

Background: Systemic lupus erythematosus (SLE) is a chronic, inflammatory, autoimmune disease that can impact single or multiple organ systems [1]. Clinical and immunologic manifestations vary widely across patients, making this heterogenous disorder difficult to diagnose and treat [1, 2]. Understanding how a treatment may affect different subgroups of patients may improve management of this complex condition. Upadacitinib, an oral selective Janus kinase inhibitor, significantly reduced disease activity compared with placebo in a phase 2 trial of patients with moderately to severely active SLE [3]. Here, we evaluated the achievement of efficacy measures of disease activity and treatment response to upadacitinib vs placebo in patients stratified by baseline demographics and clinical characteristics.
Objectives: To evaluate potential differences in treatment responses to upadacitinib vs placebo associated with baseline patient characteristics using efficacy measures of the Lupus Low Disease Activity State (LLDAS) and British Isles Lupus Assessment Group-Based Composite Lupus Assessment (BICLA).
Methods: The SLEek study was a phase 2, randomized, double-blind, placebo-controlled clinical trial that enrolled adults with moderately to severely active SLE (NCT03978520) [3]. This post hoc analysis included participants who were randomized to receive once-daily upadacitinib 30 mg or placebo for 48 weeks. We report the proportions of trial participants in each group who met the composite measures of disease improvement, LLDAS and/or BICLA. Participants were stratified into subgroups by sex, age, race, region, body mass index, medication use, clinical markers, and medical history. Missing data were handled with nonresponder imputation incorporating multiple imputation to handle missing data due to COVID-19. Between-group differences and 95% CIs were estimated according to the Cochran-Mantel-Haenszel test adjusted for baseline corticosteroid use (≤10 mg or >10 mg), Systemic Lupus Erythematosus Disease Activity Index 2000 score at screening (<10 or ≥10), baseline interferon score (high, low, or not applicable), and baseline immunosuppressant use (yes or no). As an exploratory assessment, no inferential statistics were conducted, and the reported data are descriptive.
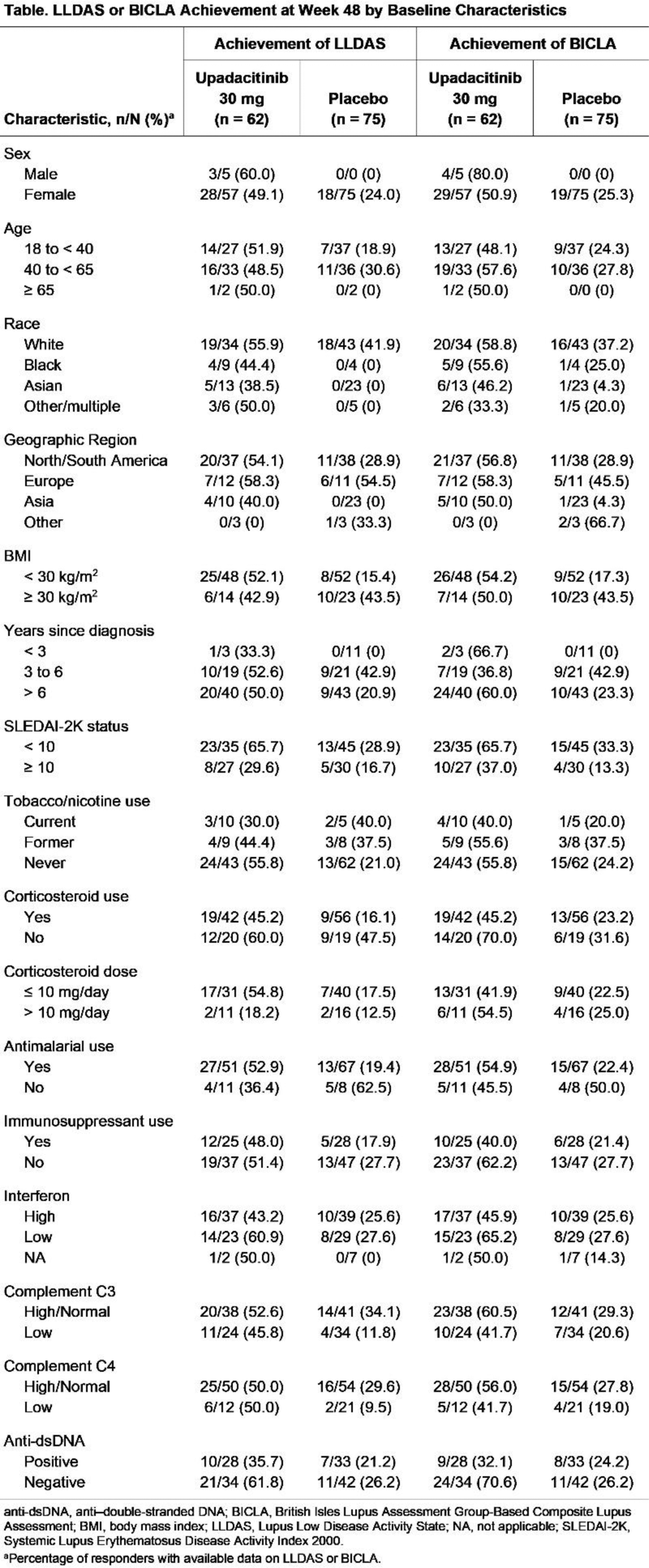
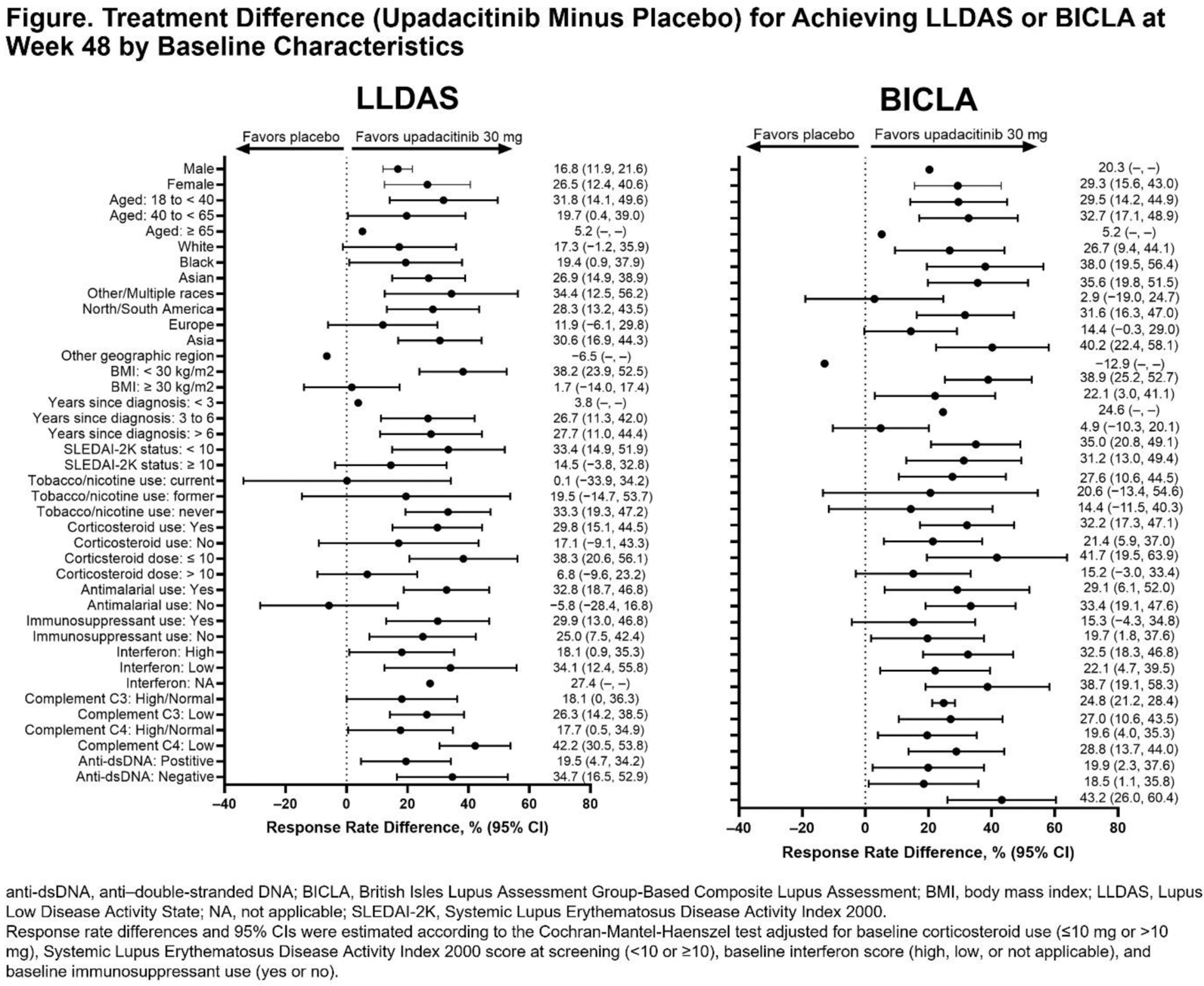
Results: A total of 137 participants (upadacitinib 30 mg, n = 62; placebo, n = 75) were included in this analysis. Most participants were White (56.2%) and female (96.4%). As previously published, at week 48, 31 participants (50.0%) treated with upadacitinib 30 mg and 18 participants (24.0%) treated with placebo achieved LLDAS; 33 participants (53.2%) treated with upadacitinib 30 mg and 19 participants (25.3%) treated with placebo achieved BICLA (3). Stratification into subgroups by baseline demographics and clinical characteristics revealed that a numerically higher proportion of participants in most subgroups treated with upadacitinib 30 mg vs placebo achieved LLDAS and/or BICLA (Table 1), with treatment differences (estimated by the Cochran-Mantel-Haenszel test) consistent with potential benefits of upadacitinib 30 mg across most subgroups (Figure 1).
Conclusion: After 48 weeks of treatment, participants with SLE treated with upadacitinib 30 mg were more likely than those receiving placebo to achieve LLDAS and/or BICLA across various baseline demographics and clinical characteristics; however, the interpretation of data in this study is limited by sample size. These findings, if corroborated in further studies, may support the potential benefit of upadacitinib treatment in a broad spectrum of patients with SLE despite the heterogeneity of the disorder.
REFERENCES: [1] Lazar S and Kahlenberg JM. Ann Rev Med . 2023;74:339–352.
[2] Fava A and Petri M. J Autoimmun. 2019;96:1–13.
[3] Merrill JT, et al. Arthritis Rheumatol. 2024;76:1518–1529.
Table 1.
Acknowledgements: AbbVie funded this study and participated in the study design, research, analysis, data collection, interpretation of data, reviewing, and approval of the abstract. All authors had access to relevant data and participated in the drafting, review, and approval of this abstract. No honoraria or payments were made for authorship. AbbVie and the authors thank all the trial investigators and the patients who participated in this clinical trial. Medical writing support was provided by Haley F Spencer, PhD, of JB Ashtin, and funded by AbbVie.
Disclosure of Interests: Marta Mosca reports receiving honoraria and/or consulting fees from AbbVie, AstraZeneca, Biogen, Bristol Myers Squibb, GlaxoSmithKline, Lilly, Novartis, Otsuka, and UCB; and has participated in safety boards for Idorsia, Karen H. Costenbader reports receiving honoraria from AbbVie, Amgen, AstraZeneca, Bain Capital, Inc, Biogen, Bristol Myers Squibb, Cabaletta Bio, GlaxoSmithKline, Neurtrolis, and UCB, and receiving research support from Gilead and Merck, Amit Saxena has served as a consultant or advisor for AbbVie, Amgen, AstraZeneca, Aurinia, Bristol Myers Squibb, and Synthekine, Michelle Petri: None declared , Andrea Rubbert-Roth reports receiving honoraria from AbbVie, Bristol Myers Squibb, Celgene, Chugai, Janssen, Lilly, MSD, Novartis, Roche, Sanofi, and UCB, Karim Masri may hold AbbVie stock or stock options, is a full-time employee of AbbVie, Christopher D Saffore may hold AbbVie stock or stock options, is a full-time employee of AbbVie, Ling Cheng may hold AbbVie stock or stock options, is a full-time employee of AbbVie, Joan Merrill has served as a consultant or advisor for AbbVie, Alexion, Almiral, Alumis, Amgen, AstraZeneca, Aurinia, Biogen, Bristol Myers Squibb, EMD Serono, Equillium, Genentech, Gilead, GlaxoSmithKline, Kezar, Lilly, Merck, Novartis, Ono, Remegen, Sanofi, Takeda, Tenent, UCB, Veloxis, and Zenas, has received research support from AstraZeneca, Aurinia, Bristol Myers Squibb, and GlaxoSmithKline.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (