

Background: T-cell LGL leukemia (T-LGLL) is a rare lymphoproliferative disorder, which is associated with rheumatoid arthritis (RA) in approximately one-third of the cases and should be suspected in patients with RA presenting with neutropenia, and/or anemia. Neutropenia is a known adverse effect of Tumor necrosis factor inhibitors (TNFi), the most used biologic drugs in RA. Recent studies have shown that long-term exposure to TNFi in patients with RA could be associated with a greater risk of developing clonal expansion of large granular lymphocytes (LGLs) [1].
Objectives: This study aimed to determine whether RA patients treated with TNFi and presenting with neutropenia are at increased risk of T-cell clonal expansion, a potential precursor to T-LGLL.
Methods: This is a retrospective cohort study including all adult patients with RA who were receiving TNFi in a Rheumatology Department of a tertiary care center during the year 2024. Demographic, clinical, laboratory and radiologic characteristics were analyzed, including gender, age, duration and activity of RA, current drug therapy, complete blood count, rheumatoid factor (RF) and anti-citrullinated protein antibodies (ACPA) levels, and radiographic evidence of erosion. Patients with at least two neutrophil count determinations below 1.5 x 10^9/L were further prospectively evaluated and underwent an additional study with flow cytometry to screen for expanded T-cell clones.
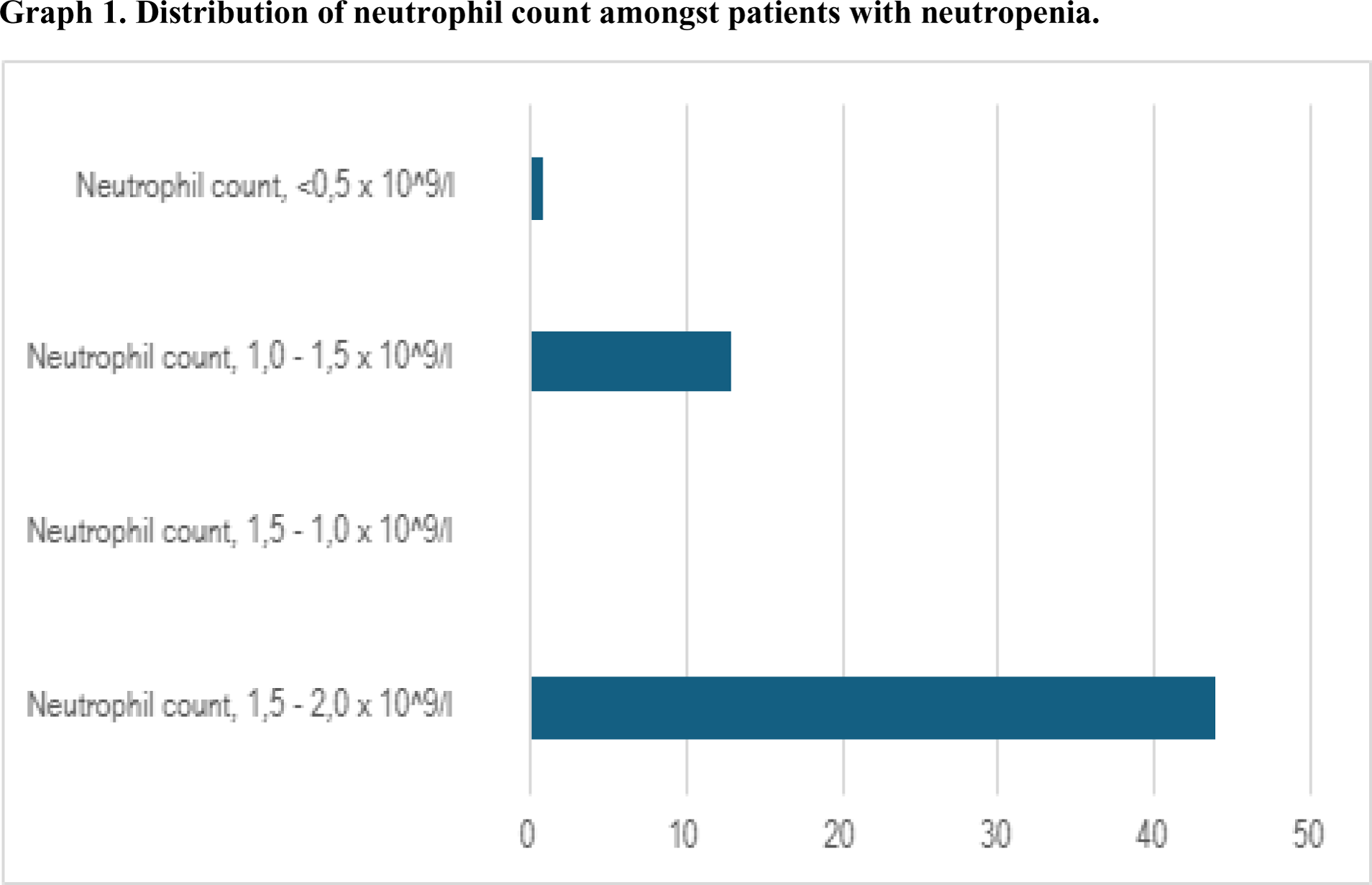
Results: A total of 316 patients with RA receiving TNFi were included. Fifty-eight patients (18.4%) had at least two neutrophil count determinations below 2.0 x 10^9/L: 44 (75.9%) were grade I neutropenias (1.5 – 2.0 x 10^9/L), 0 grade II, 13 (22.4%) grade III (1.0 – 1.5 x 10^9/L), and one patient had severe grade IV neutropenia (<0.5 x 10^9/L). Distribution of neutrophil count amongst patients with neutropenia is shown in Figure 1. Patients who presented grade III or IV neutropenia (11/316, 3.5%) were further evaluated. Their demographic data and disease characteristics are summarized in Table 1. Nine of them (81.8%) were women with a median age of 64 years (range 28-88) and a median duration of RA of 13 years (range 2.2-33). Seven patients (63.3%) were either RF or ACPA positive, and 8 of them (72.7%) had an erosive disease. The median Disease Activity Score in 28 joints (DAS28) was 2.27 (range 1.74-4.14), indicating low disease activity at the time of study inclusion. Six patients (54.5%) were treated with conventional disease-modifying antirheumatic drugs (cDMARDs) and 5 of them (45.5%) were receiving TNFi in monotherapy. All patients presented neutropenia with a median neutrophil count of 1.1 x 10^9/L (range 0.6-1.3), a median white blood cell (WBC) count of 4.7 x 10^9/L (range 3.2-5.6), median hemoglobin (Hb) levels of 12.6 g/L (range 9.9-14.1), and a median platelet count (PLT) of 252 x 10^9/L (range 68-377). Two patients (18.2%) exhibited clonal T-cell expansion with the following immunologic phenotype: both were CD57+ and had monoclonal expression of T-cell receptor constant β chain (TCRB), one of them was CD5+ and CD7+ (patient A), whereas the other one CD3 low , CD5 low , and CD7 low (patient B). Patient A was an 82-year-old woman with seropositive RA (high ACPA titers), who had a disease duration of 3.8 years and was treated with adalimumab. Patient B was a 56-year-old man with a long-standing seronegative RA of 13.2 years of evolution treated with infliximab. Both patients presented an erosive joint disease, and both manifested disease activity at the moment of the inclusion (respectively DAS28 of 2.8 and 4.1). Both were concomitantly receiving cDMARDs (namely methotrexate and/or leflunomide), along with full dosage of TNFi. No other relevant blood count changes were observed in these two patients.
Conclusion: Almost one-fifth of RA patients treated with TNFi in our cohort presented neutropenia. Moreover, two patients with grade III-IV neutropenia manifested T-cell clonal expansion. Future prospective studies are needed to better understand its clinical significance and long-term persistence.
REFERENCES: [1] Schwaneck EC, Renner R, Junker L, Einsele H, Gadeholt O, Geissinger E, Kleinert S, Gernert M, Tony HP, Schmalzing M. Prevalence and Characteristics of Persistent Clonal T Cell Large Granular Lymphocyte Expansions in Rheumatoid Arthritis: A Comprehensive Analysis of 529 Patients. Arthritis Rheumatol. 2018 Dec;70(12):1914-1922. doi: 10.1002/art.40654. Epub 2018 Oct 30. PMID: 29938921.
Distribution of neutrophil count amongst patients with neutropenia.
Demographic data and disease characteristics of patients with grade III or IV neutropenia.
| Total (n = 11) | |
|---|---|
| Female, % | 9 (81.8) |
| Age, years | 64 (28-88) |
| Duration of RA, years | 13 (2.2-33) |
| DAS28 | 2,27 (1.74-4.14) |
| RF, units/ml | 34 (4-234) |
| RF positive (>25 units/mL), % | 6 (54.5%) |
| ACPA, units/mL | 75.9 (4.6-1666.2) |
| ACPA positive (>20 units/mL), % | 7 (63.6%) |
| Erosive joint disease, % | 8 (72.7%) |
| Patients treated with cDMARDs, % | 6 (54.5%) |
| Duration of anti-TNF treatment, years | 3.9 (1-13) |
| WBC count, 10^9/L | 4.7 (3.2-5.6) |
| Neutrophil count, 10^9/L | 1.1 (0.6-1.3) |
| Hb, g/L | 12.6 (9.9-14.1) |
| PLT, 10^9/L | 252 (68-377) |
RA: rheumatoid arthritis; DAS28: Disease Activity Score in 28 joints; RF: rheumatoid factor; ACPA: anti-citrullinated protein antibodies; cDMARDs: conventional disease-modifying antirheumatic drugs; anti-TNF: anti-tumor necrosis factor; WBC: white blood cell; Hb: hemoglobin; PLT: platelet count.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (