

Background: Systemic lupus erythematosus (SLE) is a chronic autoimmune disease characterized by the production of autoantibodies against nuclear and cytoplasmic antigens, affecting multiple organs [1]. The pathogenesis of SLE involves the interplay of various factors such as genetic predisposition, autoantibody production, and inflammatory cytokine release [1]. T cell subsets play a crucial role in SLE-related autoimmunity and organ inflammation. CD49f is involved in T cell differentiation, migration, and function [2]. Although dysregulation of CD49f-positive T cells has been reported in ulcerative colitis, the role of CD49f in SLE remains underexplored [3].
Objectives: To examine the expression and function of CD49f in various subsets of CD4-positive T cells within human peripheral blood. Furthermore, to investigate alterations in regulatory T cells (Tregs) and CD4-positive T cells in SLE patients, while evaluating the clinical significance of these changes.
Methods: Fresh peripheral blood was obtained from 43 SLE patients and 21 matched healthy controls. Peripheral blood mononuclear cells (PBMCs) were isolated, and flow cytometry was employed to assess the expression of CD49f in Tregs and non-Treg CD4-positive T cells. A variety of function-associated molecules were measured to compare the characteristics of CD49f-positive and CD49f-negative subsets. CD49f expression was evaluated in both Tregs and CD4-positive T cells, with comparisons made between SLE patients and healthy controls. Furthermore, the study explored the correlations between subsets related to CD49f and clinical indicators. To assess the potential diagnostic value of these subsets for SLE, receiver operating characteristic (ROC) curve analysis was also performed.
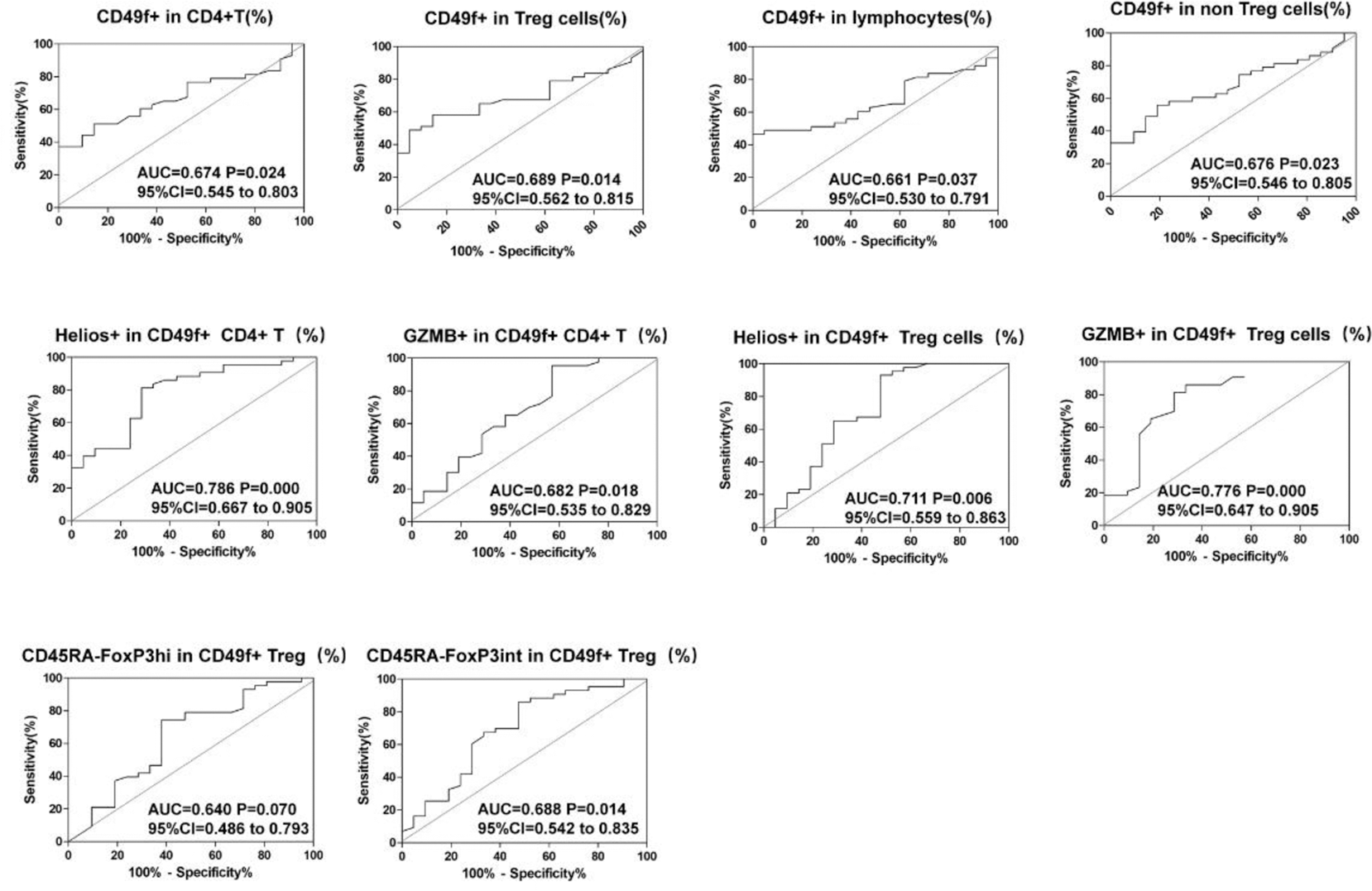
Results: The expression of CD49f was found to be significantly higher in Tregs than other CD4-positive T cell subsets. CD49f-positive CD4 T cells displayed elevated levels of CD226 and reduced expression of CD45RA. Within the CD49f-positive Tregs population, there was a higher proportion of CD45RA-negative FoxP3int cells, alongside increased expression of CD226, but less GZMB and Helios. Notably, the percentage of CD49f-positive cells was markedly increased in SLE patients. Strong correlations were observed between various clinical indicators and CD49f-related metrics. ROC analysis indicated the diagnostic value of CD49f-positive subsets, with area under the curve (AUC) values ranging from 0.640 to 0.786.
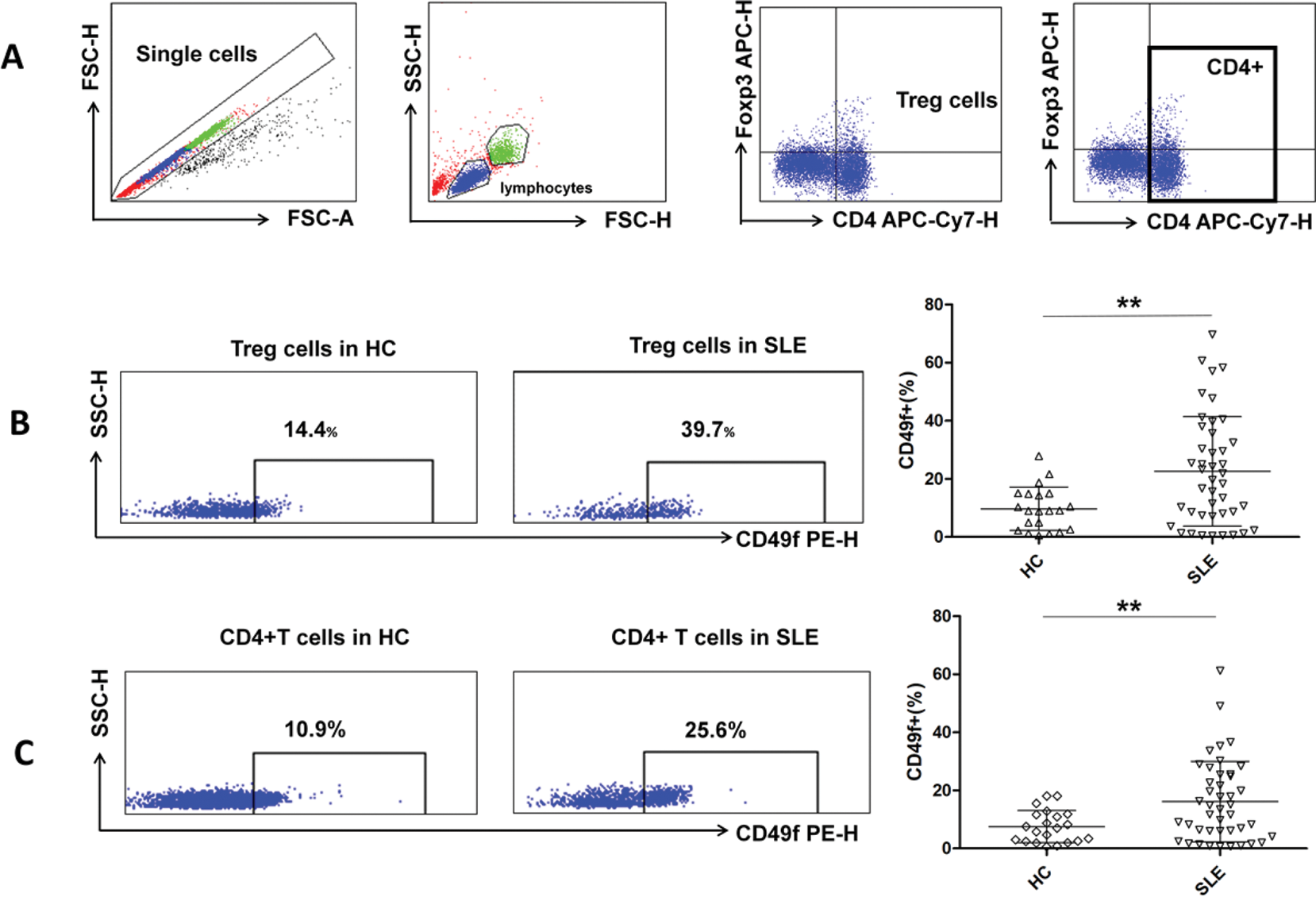
Comparison of proportions of Treg cell subpopulations between SLE patients and healthy individuals.
PBMCs were obtained from healthy controls (n = 21) and SLE patients (n = 43). Cells were collected and stained for CD4, FoxP3, CD45RA, CD49f, Helios, TIGIT and CD226.
A. Representative flow cytometry scatter plots are shown. The numbers in the plots indicate the proportions of the three subsets of CD49f+ Treg cells between healthy controls and SLE patients. Comparison of the proportions of activated Treg(CD45RA-FoxP3hi), non-Treg(CD45RA-FoxP3int) and resting Treg(CD45RA+FoxP3int) in CD49f+ Treg cells between HCs and SLE patients.
B&C. Representative flow cytometry scatter plots are shown. Helios+, TIGIT+ and CD226+ percentages in CD49f+Treg cells between healthy controls and SLE patients were compared.
Results are presented as mean values with standard deviation. *P < 0.05; **P < 0.01; ns, not significant.
ROC curve analysis of CD49f-associated cell subsets to assist in SLE diagnosis.
Flow cytometry was used to detect CD49f as well as expression of Helios, GZMB, FoxP3 and CD45RA in T cell subsets of healthy controls (n = 21) and SLE patients (n = 43). ROC curves were generated to assess the ability of various indicators to differentiate SLE patients from healthy controls, and the AUCs were calculated. The AUCs and 95 % confidence interval were calculated to evaluate their effectiveness in the diagnosis of SLE.
Conclusion: CD49f-positive CD4 T cells exhibit features indicative of activation, while CD49f-positive Tregs demonstrate diminished suppressive function. The expression of CD49f is significantly higher in SLE patients, reflecting autoimmune imbalance and providing valuable diagnostic insights.
REFERENCES: [1] Fortuna G, Brennan M T. Systemic lupus erythematosus: epidemiology, pathophysiology, manifestations, and management[J]. Dental Clinics of North America, 2013, 57(4): 631-655.
[2] Bigoni-Ordóñez G D, Czarnowski D, Parsons T, et al. Integrin α6 (CD49f), The Microenvironment and Cancer Stem Cells[J]. Current Stem Cell Research & Therapy, 2019, 14(5): 428-436.
[3] Weerakoon H, Straube J, Lineburg K, et al. Expression of CD49f defines subsets of human regulatory T cells with divergent transcriptional landscape and function that correlate with ulcerative colitis disease activity[J]. Clinical & Translational Immunology, 2021, 10(9): e1334.
Acknowledgements: We extend our heartfelt appreciation to the Rheumatic Immunology Department of Peking University People’s Hospital for granting access to the medical records that were crucial for this study.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (