

Background: Avacopan, a selective C5aR1 inhibitor, has demonstrated efficacy and safety in its development program in patients with ANCA-associated vasculitis (AAV) (specifically granulomatosis with polyangiitis [GPA] and microscopic polyangiitis [MPA]) and has been approved in combination with rituximab or cyclophosphamide for the treatment of adults with severe active GPA/MPA.
Objectives: To describe results from a physician survey assessing avacopan usage patterns in adults with severe active GPA/MPA in daily clinical practice.
Methods: A quantitative online survey was completed by physicians in Germany and the UK from April 17 to May 20, 2024. Respondents were required to be qualified in their primary specialty (nephrology, rheumatology, internal medicine with a secondary qualification in rheumatology or nephrology [Germany only], pulmonology, or ear, nose and throat [ENT] medicine) for 2 to 40 years, spend ≥60% of their time in direct patient care, be responsible for making treatment choices for adults with GPA/MPA, and have personally managed ≥5 patients with AAV in the last 12 months, including ≥2 with severe active GPA/MPA. Each respondent completed patient record forms for 2-3 GPA/MPA patients (≥18 years) with severe active disease (new-onset or relapsed) as of the most recent induction therapy, currently receiving induction therapy, or previously severe active now on maintenance therapy. Statistical significance testing was conducted with a type 1 error rate (alpha) of 5% using independent t-tests for means testing and independent p-tests for proportion testing. The survey included additional attitudinal/perceptual questions on GPA/MPA patient categorisation, ‘active’ definition, guidelines followed, and treatment attitudes, responses to which will be presented elsewhere.
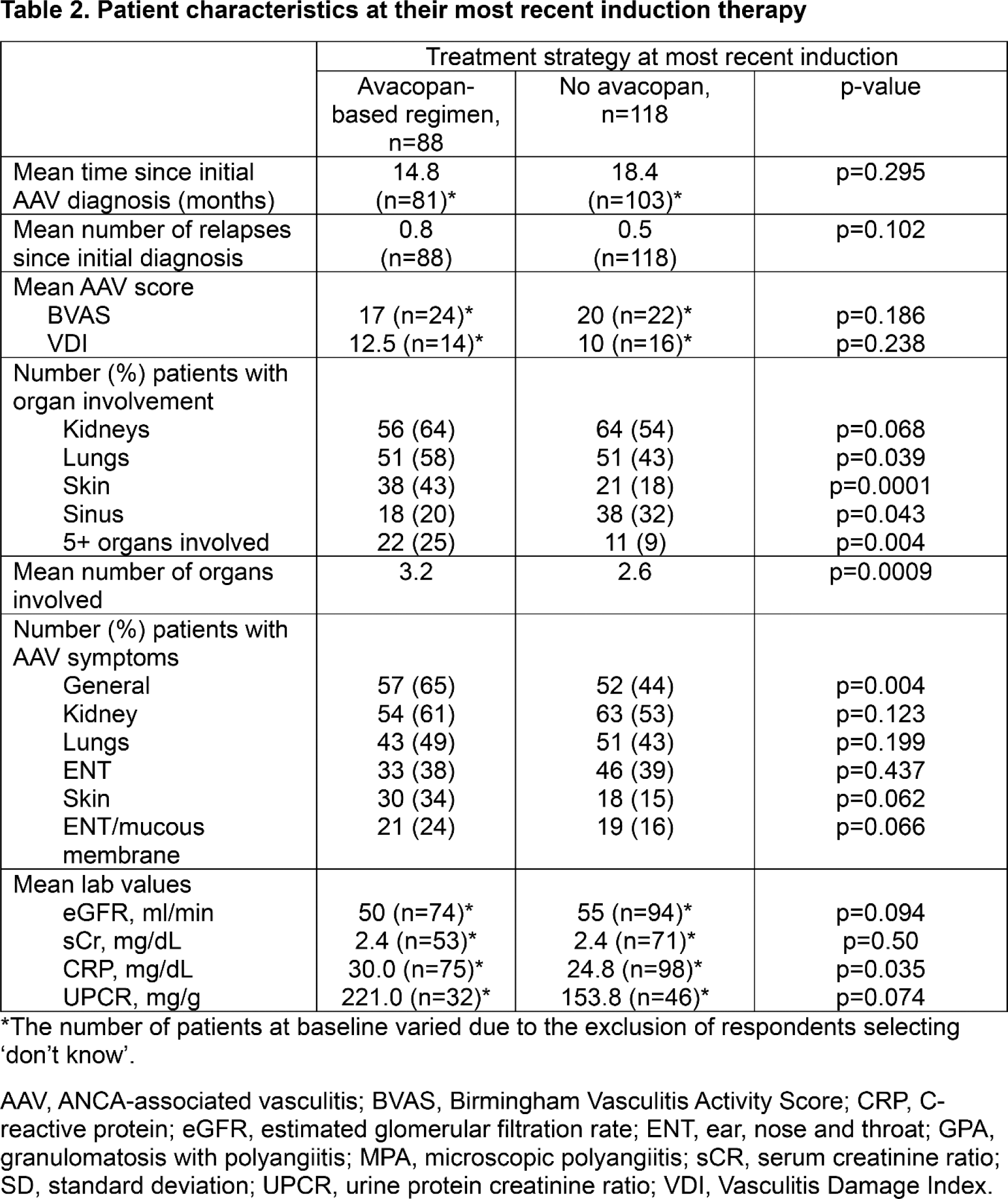
Results: Survey response rate was 51% (63% in Germany; 43% in the UK). A total of 206 patient record forms were completed by 81 respondents (40 in Germany; 41 in the UK), including 26 nephrologists, 39 rheumatologists, 9 pulmonologists and 7 ENT specialists. At their most recent induction, 93/206 patients (45.2%) were receiving avacopan-based therapy. Of these, 5 (5.4%) were continuing previous avacopan treatment and 88 (94.6%) were newly initiated. Significantly more patients in the avacopan group (vs. the non-avacopan group) had depression (25% vs 11%, p=0.008) or BMI >35 kg/m 2 (17% vs 7%, p=0.026) at their initial AAV diagnosis (Table 1). The proportion of patients with other pre-selected comorbidities at the time of AAV diagnosis, including hypertension, dyslipidemia, Type 2 diabetes, and anxiety, was similar between treatment groups. Significantly more patients in the avacopan vs. the non-avacopan group had multiple (5+) organ involvement (25% vs 9%; p=0.004), lung involvement (58% vs 43%, p=0.04), skin involvement (43% vs 13%, p=0.001), and/or presented with ‘general’ (65% vs. 44%, p=0.004) or skin (34% vs 15%, p=0.002) symptoms at the start of their most recent induction (Table 2). Patients receiving avacopan-based therapy also had significantly higher CRP values (30 mg/dL vs 24.8 mg/dL, p=0.035) and showed a trend towards lower eGFR values (50 ml/min vs 55 ml/min, p=0.094) than those treated without avacopan. Results were consistent in Germany and the UK.
Conclusion: Survey results (patient record forms) suggest broad use of avacopan as part of induction therapy in adults with severe active (new-onset or relapsed) GPA/MPA in Germany and the UK. A significantly higher proportion of patients receiving avacopan- (as opposed to non-avacopan-) based therapy had depression or were overweight at their initial AAV diagnosis. Additionally, a significantly greater proportion of these patients had multiple organ involvement, lung or skin involvement, presented with ‘general’ or skin symptoms, and/or had higher CRP values at their most recent induction. Limitations of this research include potential bias of the respondents, small sample size and incompleteness of the data in certain fields on patient record forms.

REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Udo Schneider Abbvie, Amgen, Astra Zeneca, Boehringer Ingelheim, BMS, CSL Vifor, GSK, Kyverna, Novartis, Pfizer, AstraZeneca, Boehringer Ingelheim, CSL Vifor, GSK, Wolfram-Johannes Jabs CSL Vifor, Novartis, Sanofi Genzyme, 2004-2007: Fa. Biotest, Alexion, Sanofi Genzyme, Stadapharm, Paul Warner CSL Vifor, CSL Vifor (current), Tamara Popov CSL Vifor, CSL Vifor (current), Roche, Merck.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (