

Background: Complement activation is known to play a dual role in the pathogenesis of systemic lupus erythematosus (SLE) and lupus nephritis (LN). It is protective through the effective clearance of immune complexes and cellular waste but becomes damaging when uncontrolled complement activation promotes kidney injury. Although aberrant complement activation and regulation is involved in the pathogenesis of SLE, understanding of its role in the activity of LN remains limited.
Objectives: To evaluate levels of circulating and excreted complement activation and regulatory markers in relation to SLE and LN activity, and in relation to response to treatment for active LN.
Methods: We analysed activation profiles of the classical and alternative complement pathway, as well as concentrations of complement activation-inhibitory and regulatory proteins in matched serum and urine samples from 20 SLE patients without LN (10 with active SLE and 10 in remission) from Karolinska University Hospital in Stockholm, Sweden, 21 SLE patients with active biopsy-proven LN from Cliniques universitaires Saint-Luc in Brussels, Belgium, and 15 healthy controls (HC) from Karolinska University Hospital. LN samples were analysed at two time points, i.e., before commencement of immunosuppressive therapy for active LN (T0) and after one year of therapy (T1), mainly comprising glucocorticoids and either mycophenolate mofetil or low-dose intravenous cyclophosphamide. Concentrations of complement proteins Ba, Bb, C3a, C4a, C5a, sC5b-9, factor H, and factor I were assessed using Quidel MicroVue Multiplex Complement Array. The Mann–Whitney U test was employed to compare levels of complement proteins between groups.
Results: Serum levels of complement fragments Ba, Bb, C3a, C5a, factor I, factor H did not differ significantly between patients with SLE, with or without active LN, and HC. However, serum levels of C4a and sC5b-9 were significantly higher in patients with LN compared to SLE patients without LN (Figure 1A, median 1641 vs 923 ng/mL; p=0.0003 and 2414 vs 1625 ng/mL; p=0.019, respectively), whereas serum Bb, C3a, factor I, and factor H levels were significantly higher in SLE patients without LN (median 3.93 vs 2.53 μg/mL; p=0.0005; 1863 vs 1322 ng/mL; p=0.003; 42.7 vs 22.9 μg/mL; p=0.0001; 469.5 vs 324 μg/mL; p=0.004, respectively). In LN patients, serum levels of Ba, Bb, C5a, cC5b-9, and factor I were significant reduced after one year of immunosuppressive therapy (1580.3 vs 968.7 ng/mL; p=0.0006; 3.51 vs 2.48 μg/mL; p=0.01; 31.8 vs 24.2 ng/mL; p=0.04; 3164.7 vs 2154 ng/mL; p=0.009; 29 vs 20.6 μg/mL; p=0.009, respectively). Analysis of urine samples revealed significantly higher levels of all measured complement proteins in LN patients both compared to SLE without LN and to HC (Figure 1B; p<0.0001 for all comparisons). Consistent with findings in serum, urine levels of Ba and C5a were significantly reduced in LN patients after one year of therapy (52.1 vs 22.5 ng/mL; p=0.0002; 1.92 vs 0.31 ng/mL; p=0.0001, respectively). Complement activation did not differ in patients with active SLE compared to SLE patients in remission; none of the evaluated complement proteins showed significant differences between the two groups, in serum or in urine.
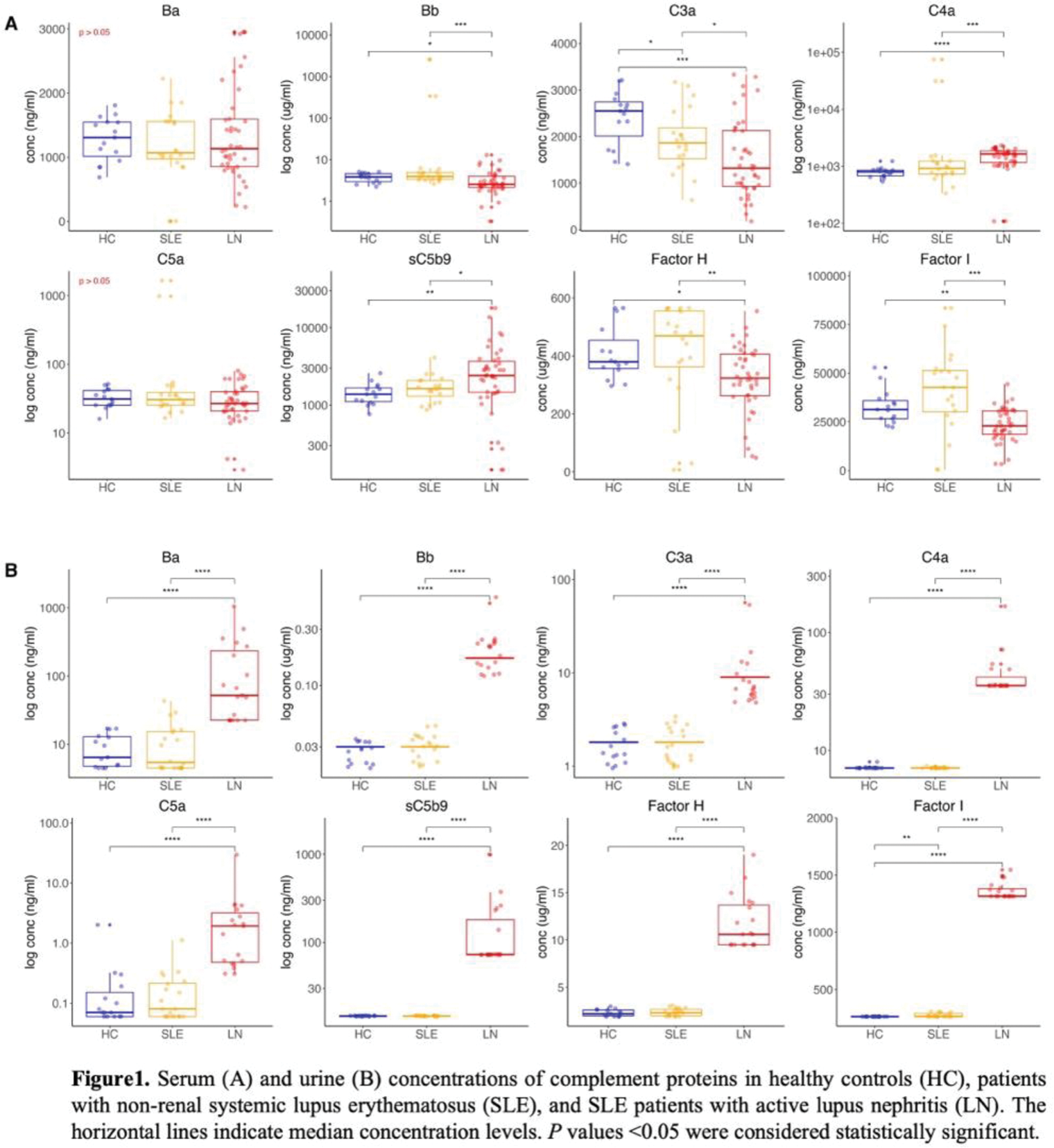
Conclusion: Our data indicate that elevated urine levels of complement activation and regulatory markers are characteristic of SLE patients with LN. Notably, urine levels of C5a and Ba were significantly reduced following therapy, suggesting their potential as biomarkers of renal activity.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (