

Background: Giant cell arteritis (GCA) is a systemic inflammatory disorder, which affects medium and large-sized vessels, most commonly seen in people over 50 years of age [1, 2]. Narrowing of vessels causes ischemia, with a range of associated symptoms and complications following [3]. GCA is primarily treated with glucocorticoids (GC) [2], however tapering schedules exceeding recommended timeframes are often needed for reasons including risk of relapse [4]. Subsequent GC treatment-related complications may follow [1]. Development of alternative therapies has been driven by a need for effective GC-sparing treatment options providing maintenance of remission and improved safety profiles [2].
Objectives: To identify and summarise all available published evidence on the clinical efficacy, and safety of pharmacological interventions for the treatment of adult patients aged ≥50 years with GCA.
Methods: A systematic literature review was conducted in July 2024. Search strategies were implemented in databases including EMBASE, MEDLINE, CDSR and CENTRAL. Additional hand searches were performed on key congress websites, clinical trial registries, HTA agency websites and bibliographic sources. Choice of included studies, data extraction and results summarization followed currently accepted and established best practices [5–7].
Results: A total of 89 publications were identified relating to 12 clinical trials across 7 treatment options (Table 1). The trials included patients with new onset and relapsing GCA. Across the included trials, where published, the mean age of patients ranged between ~69 and ~74 years of age, majority were female (~73%), and White/Caucasian (~97%) (weighted averages). Common symptoms of GCA included headaches, temporal artery abnormalities, jaw claudication and scalp tenderness. Where included in publications, PMR symptoms or features were reported in over half of patients. Tocilizumab (TOC; biologic, IL-6 inhibitor), upadacitinib (UPA; small molecule JAK inhibitor) and adalimumab (ADA; biologic, anti-TNF) were the only treatments reporting results from Phase 3 RCTs [8–12]. The trials concluded that both TOC and UPA, in combination with 26wk GC tapering schedule, demonstrated superior efficacy (sustained remission) and decrease in GC use in comparison to placebo (PLB) (+ GC taper) [8, 9]. Whereas ADA plus GC taper at 26wks was not associated with a significant GC-sparing effect nor reduced rate of relapse compared to PLB (plus GC taper at 26wks) [10]. Data for the remaining 4 treatments were based on Phase 2 RCTs [mavrilimumab (MAV; biologic, GM-CSFr antagonist), secukinumab (SEC; biologic, IL-17A inhibitor), abatacept (ABA; biologic, CLTA-4Ig)] and single-arm trial [baricitinib (BAR; small molecule JAK inhibitor)] [11–16]. MAV, ABA, and SEC in combination with a predefined GC taper all demonstrated superior sustained remission compared to PLB + GC taper. SEC and ABA reported long term remission over a 52 or 64wk treatment period, respectively. A larger randomized trial would be required to further determine the overall efficacy of BAR among the GCA population (Table 2). There was limited published data on safety. Treatments were reported to be well tolerated, without identification of new safety signals compared to their recognized safety profile. Infections or serious infections were among the most commonly reported AEs, with few discontinuing due to safety concerns overall.
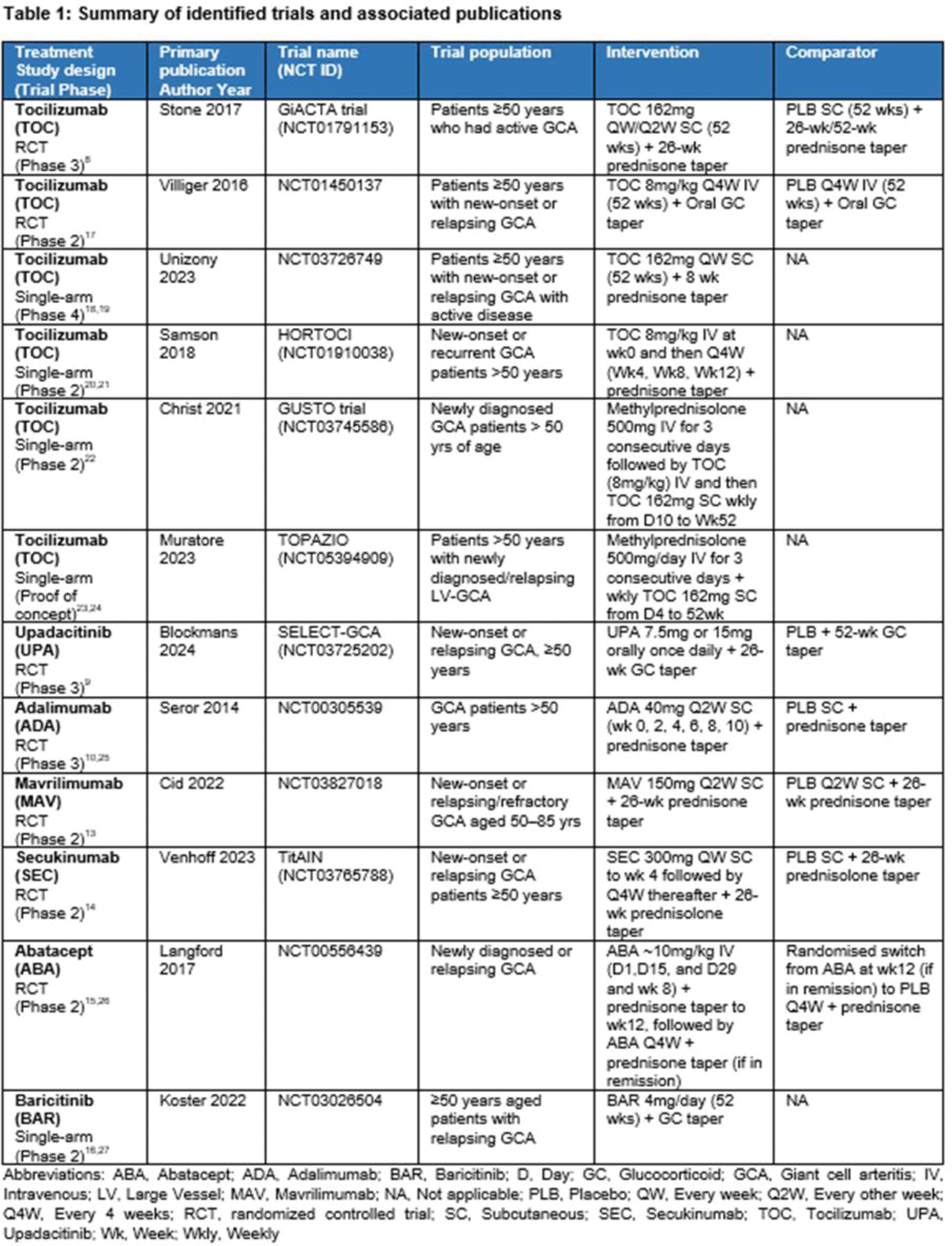
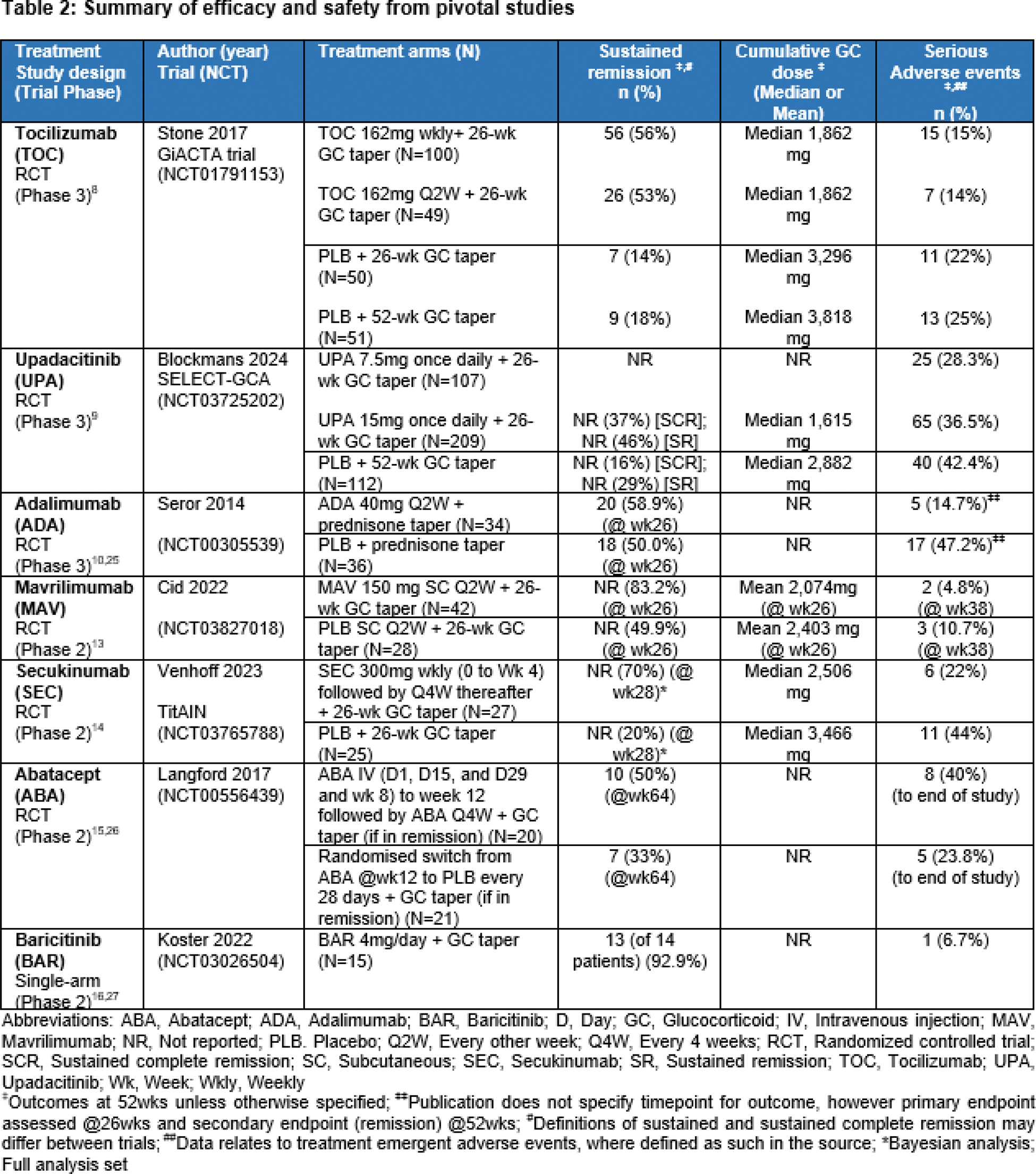
Conclusion: Based on the findings from the review there are limited treatment options available for GCA patients. TOC received regulatory approval [4], while Phase 3 results for UPA demonstrate promising efficacy and SEC is a monoclonal antibody currently in active Phase 3 development. Continued development of effective alternative glucocorticoid-sparing treatment options will support patients contraindicated or unresponsive to current treatment options and reduce the clinical burden among this older, high risk patient population.
REFERENCES: [1] Ameer MA. StatPearls. 2024.
[2] González-Gay MA. BMC Geriatr. 2019;19(1):200.
[3] Li KJ. Arthritis Res Ther. 2021;23:82.
[4] Mollan SP. Eye. 2021;35:699-701.
[5] Higgins JPT. Cochrane Handbook. 2023 [Accessed: 02 Jul 2024]. Available from:
[6] CRD, Univ of York. 2009 [Accessed: 02 Jul 2024]. Available from:
[7] Moher D. Ann Intern Med. 2009;151(4):264-269.
[8] Stone JH. NEJM. 2017;377(4):317-328.
[9] Blockmans D. Ann Rheum Dis. 2024;83(Suppl 1):232-233.
[10] Seror R. Ann Rheum Dis. 2014;73(12):2074-2081.
[11] Harrington R. Biologics. 2021;15:17-29.
[12] Bechman K. Pharmacol Res. 2019;147:104392.
[13] Cid M. Ann Rheum Dis. 2022;81(5):653-661.
[14] Venhoff N. Lancet Rheumatol. 2023;5(6):e341-e350.
[15] Langford CA. Arthritis Rheumatol. 2017;69(4):837-845.
[16] Koster MJ. Ann Rheum Dis. 2022;81(6):861-867.
[17] Villiger PM. Lancet. 2016;387(10031):1921-1927.
[18] Clinicaltrials.gov. [Accessed: 20 Sep 2024]. Available from:
[19] Unizony S. Lancet Rheumatol. 2023;5(12):e736-742.
[20] Samson M. Eur J Intern Med. 2018;57:96-104.
[21] Clinicaltrials.gov. [Accessed: 20 Sep 2024]. Available from: clinicaltrials.gov/study/NCT01910038
[22] Christ L. Lancet Rheumatol. 2021;3(9):e619-626.
[23] Muratore F. Ann Rheum Dis. 2023;82(Suppl 1):633.
[24] Clinicaltrials.gov. [Accessed: 20 Sep 2024]. Available from: clinicaltrials.gov/study/NCT05394909
[25] Clinicaltrials.gov. [Accessed: 20 Sep 2024]. Available from: clinicaltrials.gov/study/NCT00305539
[26] Clinicaltrials.gov. [Accessed: 20 Sep 2024]. Available from: clinicaltrials.gov/study/NCT00556439
[27] Clinicaltrials.gov. [Accessed: 20 Sep 2024]. Available from: clinicaltrials.gov/study/NCT03026504
Acknowledgements: NIL.
Disclosure of Interests: Himanshu Modi Current employee of Novartis Healthcare Pvt. Ltd, Sarah Jane McKenna Shareholder of Novartis Ireland and Sandoz, Current employee of Novartis Ireland Ltd, Jessica Commane Shareholder of Novartis Ireland and Sandoz, Current employee of Novartis Ireland Ltd, Udaya Sri Lakkakula Current employee of Novartis Healthcare Pvt. Ltd.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (