

Background: Sodium-glucose cotransporter type 2 inhibitors (SGLT2I) have substantial benefits in diabetes mellitus (DM), heart failure (HF), and kidney disease (KD). In pivotal trials, SGLT2Is reduced serum urate (SU) levels and observational data reported prevention of gout development and flares [PMID 38472344]. However, the evidence in patients with gout under other urate-lowering drugs (ULD) is scarce.
Objectives: To evaluate the urate outcomes in patients with gout treated with SGLT2I plus ULD in clinical practice.
Methods: Retrospective, single-center observational study, enrolling patients with gout from a crystal arthritis specialized clinic. We selected those receiving combined treatment with ULD and SGLT2I, regardless of the indication. Cases with no serum urate (SU) levels available in the 6 months before and after combined treatment or starting renal replacement therapy were excluded. We focused on SU levels before and after initiating SGLT2I - analyzed by Wilcoxon test - and the achievement of SU targets (<6 and <5 mg/dL). We also collected the used allopurinol dose during SGLT2I therapy and the pre-estimated dose according to the Easy-Allo tool [PMID 38359899], as well as other relevant clinical and laboratory variables.
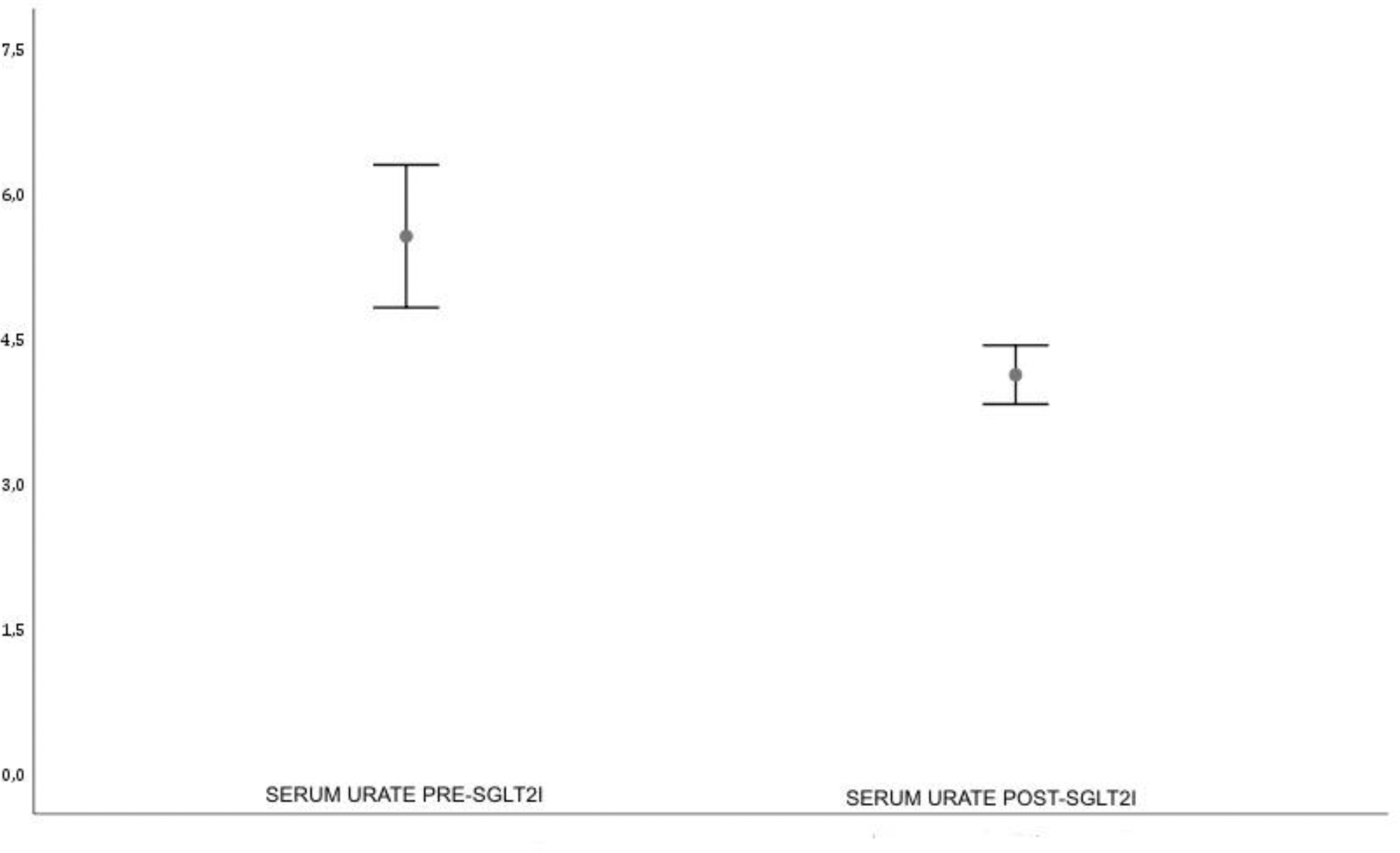
Results: Forty-six patients were included: median age 76 years (IQR 15.25), 82.6% men, with high comorbidity (91.1% with DM, 58.7% with KD, 45.7% with HF) and 10 years (IQR 20) of gout duration. As ULD, 66.7% (n=30) of patients were treated with allopurinol, 28.9% (n=13) with febuxostat, and 4.4% (n=2) with benzbromarone. Regarding the SGLT2I, 58.7% (n=27) received dapagliflozin, 30.4% (n=14) empagliflozin, and 10.9% (n=5) canagliflozin. ULD were initially prescribed in 76.1% (n=35), SGLT2I in 17.4% (n=8), both simultaneously in 4.3% (n=2), and one patient was only treated with SGLT2I (2.2%) The target of SU <6 mg/dL was achieved in 97.7% (95%CI 88.2-99.6%) of the patients, while 81.4% also reached <5 mg/dL (95%CI 65.5-88.9%). The achievement of the <5 mg/dL target was unrelated to the first prescribed medication (ULD 81.3% vs SGLT2I 87.5%, p=1.000) or the type of xanthine-oxidase inhibitor (allopurinol 79.3% vs febuxostat 91.7%, p=0.651). The start of SGLT2I showed a median SU reduction of 0.85 mg/dL (IQR 3.02, p=0.001 for the before-after comparison) [Figure 1]. The SU reduction was observed regardless of being already on ULD (median 0.80, IQR 2.90, p=0.021) or not (median 3.00, IQR 6.95, p=0.018) or of changes in diuretics use (p=0.692). No significant differences in allopurinol dose were found pre- and post-SGLT2I use but with a trend toward a lower dosage than pre-estimated by the Easy-Allo tool. The use of SGLT2I showed significant reductions in fasting glucose, HbA1c, and C-reactive protein levels, increased urine glucose excretion, and no differences in urine albumin and glomerular filtration rate. We detected a significant reduction in diuretics use pre- and post-SGLT2I (60.9% vs 54.3%, p<0.001).
SU reduction in mg/dL after the start of SGLT2I (in brackets, 95%CIs).
Conclusion: The combination of SGLT2I and ULD in patients with gout in clinical practice achieved significant SU level reductions and targets of SU <6 and <5 mg/dL. A trend towards a lower dosage requirement of allopurinol was noted, in addition to a reduced use of diuretics. These promising results require confirmation by further intervention studies.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Jose Domenech: None declared, Ivana Garcia-Loiseau: None declared, Cristina Rodríguez: None declared, Pablo Riesgo: None declared, Oscar Moreno-Pérez: None declared, Mariano Andrés Menarini, SOBI, Grunenthal.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (