

Background: UCTD (Undifferentiated Connective Tissue Disease) describes a clinical condition characterized by the presence of isolated features of CTD (Connective Tissue Diseases) without meeting diagnostic criteria for any specific disease. Studies have indicated a higher risk of adverse obstetric events in these patients. In the case of SLE (Systemic Lupus Erythematosus), there is an elevated risk of postpartum flares, suggesting that pregnancy in UCTD patients could act as a triggering factor for flares and for meeting the diagnostic criteria for a specific CTD.
Objectives: The primary objective of this study is to analyse maternal and fetal outcomes in a Spanish cohort of UCTD patients. Secondary objectives include identifying potential risk factors associated with adverse maternal and obstetric events in UDCT patients, comparing maternal and fetal outcomes with those of a cohort of CTD patients, defined as SLE, and determining the percentage of UCTD patients who evolve into a defined CTD after pregnancy.
Methods: This is a multicenter retrospective observational study of UCTD and SLE patients who became pregnant between January 1, 2005, and December 31, 2023, evaluated in specialized pregnancy clinics at two hospitals: Hospital Universitario de Canarias and Hospital Universitario Marqués de Valdecilla.
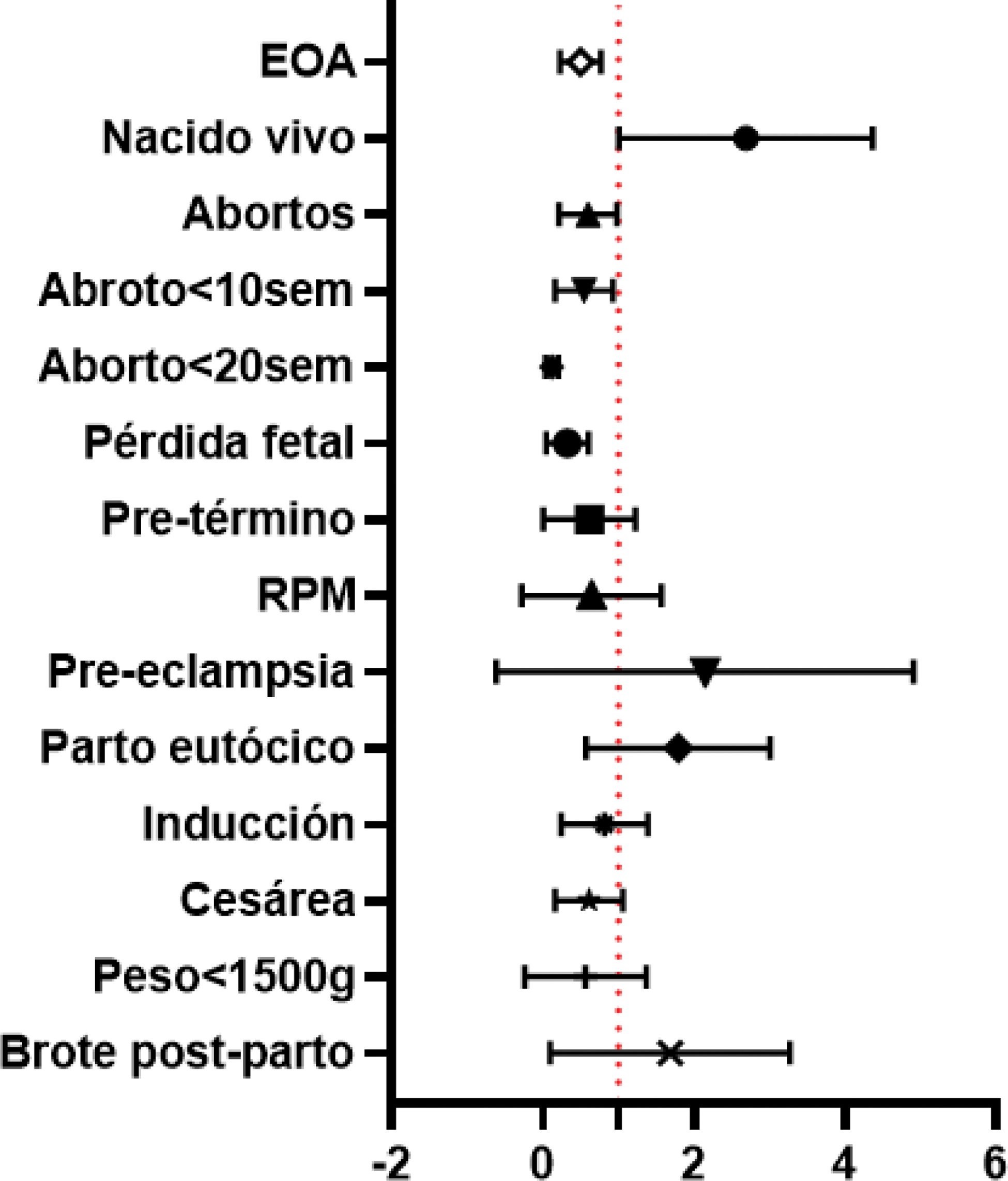
Results: A total of 84 pregnancies in UCTD patients and 219 pregnancies in SLE patients were included. Table 1 shows demographic and disease characteristics at the time of conception. Adverse obstetric events (AOEs) occurred in 52% of UCTD patients and 75% of SLE patients (OR: 0.45, 95% CI: 0.25–0.79, p=0.01), with a higher live birth rate in the UCTD group (76% vs. 56%, OR: 2.33, 95% CI: 1.21–4.51, p=0.012) and a lower risk of spontaneous abortion (18% vs. 32%, p=0.019), particularly in cases of miscarriage before 10 weeks (80% vs. 86%, OR: 0.46, 95% CI: 0.22–0.96, p=0.038) and before 20 weeks (4% vs. 14%, OR: 0.21, 95% CI: 0.05–0.094, p=0.041). The UCTD group also had a lower risk of fetal loss (7% vs. 23%, OR: 0.24, 95% CI: 0.09–0.63, p=0.04). Preterm delivery before 37 weeks (6% vs. 14%, p=0.05), extreme preterm delivery before 34 weeks (0% vs. 5%, p=0.036), and pathological Doppler ultrasound findings (0% vs. 18%, p=0.016) were less frequency in UCTD patients. Gestational age (39 [38–40] vs. 38 [37–40] weeks, p=0.001) and birth weight (3206 ± 406 vs. 2947 ± 599 g, β-coefficient: 204, 95% CI: 8.4–400.2, p=0.04) were higher compared with SLE (Figure 1). Multivariable analysis showed that acetylsalicylic acid (ASA) use reduced the risk of miscarriage in UCTD patients (OR: 0.06, 95% CI: 0.01–0.46). The presence of anti-Ro antibodies was associated with a higher risk of AOEs in UCTD patients (OR: 3.21, 95% CI: 1.1–9.7), while anti-dsDNA antibodies increased risk in SLE patients (OR: 2.84, 95% CI: 1.22–6.65). After five years, 42% of UCTD patients progressed to a defined CTD.
Conclusion: In UCTD patients, ASA use decreases the risk of miscarriage, whereas anti-Ro antibodies increase complications, as do anti-dsDNA antibodies in SLE patients. These findings underscore the importance of multidisciplinary management in UCTD and SLE patients, with special attention to immunological profiles. Further studies are needed to identify and evaluate other risk factors associated with AOEs in these conditions.
Demographic and clinical characteristics at conception.
| UCTD
| SLE
| p | ||||
|---|---|---|---|---|---|---|
| Age at conception (SD) | 39 ± 2.4 | 34±10.4 | <0.001 | |||
| Caucasian race, n (%) | 78 (93) | 202 (92) | 0.86 | |||
| Comorbilities n(%) | ||||||
| Obesity (BMI>30) | 10 (12) | 25 (11) | 0.79 | |||
| Smokers | 14 (17 ) | 72 (33 ) | 0.004 | |||
| Hypertension | 2 (2 ) | 27 (12 ) | 0.008 | |||
| Dislipydemia | 3 (4) | 13 (6) | 0.57 | |||
| Diabetes Mellitus | 0 (0) | 5 (2) | 0.33 | |||
| Thyroid disease | 15 (18) | 34 (16) | 0.62 | |||
| Gynecological | 22 (26) | 40 (18) | 0.15 | |||
| Myomas | 5 (23) | 17 (43) | 0.59 | |||
| Polycystic ovary syndrome | 6 (27) | 7 (18) | 0.13 | |||
| Endometriosis | 3 (14) | 5 (13) | 0.69 | |||
| Uterine anomalies | 7 (32 ) | 5 (13 ) | 0.016 | |||
| Total number of total pregnancy n(%) | ||||||
| 1 | 18 (21) | 42 (19) | ||||
| 2 | 19 (23) | 85 (39) | 0.025 | |||
| ≥3 | 47 (56) | 92 (42) | ||||
| Previous Miscarriage* | 14 (35) | 37 (47) | 0.12 | |||
| Disease durataion (years), median (IQR) | 2.5 (1-5.2 ) | 7 (3-13 ) | <0.001 | |||
| Thrombophilia, n(%) | 3 (4) | 7 (3) | 0.99 | |||
| Thrombosis, n(%) | 4 (5) | 19 (9) | 0.34 | |||
| Historical previous kidney disease n(%) | 5 (6 ) | 38 (17 ) | 0.011 | |||
| Secondary APS n(%) | - | 40 (19) | - | |||
| Antiphospholipid antibodies n(%) | ||||||
| Anti-beta-2-glycoprotein IgG | 33 (39) | 128 (58) | 0.66 | |||
| Anti-beta-2-glycoprotein IgM | 35 (42) | 119 (54) | 0.21 | |||
| Anti cardiolipin IgG | 35 (42 ) | 134 (61 ) | 0.05 | |||
| Anti cardiolipin IgM | 37 (44 ) | 126 (58 ) | 0.024 | |||
| Lupic anticoagulant | 25 (30 ) | 95 (43 ) | 0.013 | |||
| Triple positive | 0 | 28 | <0.001 | |||
| High risk antibodies | 5 (6 ) | 80 (37 ) | <0.001 | |||
| Anti Ro, n(%) | 22 (26) | 64 (29) | 0.77 | |||
| Anti La, n(%) | 9 (11) | 25 (11) | 0.77 | |||
| Anti Ro+AntiLa | 9 (11) | 25 (11) | 0.86 | |||
| Anti-dsDNA, n(%) | 5 (6 ) | 67 (31 ) | <0.001 | |||
| Anti-dsDNA titre, median (IQR) | 62 (55-209) | 182 (82-390) | 0.246 | |||
| Low complement | 3 (4 ) | 50 (23 ) | <0.001 | |||
| Hematological disorders at conception* | ||||||
| Anemia | 2 (5) | 8 (10) | 0.49 | |||
| Leukopenia | 4 (10 ) | 24 (31 ) | 0.012 | |||
| Lymphopenia | 2 (5 ) | 16 (21 ) | 0.030 | |||
| Thrombocytopenia | 2 (5) | 11 (14) | 0.22 | |||
| SLDAI median (IQR) | - | 1 (0-4) | - | |||
APS: Antiphospholipid Syndrome. BMI: Body Mass Index. UDTC: undifferentiated Connective Tissue Disease. SLE: Systemic Lupus Erythematosus. SLDAI: Systemic Lupus Erythematosus Disease Activity Index. Triple positive: Anti-beta-2-glycoprotein (IgG or/and IgM) and Anti-cardiolipin (IgG or/and IgM) and Lupic anticoagulant (LAC) positive confirmed. High risk antibodies: Positive LAC or Triple Positivity or Persistently Positive aCL at Medium-High Titter. Haematological disorder at conception: from the last period date to six months previous. Gynecological comorbidities including all above and other (salpingectomy for ectopic pregnancy). *(% based on n (UCTD: 41; SLE: 78).
Forest plot of obstetric outcomes. UDCT vs SLE.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (