

Background: Progressive pulmonary fibrosis (PPF) is known to be associated with high mortality, but there are few data on the course of PPF in patients with interstitial lung disease (ILD) due to systemic autoimmune rheumatic diseases (SARDs).
Objectives: To evaluate time to death or lung transplant among patients with SARDs and PPF.
Methods: The ILD-PRO Registry enrolled patients who had an ILD other than idiopathic pulmonary fibrosis, reticular abnormality and traction bronchiectasis (with or without honeycombing) on HRCT, and met criteria for ILD progression within the prior 24 months. Patients were followed prospectively while receiving usual care. The Kaplan–Meier method was used to analyze time to death or lung transplant over 24 months in patients with the following SARDs: rheumatoid arthritis (RA), systemic sclerosis (SSc), idiopathic inflammatory myopathies (IIMs), interstitial pneumonia with autoimmune features (IPAF), mixed or undifferentiated connective tissue disease (CTD), Sjögren’s disease, and other SARDs.
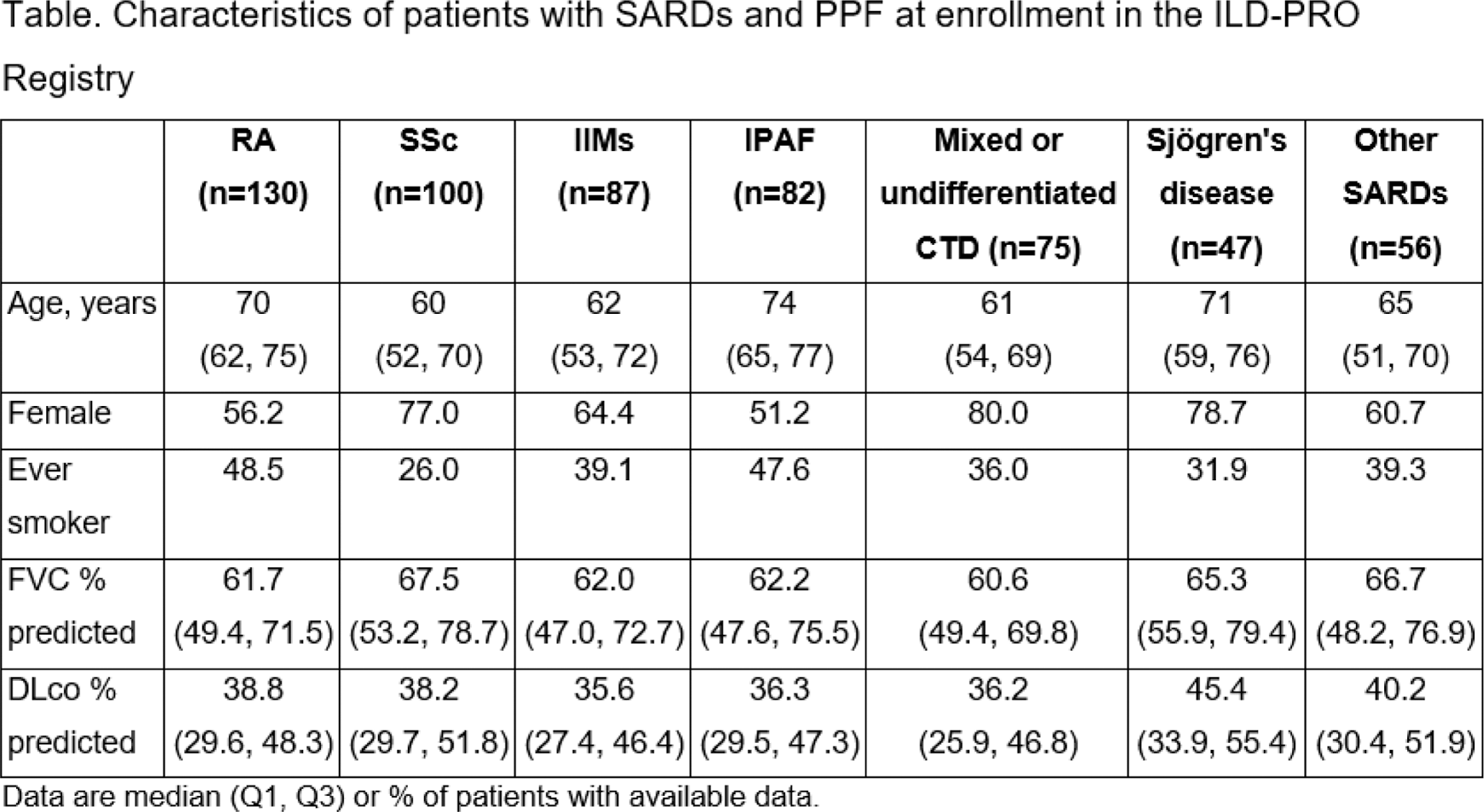
Results: A total of 577 patients in the ILD-PRO Registry had SARDs: 130 (22.5%) RA, 100 (17.3%) SSc, 87 (15.1%) IIMs, 82 (14.2%) IPAF, 75 (13.0%) mixed or undifferentiated CTD, 47 (8.1%) Sjögren’s disease, and 56 (9.7%) other SARDs. Patient characteristics at enrollment are summarized in the Table 1. The probability of transplant-free survival at 24 months was 60.8% for RA, 83.0% for SSc, 80.9% for IIMs, 69.3% for IPAF, 78.8% for mixed or undifferentiated CTD, 89.2% for Sjögren’s disease and 78.2% for other SARDs (Figure 1).
Conclusion: Patients with SARDs and PPF enrolled in the ILD-PRO Registry had a high probability of death or lung transplant over the following two years. The probability of transplant-free survival varied by type of SARD, with the worst survival in patients with RA and IPAF.
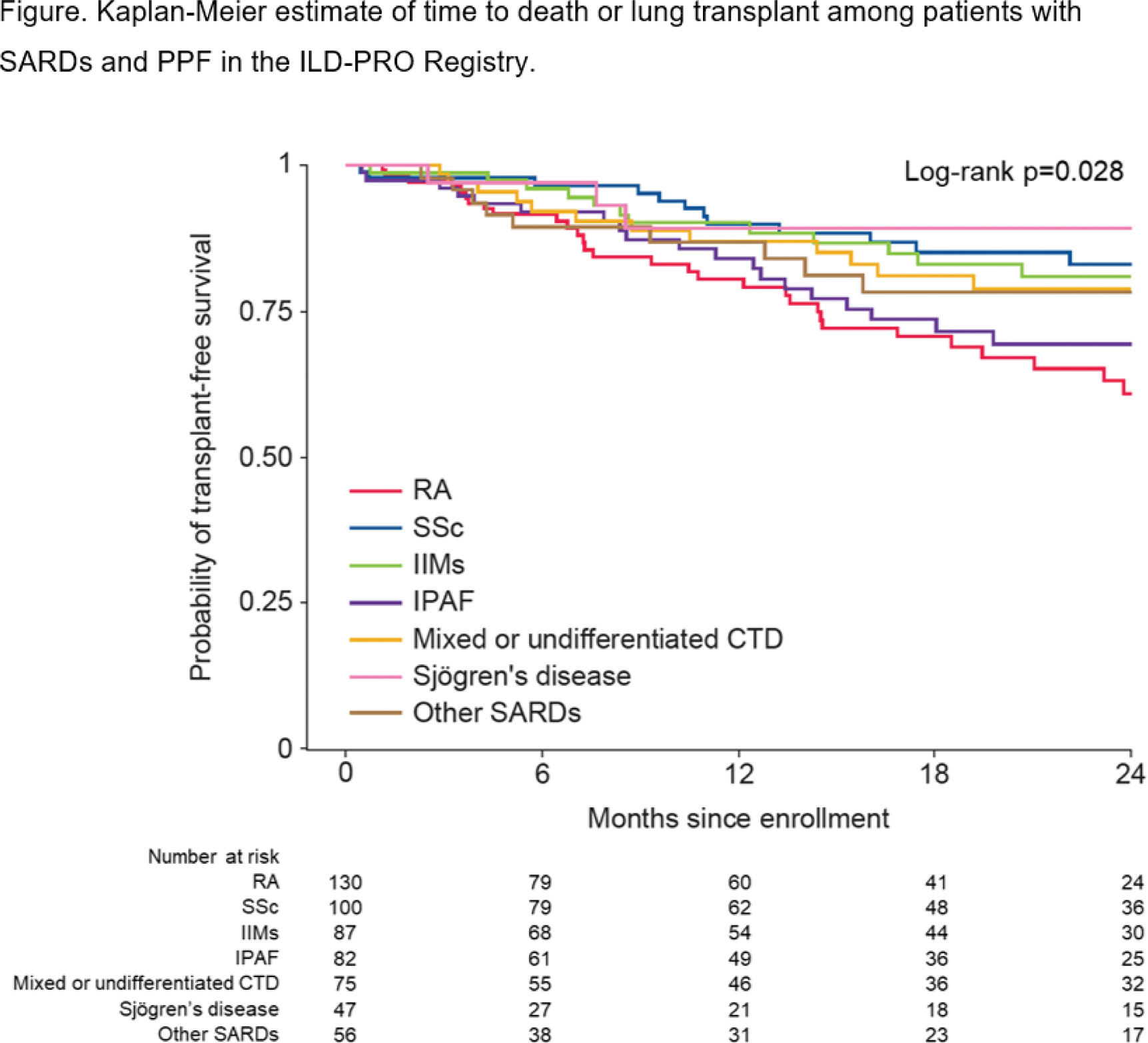
Table 1.
REFERENCES: NIL.
Acknowledgements: The IPF-PRO/ILD-PRO Registry is supported by Boehringer Ingelheim Pharmaceuticals, Inc. and run in collaboration with the Duke Clinical Research Institute and enrolling centers.
Disclosure of Interests: Sonali J Bracken is an employee of Duke University Medical Center, which is a site in the IPF-PRO/ILD-PRO Registry, Aparna C Swaminathan has served on an advisory board for United Therapeutics, has received grant support from Bristol Myers Squibb and Genentech and reports that Duke Clinical Research Institute (my employer) receives funding support from Boehringer Ingelheim Pharmaceuticals, Inc. to coordinate the IPF-PRO/ILD-PRO Registry, Jeremy M Weber reported that Duke Clinical Research Institute (my employer) receives funding support from Boehringer Ingelheim Pharmaceuticals, Inc. to coordinate the IPF-PRO/ILD-PRO Registry, Megan L Neely reported that Duke Clinical Research Institute (my employer) receives funding support from Boehringer Ingelheim Pharmaceuticals, Inc. to coordinate the IPF-PRO/ILD-PRO Registry, Scott M Palmer has served as a consultant for Sanofi, has received grant/research support from Boehringer Ingelheim, and reported that Duke Clinical Research Institute (my employer) receives funding support from Boehringer Ingelheim Pharmaceuticals, Inc. to coordinate the IPF-PRO/ILD-PRO Registry, Erin Wilfong has served as a consultant for AstraZeneca, Boehringer Ingelheim, Cabeletta Bio, Merck, and has received grant/research support from Boehringer Ingelheim, Ann Chauffe is an employee of Boehringer Ingelheim Pharmaceuticals, Inc., Elizabeth R Volkmann has served as a consultant for AbbVie, Boehringer Ingelheim, GlaxoSmithKline, and has received grant/research support from AstraZeneca, Atyr Pharma, Boehringer Ingelheim, GlaxoSmithKline, Horizon, Prometheus.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (