

Background: Psoriatic arthritis (PsA), a chronic inflammatory joint disorder associated with psoriasis, has drawn significant attention due to its prevalence and the considerable impact it has on patients’ physical function, quality of life, and psychological well-being. Guselkumab have shown greater persistence compared to traditional treatments like tumor necrosis factor alpha (TNF-α) inhibitors, offering sustained benefits for managing joint and skin symptoms [1, 2]. Here we explore the efficacy and safety of guselkumab in clinical practice.
Objectives: To evaluate treatment persistence in northern Spanish patients from real-world clinical practice who previously failed multiple therapeutic strategies and initiated guselkumab under conditions markedly different from those of clinical trials 6 (Discover I, Discover II and Cosmos) [3].
Methods: Retrospective, multicentric, observational study in patients who met EULAR and ACR criteria for PsA, who received at least one dose of Guselkumab up to 165w. Patient demographic, baseline data and disease profiles were recorded and response variables were statistically analyzed. In addition, adverse events were recorded. Qualitative variables were expressed in absolute (n) and relative (percentages) values and quantitative variables as mean and standard deviation (SD).
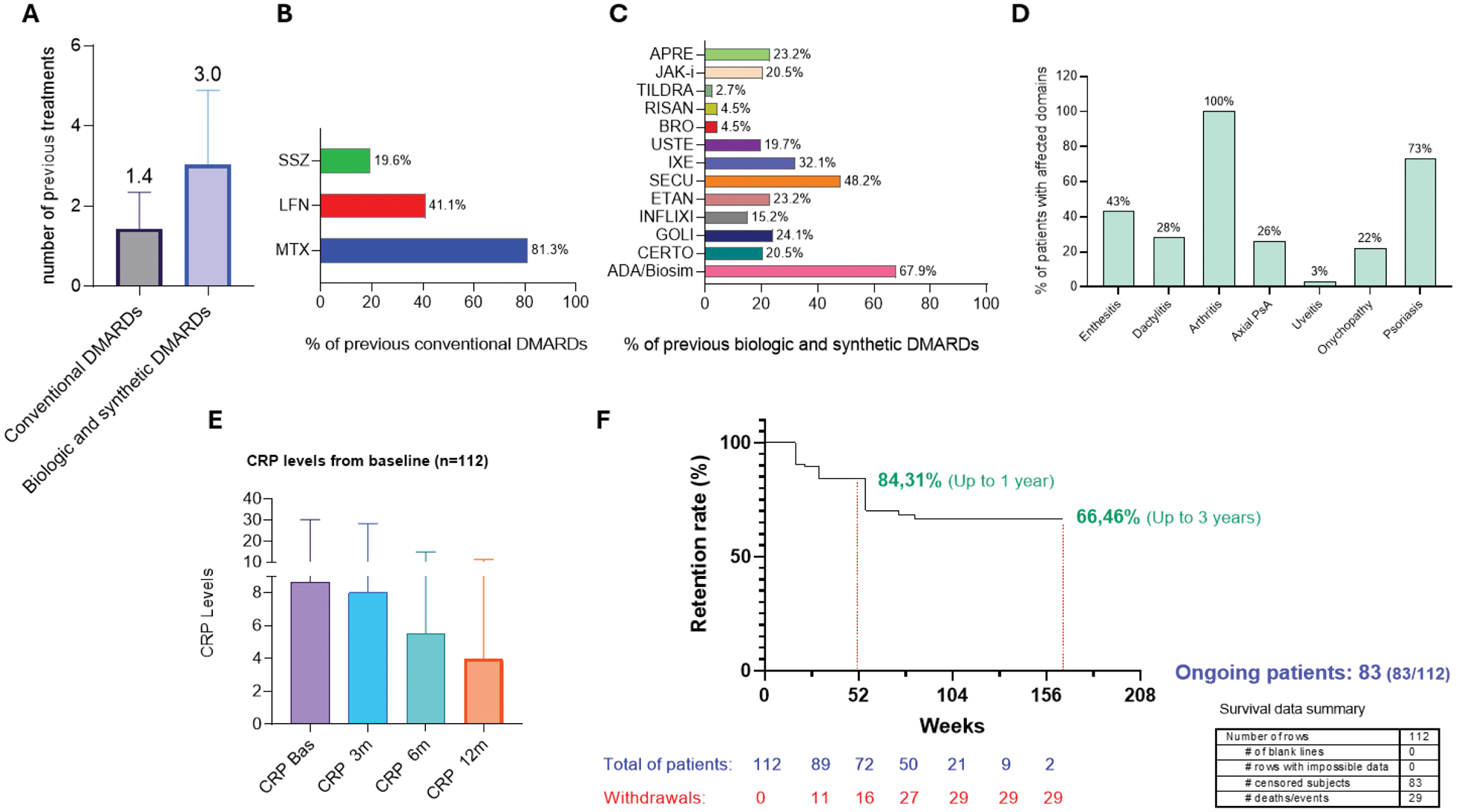
Results: One hundred and twelve patients with moderate-to-severe PsA were included. A mean of 11 (8.1) years elapsed since psoriatic arthritis was diagnosed, while the mean time from guselkumab initiation was 14 (8.8) months. The maximum follow-up period was 3 years. The baseline characterisitics of the study population are shown in Table 1. The primary affected domains were also documented: psoriasis was present in the 73.2% of the sample, enthesitis in 42.9%, dactylitis in 27.7%, axial disease in 25,9% and nail psoriasis in 22.3% of them (Figure 1A). All 112 patients were previously treated with conventional systemic disease-modifying antirheumatic drugs (csDMARDs), being methotrexate (81.3%), leflunomide (41.1%) and sulfasalazine (19,6%) the most frequent treatments used respectively. A mean of 1.4 csDMARDs and 3.0 bDMARDs and tsDMARDs were used in these patients, being adalimumab (67.9%), etanercept (23.2%), golimumab (24.1%), infliximab (15.2%), certolizumab (20.5%), secukinumab (48.2%), ixekizumab (32.1%) and ustekinumab (19.2%) the most frequent therapeutic agents used. Regarding tsDMARDs JAK-i and apremilast were used in 23.2% and 20.5% of patients respectively (Figure 1B-C-D). Regardless of prior treatment history, guselkumab enabled to achieve significant improvements in joint symptoms compared to baseline. Molecular markers of inflammation confirmed clinical improvement through a significant reduction by the decreased levels of C-reactive protein (CRP) (Figure 1E). The survival rate of guselkumab in these cohort was 84.31% for the first year and 66.46% for the second and third year of treatment. 16 withdrawals were observed in the first 12 months due to lack of efficacy increasing to 29 in the second year of treatment. Of the initial 112 patients, 83 are still undergoing treatment, of whom 72 have already exceeded 52 weeks of guselkumab therapy. (Figure 1F). No dropouts related with adverse events or guselkumab safety profile were detected.
Table 1. Demographic characteristics of the treated population. Qualitative variables were expressed in absolute (n) and relative (percentages) values and quantitative variables as mean and standard deviation (SD).

Main PsA comorbidities (A), previous DMARD synthetic and biologic treatments to guselkumab (B,C,D), CRP reduction levels from baseline to 12 months (E) and survival curve for guselkumab treatment up to 165 weeks (F).
Conclusion: In this study, we present a cohort of patients from the northern of Spain treated with guselkumab under real-world clinical practice conditions with long-term effectiveness data. The drug showed high persistence (84.31% in the first year) in a more complex patient profile than those examined in clinical trials. There were no notable adverse events that compromised patient safety or justified drug withdrawal. These data could provide additional and more reliable information to Spanish clinicians using guselkumab in refractory patients and demonstrate the therapeutic potential and the high persistence that could be achieved by using the drug in earlier treatment lines.
REFERENCES: [1] Coates LC, Kavanaugh A, Mease PJ, et al. Group for Research and Assessment of Psoriasis and Psoriatic Arthritis 2015 treatment recommendations for psoriatic arthritis. Arthritis Rheumatol. 2016 May;68(5):1060-71. doi: 10.1002/art.39573.
[2] Gossec L, Coates LC, de Wit M, et al. Management of psoriatic arthritis in 2016: a comparison of EULAR and GRAPPA recommendations. Nat Rev Rheumatol. 2016 Dec;12(12):743-750. doi: 10.1038/nrrheum.2016.183. Epub 2016 Nov 10.
[3] Rahman P, Boehncke WH, Mease PJ, et al. Safety of Guselkumab With and Without Prior Tumor Necrosis Factor Inhibitor Treatment: Pooled Results Across 4 Studies in Patients With Psoriatic Arthritis. J Rheumatol. 2023;50(6):769-780. doi:10.3899/jrheum.220928.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (