

Background: RGB-19, a proposed biosimilar to tocilizumab, is a monoclonal antibody that competitively inhibits the binding of interleukin-6 (IL-6) to its receptor.
Objectives: To demonstrate the pharmacokinetic (PK) equivalence and compare the pharmacodynamics (PD), safety, and immunogenicity of RGB-19 to its reference (hereafter tocilizumab), in healthy adults.
Methods: In this Phase 1 cross-over study, healthy Japanese adult males, with a body mass index of ≥18.5 to <25.0 kg/m 2 and no previous exposure to tocilizumab were randomly allocated 1:1 to Sequences A or B to receive a single subcutaneous (SC, upper arm) injection of either RGB-19 162 mg or tocilizumab 162 mg in the first period. As per the two-period, (Period 1: Day -1 to 42; Period 2: Day 42 to 85) two-sequence cross-over design, participants in Sequence A received RGB-19 in Period 1 and switched to tocilizumab in Period 2, while participants in Sequence B received tocilizumab in Period 1 and RGB-19 in Period 2. The primary PK endpoints were the maximum serum drug concentration (C max ) and area under the curve from immediately prior to administration, to infinity (AUC inf ), both assessed using an analysis of variance model. Additional secondary PK, PD, safety (minimum 85-day follow-up), and immunogenicity outcomes were also assessed.
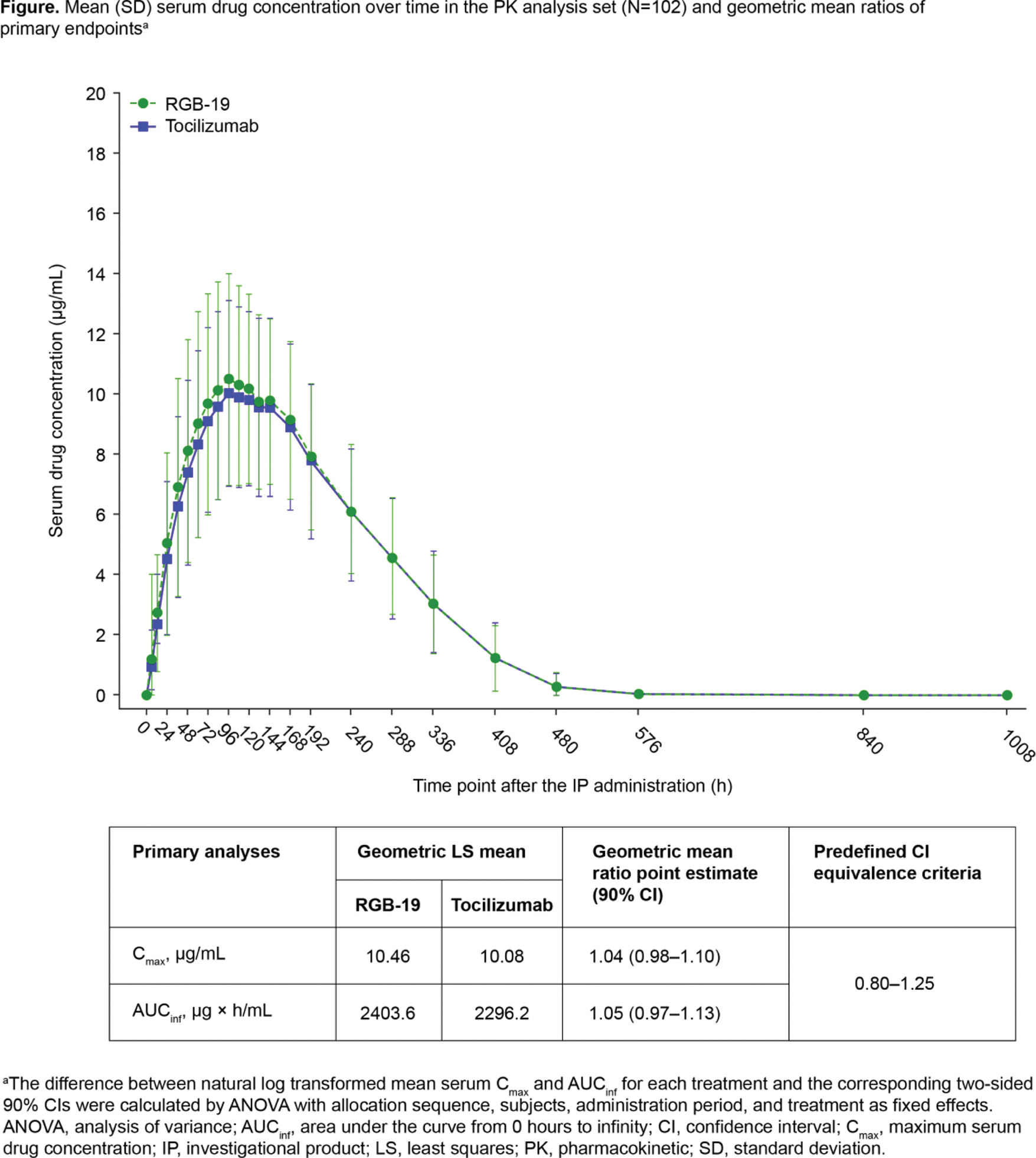
Results: A total of 110 participants aged 20–40 years were randomised in Period 1 and treated (Sequence A and B n=55 each). Baseline characteristics were well balanced between the two treatment groups. PK equivalence was demonstrated between RGB-19 and tocilizumab, with results for C max and AUC inf meeting the prespecified equivalence criteria (Figure 1). No clinically significant differences between treatments were observed in key secondary PK and PD parameters (Table 1). No significant sequence effects were noted, with unquantifiable serum drug concentration prior to drug administration in Period 2. PK parameter variability was comparable between treatment periods, suggesting no carry-over effect. The evaluation of bioequivalence was not affected by period effects. RGB-19 was well tolerated, with a similar pattern of treatment-emergent adverse events (AEs) to tocilizumab. The most frequently reported AE in both groups was decreased neutrophil count, no difference was noted in the incidence of infections or infestations between groups. There were no serious AEs or AEs leading to death in either group. Immunogenicity outcomes were similar for both treatments, with a comparable incidence of anti-drug antibody (ADA) positivity (total number of ADA positive participants across both treatment periods: RGB-19 n=61; tocilizumab n=62). ADA status did not impact C max or AUC inf outcomes for participants in either group.
Conclusion: In this study, PK equivalence of the proposed biosimilar RGB-19 was demonstrated, with comparable PD, safety, tolerability and immunogenicity to that of reference tocilizumab.
REFERENCES: NIL.
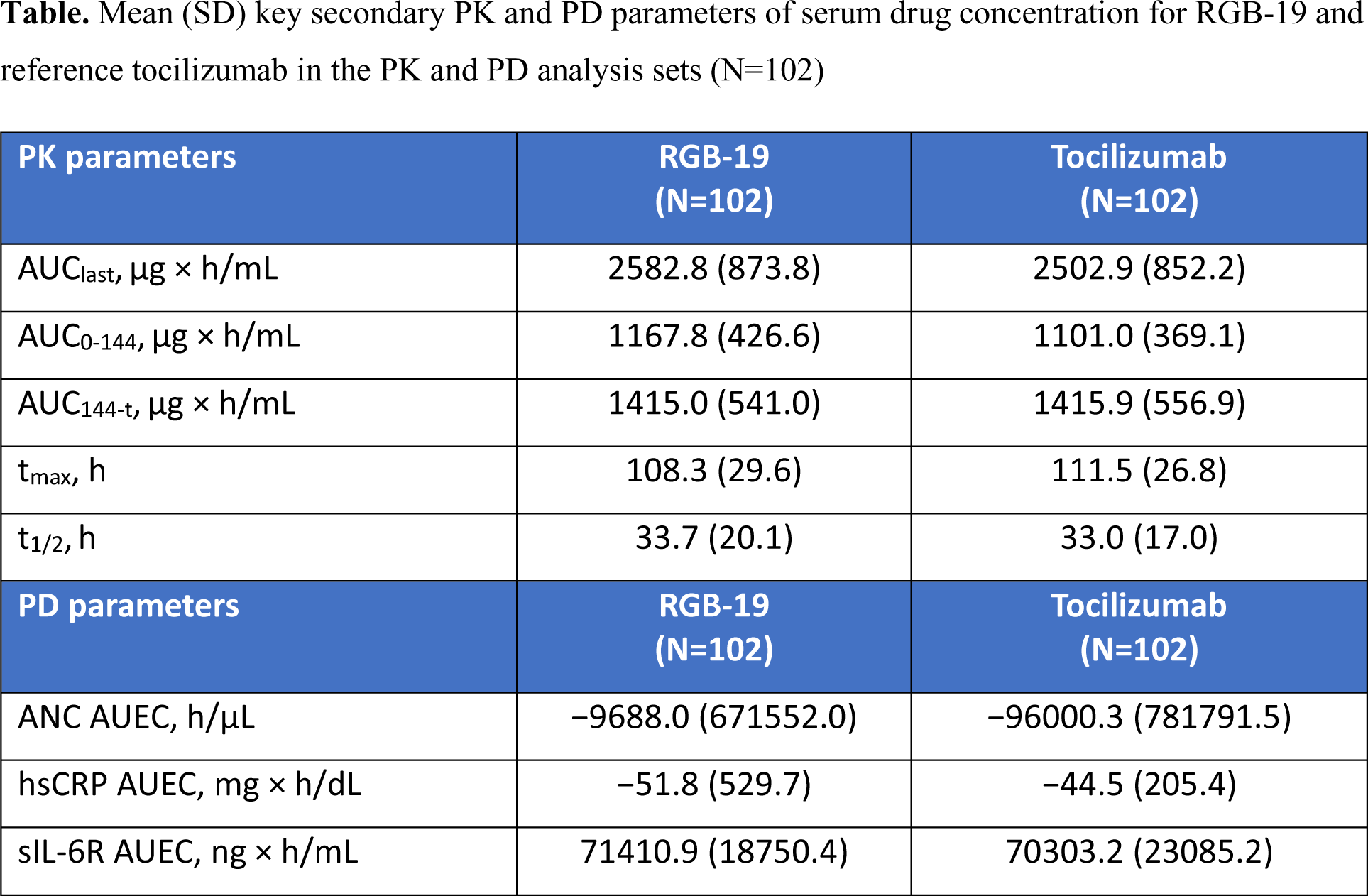
ANC, absolute neutrophil count; AUC 0-144 , area under the curve from 0 (immediately before administration) to 144 hours after administration; AUC 144-t , area under the curve from 144 hours after administration to the last quantifiable time; AUC last , area under the curve from 0 hours (immediately before administration) to the last quantifiable time; AUEC, area under the effect-time curve; hsCRP, high-sensitivity C-reactive protein; PD, pharmacodynamic; PK, pharmacokinetic; sIL-6R, soluble interleukin-6 receptor; SD, standard deviation; t 1/2 , elimination half-life; t max , time to maximum serum concentration.
Acknowledgements: Study funded by Gedeon Richter and Mochida Pharmaceutical. Medical writing support provided by Timothy Davies, PhD, of Avalere Health, and funded by Gedeon Richter.
Disclosure of Interests: Paul Emery has provided expert advice to Abbvie, Activa, Astra-Zeneca, BMS, Boehringer Ingelheim, Galapagos, Gilead, Immunovant, Janssen, Lilly, and Novartis, and contributed to clinical trials of Abbvie, BMS, Lilly, Novartis, and Samsung, Ernest Choy has received research grants, speaking fees, consultancies or honoraria from Abbvie, Bio-Cancer, Biocon, Biogen, Chugai Pharma, Eli Lilly, Fresenius Kai, Galapagos, Gedeon Richter, Gilead, Janssen, Pfizer, Sanofi, UCB and Viatris, Tomoko Hasunuma: None declared , Masato Okada has received speaking fees and/or honoraria from Astellas, Eli Lilly and Company, GSK, and Janssen, Zsofia Polgari is a Gedeon Richter employee, Karoly Horvát-Karajz is a Gedeon Richter employee, Gordana Dancer is a Gedeon Richter employee, Attila Kónya is a Gedeon Richter employee, Yusuke Karibe is a Mochida Pharmaceutical employee, Kazuya Uchida is a Mochida Pharmaceutical employee, Suguru Masuda is a Mochida Pharmaceutical employee, Joachim Kiefer is a Gedeon Richter employee, Gerd R. Burmester has received honoraria for lectures and consulting from Celltrion, Chugai, Fresenius, Gedeon Richter, and Sanofi.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (