

Background: As up to 20% of patients with lupus nephritis (LN) develop end-stage kidney disease (ESKD) within 10 years of diagnosis [1], treating early may slow damage progression and prevent recurrent flares. This is further supported by evidence that kidney function improvements within 6 months of treatment predict favourable long-term outcomes [2]. Current EULAR 2023 [3], KDIGO 2024 [4] and ACR 2024 [5] recommendations for LN state that belimumab should be considered for initial triple therapy in many patients, but there may be hesitancy among some clinicians to include biologics such as belimumab in initial therapy.
Objectives: To evaluate the available clinical trial and real-world evidence for belimumab as an initial therapy in LN.
Methods: Narrative review guided by LN experts, inclusive of recent publications related to Phase 3 trials and real-world studies of belimumab therapy in LN.
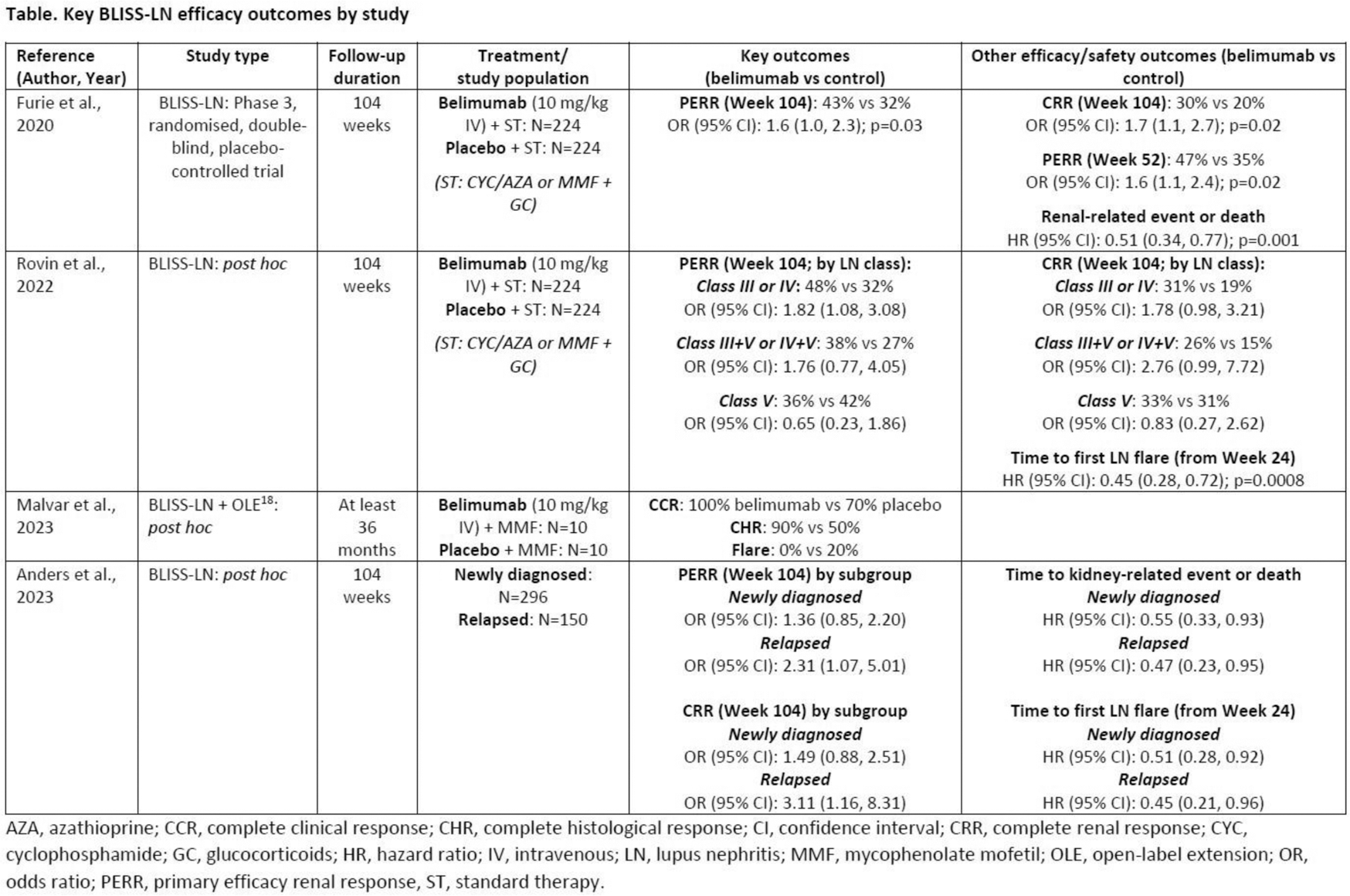
Results: The 2-year Phase 3 BLISS-LN study found patients receiving belimumab induction therapy (with standard therapy) achieved primary efficacy and complete renal responses (PERR and CRR, respectively) earlier than standard therapy alone, with differences observed as early as Week 12–24 [6]. BLISS-LN secondary analyses further supported the early impact of belimumab on outcomes relative to standard therapy, including reduced risk of recurrent flares (defined as impaired kidney function, increase in proteinuria, or kidney-related treatment failure) from Week 24 [7]. Further BLISS-LN post hoc analyses reported a higher proportion of patients achieving histologic remission with belimumab treatment [8], and newly diagnosed and relapsed patients receiving belimumab both had approximately 50% lower risks of kidney-related events (or death) and flares versus placebo [9]. The key efficacy findings of BLISS-LN and its analyses are summarised in Table 1. The BeRLiSS-LN real-world observational study found that hypertension, high baseline serum creatinine and high proteinuria were negative predictors of achieving PERR after 6 months of belimumab treatment, with high baseline proteinuria and achieving PERR at 6 months predictors of achieving PERR after 12 months of treatment [10]. In line with this, belimumab may be most effective at the early stages of LN [11–13]. In a small study of Japanese patients from the LOOPS registry, those who added belimumab to their LN induction therapy achieved significantly lower glucocorticoid doses, fewer adverse events and better disease control than standard therapy alone [12]. Importantly, earlier initiation of belimumab, especially within 6 weeks of the start of induction therapy, improved the rate of CRR achievement at Week 52 [12]. Other real-world studies have also reported data in favour of earlier treatment with belimumab to facilitate the improvement of patient outcomes [11, 13] and help reduce glucocorticoid use [14]. Steroid-sparing effects, kidney function preservation and disease-modifying abilities of belimumab in LN over a 60-month period will be evaluated in the ongoing OBSErve-LN real-world observational study (GSK Study 216147, NCT06527872). Further studies in systemic lupus erythematosus also found that treatment with belimumab may protect against kidney flares in LN-naïve patients, although this benefit may be dose- and formulation-dependent [15, 16]. Finally, a historical control study reported that in 101 paediatric patients (age 5–17 years) with newly diagnosed LN, belimumab treatment (N=38) improved efficacy endpoints at 12 months versus standard therapy (N=63) [17]. Significantly more patients treated with belimumab attained PERR (97.1% vs 80.0%; p=0.046), CRR (94.1% vs 76.6%; p=0.031), reduced glucocorticoid dose to ≤5 mg/day (42.9% vs 19.6%; p=0.017), and achieved Lupus Low Disease Activity State (LLDAS; 75.0% vs 18.6%; p<0.001) and Definition Of Remission In SLE (DORIS; 34.4% vs 11.9%; p=0.01), thus lending further evidence of better disease control with early belimumab treatment [17].
Conclusion: Clinical and observational studies support the use of belimumab as an appropriate initial therapy for active LN, aligning with the EULAR 2023, KDIGO 2024 and ACR 2024 recommendations. Early effective treatment in LN is critical to help achieve treatment goals and reduce the risk of kidney flares, each of which represents a degree of irreversible nephron loss, to prevent progression to ESKD.
REFERENCES: [1] Tektonidou MG et al. Arthritis Rheumatol 2016;68:1432–41.
[2] Korbet SM et al. Nephrol Dial Transplant 2013;28:2313–18.
[3] Fanouriakis A et al. Ann Rheum Dis 2024;83:15–29.
[4] KDIGO Lupus Nephritis Work Group. Kidney Int 2024;105:S1–69.
[5] ACR. 2024 ACR Guideline for the Screening, Treatment, and Management of Lupus Nephritis [Accessed Nov 2024].
[6] Furie R et al. N Engl J Med 2020;383:1117–28.
[7] Rovin BH et al. Kidney Int 2022;101:403–13.
[8] Malvar A et al. Lupus 2023;32:1394–1401.
[9] Anders HJ et al. Nephrol Dial Transplant 2023;38:2733–42.
[10] Gatto M et al. J Autoimmun 2021;124:102729.
[11] Lin Z et al. PeerJ 2024;12:e18028.
[12] Sakai H et al. Rheumatology (Oxford ) 2024;Sept:keae495.
[13] Sishi L et al. Rheumatology (Oxford ) 2023;Dec:kead707.
[14] Tan M et al. Kidney Dis (Basel ) 2023;9:218–28.
[15] Parodis I et al. Kidney Int Rep 2023;8:1822–30.
[16] Gomez A et al. Rheumatology (Oxford ) 2024;63:338–48.
[17] Gong Y et al. Lupus Sci Med 2024;11:e001350.
[18] Furie R et al. Clin J Am Soc Nephrol 2022;17:1620–30.
Table 1.
Acknowledgements: Medical writing support was provided by Liam Campbell, PhD, and Marta Budzinska, PhD, of Fishawack Indicia Ltd, UK, part of Avalere Health, and was funded by GSK.
Disclosure of Interests: Michelle Petri Paid instructor: unbranded lectures for Aurinia, MedShr and Arthros-FocusMedEd, Paid speaker: Alexion, Vifor Pharma, Roche, GSK, Paid consultant: BPR Scientific Advisory Committee, Alexion, Amgen, AnaptysBio, Argenx, AstraZeneca, Aurinia, AXDEV, Biogen, Boston Pharmaceuticals, Caribou Biosciences, CVS Health, Eli Lilly, Gilead Biosciences, GSK, Idorsia Pharmaceuticals, Janssen, Kezar Life Sciences, Kira Pharmaceuticals, Momenta Pharmaceuticals, Nimbus Lakshmi, Proviant, Sanofi, SinoMab and UCB, and consulting fees for participation in a data safety monitoring board or advisory board for EMD Serono, Emergent BioSolutions, IQVIA and PPD Development, Financial grants: AstraZeneca, Aurinia, Eli Lilly, Exagen, GSK, Janssen, Thermo Fisher, Ana Malvar Paid speaker: GSK, Pfizer, Novartis, Roche, Paid consultant: GSK, Roche, Biogen, BMS, Novartis, Pfizer, MUNTHER KHAMASHTA Shares: GSK, Employee: GSK, David D’Cruz Paid speaker: GSK, CSL Vifor, Paid consultant: GSK, CSL Vifor, Financial grants: CSL Vifor, Roger A. Levy Shares: GSK, Employee: GSK, Hans-Joachim Anders Paid speaker: GSK, Paid consultant: GSK.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (