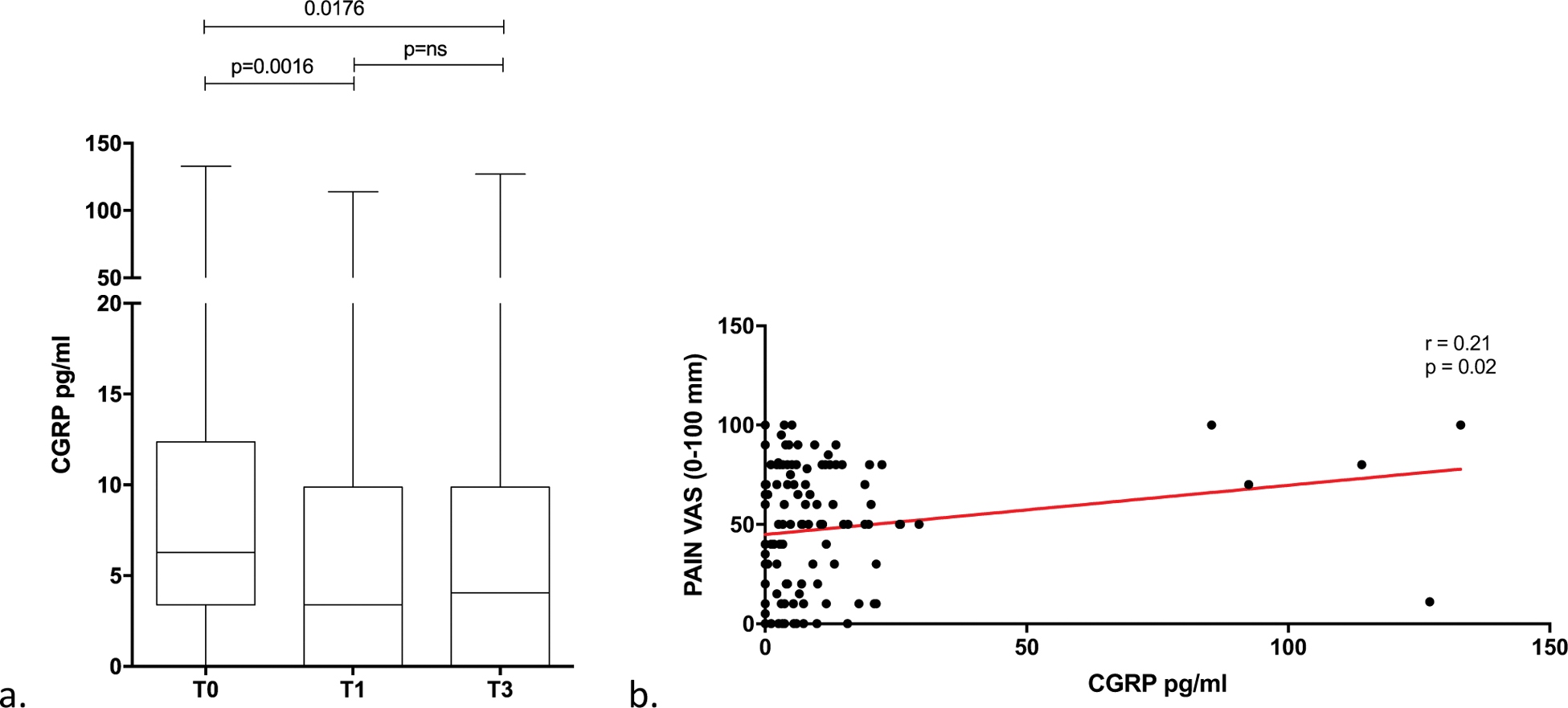
Results: We enrolled 43 patients (F:M =36:7, median age=58, IQR 11 years, median disease duration =144, IQR 150) starting baricitinib. We defined responders (R) patients those who achieved at least a EULAR moderate response (1.2 point reduction) of DAS28_CRP from baseline value and not-responders (NR) those who did not. At baseline pain VAS score did not differ significantly between R and NR. Already after 4 weeks of treatment all patients showed a significant reduction of pain (median pain score was 8(2) at baseline, 4(5) and 2(5) after 4 and 12 weeks, respectively; p=0.0014 and p<0.0001 vs baseline, respectively). R patients had lower pain VAS scores after 4 and 12 weeks of follow-up (p=0.0038 and p=0.0026) compared to NR. CGRP was significantly reduced in the whole cohort at 4 (p=0.0016) and 12 weeks (p=0.018) (Figure 1a). NR patients showed higher CGRP serum levels compared to R, after one month of baricitinib. Levels of CGRP significantly correlated with pain VAS (p=0.0152, r=0.22) (Figure 1b) and PGA (p=0.02, r=0.21), but not with ESR, CRP or disease activity (DAS28_CRP).
Conclusion: With the same disease activity, we found higher levels of CGRP in active RA patients with higher levels of pain. Interestingly, CGRP was reduced to a greater extent in patients who responded to the JAK inhibitor. This preliminary result sheds light on a possible additional mechanism of baricitinib efficacy, linked to modulation of neurotransmission other than to control of inflammation. On the other hand, in NR patients, other mechanisms underlying pain may contribute to non-response to therapy.
REFERENCES: [1] Walsh DA, et al. Nat Rev Rheumatol . Oct 2014;10(10):581-92.
[2] Russell FA, et al. Physiol Rev . Oct 2014;94(4):1099-142.
[3] Raap T, et al. J Rheumatol. Nov 2000;27(11):2558-65.
a. Reduction of serum CGRP in patients treated with baricitinib, from baseline (T0) and after 1 and 3 months of treatment (T1, T3). b. correlation between levels of pain and serum CGRP.
Acknowledgements: NIL.
Disclosure of Interests: Cristina Garufi: None declared, Silvia Mancuso: None declared, Letizia Caruso: None declared, Fulvia Ceccarelli: None declared, Simona Truglia: None declared, Fabrizio Conti Eli Lilly, Abbvie, UCB, Pfizer, Galapagos, Francesca Romana Spinelli Eli Lilly, Abbvie, UCB, Galapagos