

Background: Avacopan, the first orally active complement 5a receptor (C5aR) inhibitor, has emerged as a promising therapeutic option for ANCA-associated vasculitis (AAV). By selectively targeting C5aR, avacopan reduces neutrophil activation and vascular inflammation. ADVOCATE study have demonstrated that avacopan effectively induces and sustains remission in AAV while minimizing glucocorticoid-related toxicities [1]. Despite these promising results, the efficacy and safety of avacopan in real-world clinical settings remain unclear. Recent studies and case reports, have raised concerns regarding avacopan-induced severe liver injury, such as vanishing bile duct syndrome, particularly in Japan [2, 3].
Objectives: To assess the safety profile and incidence of adverse events associated with avacopan in AAV using data from a nationwide registry in Japan.
Methods: This multicenter retrospective cohort study analyzed data from the Japan Collaborative Registry of ANCA-Associated Vasculitis (J-CANVAS) between January 2017 and March 2023. Patients with granulomatosis with polyangiitis and microscopic polyangiitis (MPA), newly diagnosed or with severe relapse, were included. Disease classification followed the 2012 International Chapel Hill Consensus Conference definitions and the European Medicines Agency algorithm. Baseline clinical characteristics, including the Birmingham Vasculitis Activity Score (BVAS) 3.0, treatments, and prognoses (serious adverse events [SAEs], serious infections), were documented. The follow-up period ranged from the disease onset to death, loss to follow-up, severe relapse, or March 2024. Weighted Cox proportional hazard models were used to compare the risk of serious infections between the avacopan-treated and the non-avacopan-treated groups. The imbalance of clinical characteristics between the two groups was adjusted by overlap weighting with propensity scores calculated from age, sex, diagnosis, diabetes mellitus, smoking, pulmonary involvement, renal involvement, BVAS, C-reactive protein, intravenous cyclophosphamide use, rituximab use, methylprednisolone pulse use, the initial dose of prednisolone, and use of trimethoprim-sulfamethoxazole. The analysis employed the average treatment effect in the overlap population to estimate treatment effects and minimize comparison bias. After weighting, the balance in these variables was assessed by the standardized mean differences. We also collected data on SAEs associated with avacopan and analyzed the risk factors for avacopan-induced liver dysfunction in the avacopan-treated group. SAEs were defined as events leading to avacopan discontinuation, and serious infections were defined as those requiring or prolonging hospitalization. Baseline characteristics were compared using Wilcoxon’s rank-sum test for continuous variables and Fisher’s exact test for categorical variables between patients with and without SAEs. Kaplan-Meier analysis was performed to estimate SAE-free survival.
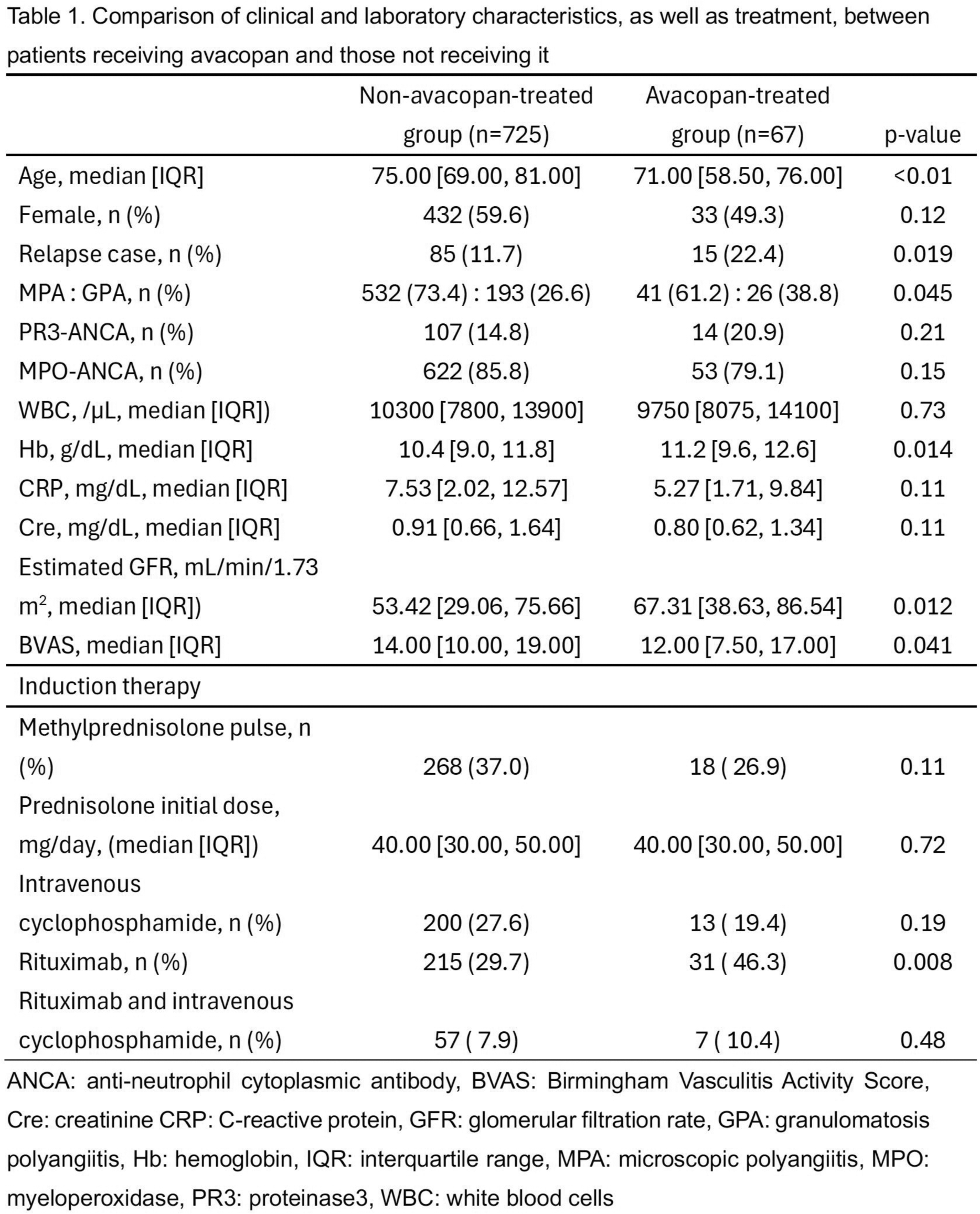
Results: Among 792 AAV patients, 67 received avacopan, including 18 as induction therapy. Other reasons for initiating avacopan included persistence or worsening of disease activity (27 cases) and elevated ANCA titers (12 cases). The baseline characteristics were summarized in Table 1. The avacopan-treated group had younger age (median: 71 vs. 75 years, p < 0.01), a higher proportion of relapse cases (15 [22.4%] vs. 85 [11.7%] cases, p = 0.019), less MPA cases (41 [61.2%] vs. 532 [73.4%] cases, p = 0.045), higher eGFR (67.31 vs. 53.42 mL/min/1.73 m², p = 0.012), higher BVAS (14.0 vs. 12.0, p = 0.041), and more rituximab use (31 [46.3%] vs. 215 [29.7%] cases, p = 0.008) than those of the non-avacopan-treated group. There were no significant differences in MPO-ANCA positivity (53 vs. 622 cases, p = 0.15). Serious infections occurred in 2 patients in the avacopan-treated group and 123 patients in the non-avacopan-treated group. The adjusted hazard ratio for the incidence of serious infections was comparable between the two groups (HR: 0.44, 95% CI: 0.11–1.76; Figure 1a). Twelve SAEs were identified in the avacopan-treated group, with liver dysfunction being the most common (8 cases). Kaplan-Meier analysis of SAE-free survival is shown in Figure 1b. Patients who developed avacopan-induced liver dysfunction were significantly older than those without avacopan-induced liver dysfunction (median: 75.5 vs. 69.0 years, p = 0.037).
Conclusion: This study provides real-world data on the safety profile of avacopan in Japanese patients with AAV. The risk of serious infections in the avacopan-treated patients was comparable to non-avacopan-treated patients. However, liver dysfunction emerged as the most frequent SAE of avacopan. Patients with liver dysfunction were older than patients without liver dysfunction. These findings highlight the importance of careful monitoring of liver function, especially in older patients receiving avacopan.
REFERENCES: [1] N Engl J Med. 2021;384:599-609.
[2] J Rheumatol. 2024;51:1146-1148.
[3] Rheumatology (Oxford). 2024:keae689.
Acknowledgements: NIL.
Disclosure of Interests: Tomohisa Uchida Asahi Kasei Pharma Co., Ltd., Chugai Pharmaceutical Co., Ltd, Mitsubishi-Tanabe Pharma Co., Shoichi Fukui: None declared , Kunihiro Ichinose Chugai, Nihon-Shinyaku, AbbVie, Asahi-Kasei Pharma, Astellas, AstraZeneca, Ayumi, Bristol Myers Squibb, Boehringer Ingelheim, Chugai, Eisai, Eli Lilly, Glaxo SmithKline, Nihon-Shinyaku, Otsuka Pharmaceutical, Pfizer, Taisho, Tanabe-Mitsubishi, UCB, Otsuka Pharmaceutical, Asahi-Kasei Pharma, Asahi-Kasei Pharma, Chugai, Otsuka, Japan Blood Products Organization, Hiroyuki Ide Eisai, Yoshika Tsuji: None declared , Toshimasa Shimizu: None declared , Masataka Umeda Otsuka Pharmaceutical Co., Ltd., AstraZeneca plc, AbbVie GK, Asahi Kasei Pharma Corporation, GlaxoSmithKline plc., Remi Sumiyoshi: None declared , Takashi Igawa: None declared , Tomohiro Koga: None declared , Shin-ya Kawashiri: None declared , Naoki Iwamoto: None declared , Mami Tamai: None declared , Tomoki Origuchi: None declared , Satoshi Omura Asahi Kasei, UCB Japan, Janssen, Daiki Nakagomi AbbVie, Asahi-Kasei Pharma, AstraZeneca, Chugai, Eisai, Novartis, Taisho, Taisho, Asahi-Kasei Pharma, Chugai, Yoshiyuki Abe Asahi-Kasei Pharma, Chugai, Eisai, Glaxo SmithKline, Kissei, Makoto Wada: None declared , Naoho Takizawa Chugai, AbbVie, Asahi-Kasei Pharma, Astellas, AstraZeneca, Boehringer Ingelheim, Chugai, Eisai, Eli Lilly, Glaxo SmithKline, Janssen, Pfizer, Taisho, Tanabe-Mitsubishi, UCB, Atsushi Nomura: None declared , Yuji Kukida AbbVie, Asahi Kasei Pharma, Boehringer Ingelheim, Chugai, Daiichi-Sankyo, Eisai, Mitsubishi Tanabe and Taisho, Naoya Kondo: None declared , Yamano Yasuhiko Boehringer Ingelheim、Bristol Myers Squibb、GlaxoSmithKline, Hirosuke Takagi Janssen, Kissei, Taisho, AbbVie, Asahi-Kasei Pharma, Astellas, Eli Lilly, Kissei, Tanabe-Mitsubishi, UCB, Koji Endo: None declared , Shintaro Hirata AbbVie, Asahi-Kasei Pharma, Astellas, AstraZeneca, Ayumi, Bristol Myers Squibb, Boehringer Ingelheim, Chugai, Daiichi-Sankyo, Eisai, Eli Lilly, Gilead Sciences, Glaxo SmithKline, Janssen, Nihon-Shinyaku, Novartis, Otsuka Pharmaceutical, Pfizer, Taisho, Tanabe-Mitsubishi, UCB, Eisai, Asahi-Kasei Pharma, Chugai, Otsuka, Taisho, Naoto Azuma: None declared , Tohru Takeuchi AbbVie, Asahi-Kasei Pharma, AstraZeneca, Boehringer Ingelheim, Chugai, Eisai, Eli Lilly, Glaxo SmithKline, Janssen, Nihon-Shinyaku, Otsuka Pharmaceutical, Taisho, Tanabe-Mitsubishi, Taisho, Asahi-Kasei Pharma, Chugai, Boehringer Ingelheim, Kazuro Kamada: None declared , Ryo Yanai: None declared , Yusuke Matsuo: None declared , Yasuhiro Shimojima: None declared , Ryo Nishioka Kissei Pharmaceutical Co., Ltd, Ryota Okazaki Eli Lilly, Asahi-Kasei Pharma, Tomoaki Takata Kyowa Kirin, Kyowa Kirin, Chugai Pharmaceutical, Mitsubishi Tanabe Pharma, Mayuko Moriyama Eisai, Eisai, Eli Lilly, Taisho, Tanabe-Mitsubishi, Ayuko Takatani: None declared , Yoshia Miyawaki: None declared , Tsuyoshi Shirai: None declared , Hiroaki Dobashi Abbvie, Eli Lilly, Astellas, Ayumi Pharmaceutical, Chugai Pharmaceutical, Eisai Pharmaceutical, Asahi Kasei Pharma, UCB Pharmaceutical, and KISSEI Pharmaceutical, Asahi Kasei Pharma, Chugai Pharmaceutical, and AbbVie Japan, Takafumi Ito: None declared , Isao Matsumoto: None declared , Toshihiko Takada: None declared , Toshiko Ito-Ihara: None declared , Takashi Kida: None declared , Nobuyuki Yajima: None declared , Takashi Kawaguchi: None declared , Yutaka Kawahito Pfizer Japan Inc., Asahi Kasei Pharma Co., Ltd., AbbVie Japan GK, Astellas Pharma Inc., AstraZeneca plc., Ayumi Pharmaceutical Co., Boehringer Ingelheim Japan, Inc., Bristol Myers Squibb Co., Ltd., Chugai Pharmaceutical Co., Ltd., Eisai Co., Ltd., Daiichi Sankyo., GlaxoSmithKline K.K., Janssen Pharmaceutical K.K., Kissei Pharmaceutical Co., Ltd., Mitsubishi-Tanabe Pharma Co., Nippon Kayaku Co., Ltd., Novartis Pharma K.K., Pfizer Japan Inc., Taisho Toyama Pharmaceutical Co., Ltd., UCB Japan Co. Ltd. and Viatris Pharmaceuticals Japan Inc., Asahi KASEI Pharma Co., Ltd., Boehringer Ingelheim Japan, Inc., Chugai Pharmaceutical Co., and Eisai Co., Ltd., Atsushi Kawakami AbbVie, Asahi Kasei, Chugai, Eisai, Eli Lilly, ONO, Taisho, Boehringer Ingelheim, Pfizer, AbbVie, Asahi Kasei, AYUMI, Boehringer Ingelheim, Bristol-Myers Squibb, Celltrion, Chugai, Daiichi Sankyo, Gilead, Neopharma, ONO, Taisho.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (