

Background: Patients suffering from SLE are at risk for physical, emotional, and social challenges that negatively impact their quality of life. Non-adherence to treatment and follow-up is complex and multifactorial and often impairs remission achievement and/ or may lead to disease flares, which carry a significant risk of irreversible damage. Methods to improve adherence are scarce and not well studied.
Objectives: To conduct a structured peer mentoring program for patients with SLE aiming to improve coping skills and adherence to treatment, better disease control, and improved quality of life. This interim report focuses on the mentors’ perspective of the program.
Methods: Adult SLE patients treated at the Lupus clinic of a tertiary medical center were invited to participate in a mentoring program as mentors or mentees. Mentors were selected based on a high level of coping/self-efficacy as assessed by the primary investigators (TE, DP) and a sociologist regarding their competence, maturity, emotional stability, verbal communication skills, and willingness to participate. Mentees were selected by their treating rheumatologist if considered to have poor adherence and/or a low level of coping/self-efficacy. Mentors underwent a structured mentoring course consisting of 4 four-hour group training sessions conducted by a dedicated sociologist during the first 6 months of the intervention program. The mentors had ongoing access to consultation, as needed, with the sociologist and the PIs by telephone. Each mentor was matched to a mentee by the PIs and sociologist, considering multiple personal and disease-related aspects of the pairs. Mentor-mentee meetings were held in person, by video or telephone calls, and instructed to occur at least once a week. All the participants were asked to respond to questionnaires evaluating coping skills, quality of life, self-efficacy, self-esteem, anxiety, and depression at 3 time points during the study (0, 3, 6 months). Disease activity assessed by SLEDAI and BILAG and medication adherence assessed by the ProMas were scored by the PIs at the same time points.
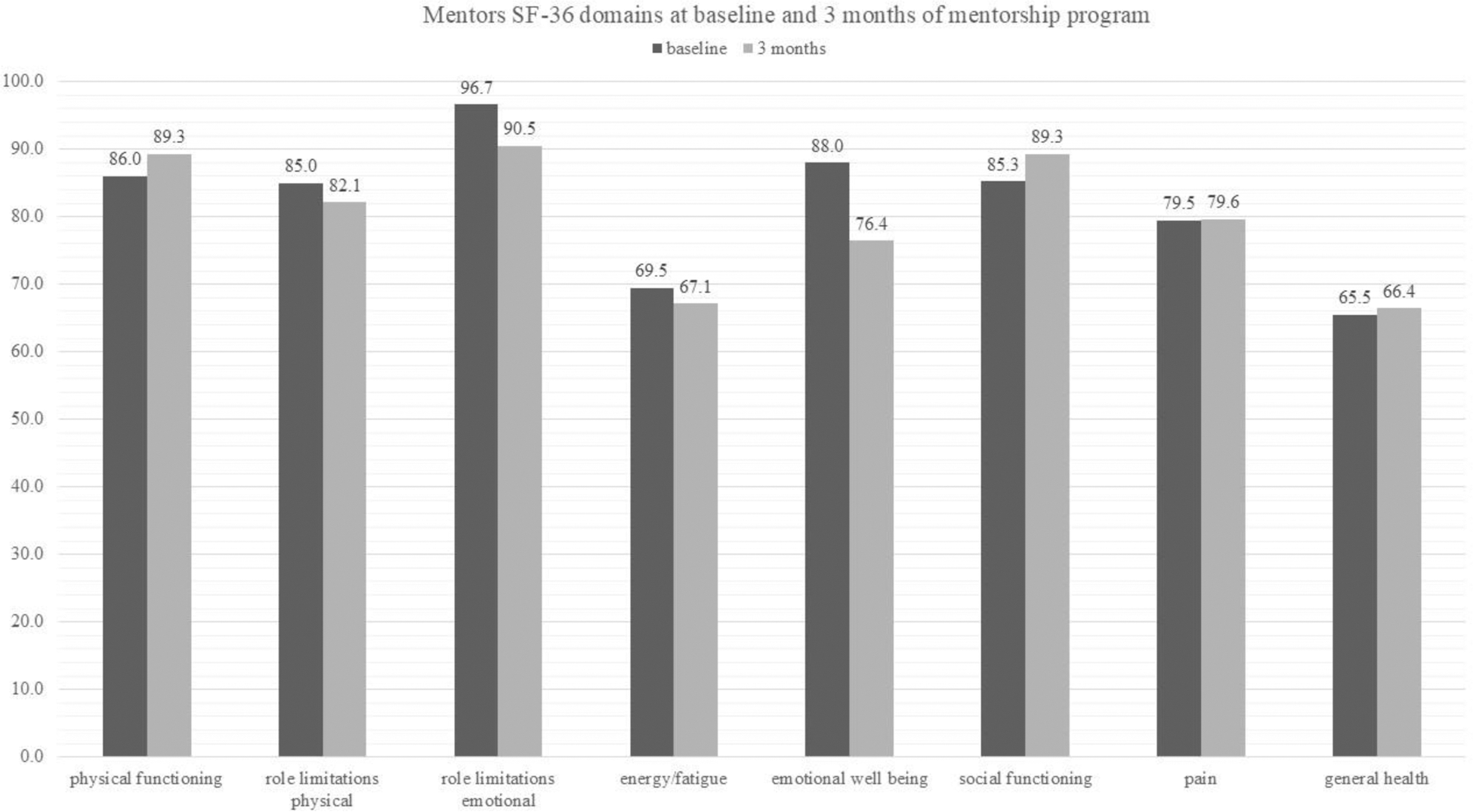
Results: Thirteen mentors started the mentoring program; one mentor left the program due to a stroke, and one mentee chose to stop after one call with her mentor. Twelve pairs completed 6 months of the intervention. Mentors were older than mentees, had lower disease activity, and took less prednisone. ProMAS scores of the mentors at baseline were significantly higher than those of the mentees, indicating better medication adherence (p=0.05) (Table 1). After 6 months of the intervention, mentors expressed numerous positive feelings of mutual connection , friendship, (“an open communication channel”, “a powerful connection”), motherhood (“the mentee was like my child”); reciprocation (“what I gave her, I got back”); empathy (“I found it easy to understand her difficulties”, “my mentee has similar goals as mine”, “reassured knowing that other people are going through similar things”, “my mentee was my mirror”, “I saw another perspective of my own coping”); kinship (“I enjoyed meeting more women like me”); altruism (“I felt like a hero, I felt strong”, “I contributed from my experience”, “The ability to give fulfilled me”, “doing this is a blessing”) and positive wellbeing (“the process made me appreciate my day-to-day life and the fact that I respond well to the medications”, “each meeting fulfilled me”, “the mentorship helped me and made me feel good”). Nevertheless, three mentors experienced challenges in the mentorship process: one did not succeed in establishing a connection with two different mentees, another felt that her mentee did not confide in her and that she was not helping her, and one mentee decided to stop the program after one meeting. The mentor was paired with a new mentee with a successful experience. SF-36 functional and emotional domain scores were high, while energy/fatigue, pain, and general health scores were moderate at baseline, with no statistically significant change at 3 months (Figure 1). Emotional well-being declined numerically at 3 months due to a decrease in the scores of two mentors, with difficulties not related to their disease or the mentorship program.
Demographic and clinical characteristics of mentors and mentees
| Mentors n=12 (%) | Mentees n=12 (%) | pV | |
|---|---|---|---|
| Age | 43.75±7.57 | 36.17±11.7 | 0.14 |
| Female | 12 (100) | 11 (91.67) | 0.31 |
| Baseline medications | HCQ: 12 (100)
| HCQ: 11 (91.67)
| |
| Baseline prednisone dose | 2±5.54 | 8.75±13.25 | 0.13 |
| 3 M prednisone dose | 1.92±5.51 | 2.92±3.2 | 0.61 |
| 6 M prednisone dose | 1.75±5.51 | 2.32±2.56 | 0.77 |
| Baseline SLEDAI2K | 3.67±2.9 | 5.82±4.39 | 0.2 |
| 3 M SLEDAI2K | 3.83±3.1 | 4.45±3 | 0.65 |
| 6 M SLEDAI2K | 3.5±2.06 | 4.2±3.03 | 0.57 |
| Baseline ProMAS* | 10.7±2.9 | 6.8±3.66 | 0.05 |
| 3 M ProMAS | 10.43±2.77 | NA |
* ProMAS categories: 0-4 low, 5-9 medium-low, 10-14 medium-high, 15-18 high
Conclusion: An interventional approach to target adherence in SLE is much needed. In this mentorship program, the mentors reported positive feelings of empowerment, suggesting that this type of intervention shows promise for both mentors and mentees. Baseline and 3-month ProMAS scores disclosed medium-high adherence of the mentors. Six-month results regarding the mentees’ and the mentors’ outcomes will be reported at the completion of the 12-month program.
REFERENCES: NIL.
Acknowledgements: This study was supported by AstraZeneca.The company was not involved in the planning or organization of the mentorship program, aquisition or analysis of the data.
Disclosure of Interests: Tali Eviatar AstraZeneca, GSK, AstraZeneca, Merav Tshuva AstraZeneca, Victoria Furer: None declared , Chen Avaki: None declared , Tali Ofir-Dovrat: None declared , Marina Anouk: None declared , Ori Elkayam: None declared , Daphna Paran AstraZeneca, AstraZeneca.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (