

Background: Immunosuppressive therapies (ISs) are pivotal in achieving remission in systemic lupus erythematosus (SLE) but contribute to long-term damage and morbidity. Discontinuation of ISs is often followed by relapse, with a lack of robust predictors to guide withdrawal decisions. Recent evidence suggests that biomarkers like BLyS may predict flares, but further validation is needed to optimize therapy management in remitted SLE patients.
Objectives: Evaluation of the optimal combination of flare predictors in remitted SLE patients who discontinued immunosuppressive therapies.
Methods: A retrospective study was conducted on fifty-seven SLE patients diagnosed after 1990 and followed until 2023 who discontinued IS therapies after achieving remission. Remission was defined as clinical SLE Disease Activity Index (c-SLEDAI)-2K=0 on a stable background therapy and/or prednisone ≤5 mg/day. Flares were determined according to SLEDAI-Flare-Index. A multivariable logistic regression model was used to evaluate the association between occurrence of flare and time variables concerning remission, therapies implemented for induction and maintenance of remission, and serological biomarkers (anti-dsDNA, C3, C4, anti-C1q, anti-PTX3, APRIL, BLyS, C3a, and C3c) measured within 6 months after IS discontinuation (Table 1). Then we tried to create a scoring system that would predict the risk of relapse: the weight of each variable in the scoring model was assigned based on the regression coefficient, standardised to make it more interpretable. Thereafter, we chose the set of variables and the score cut-off that would maximise the area under the curve (AUC), and at the same time the sensitivity of the scoring system to predict the greatest number of flares.
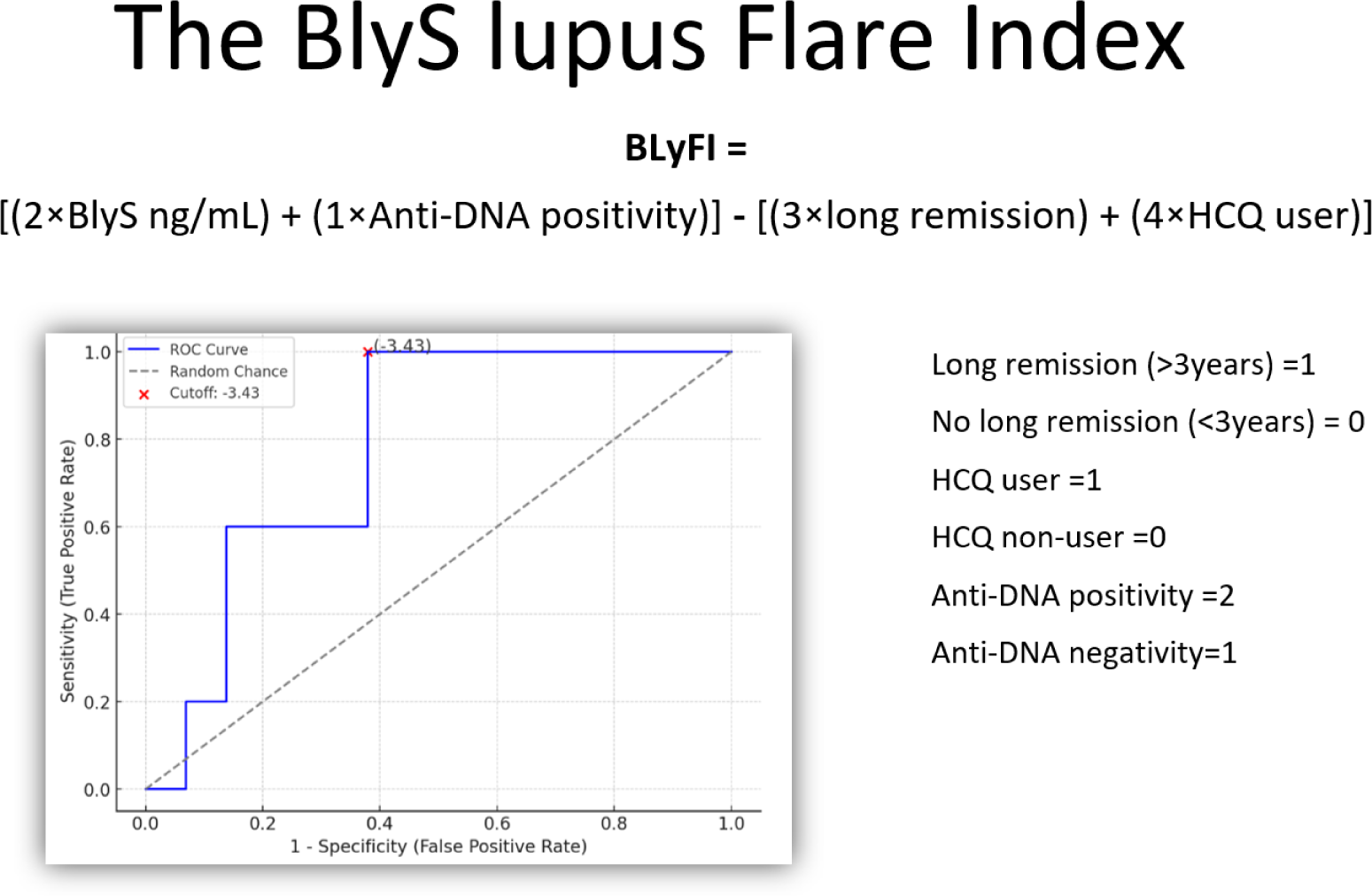
Results: Fifteen patients (26.3%) experienced a flare, on average 4.35 (± 4.1) years after IS discontinuation. BLyS serum levels were higher in patients with flares (median=0.869 ng/L, IQR=0.669-1.262, p=0.033). Hydroxychloroquine use and anti-PTX3 positivity resulted as protective factors (B=2.667, S.E.=±1.013, p=0.008) (B=1.896, S.E.=±0.967, p=0.050). BlyS value, anti-DNA positivity, duration of remission greater than three years, and hydroxychloroquine maintenance therapy provided the best ponderous combination to create a predictive score for relapses named the BlyS lupus Flare Index (BlyFI) (Figure 1). The best threshold value of the BlyFI for predicting a higher risk of relapse following discontinuation of IS was -3.43 (AUC=0.779, SE=100%, SP=62.9%).
Conclusion: High levels of BlyS, anti-PTX3 negativity, and discontinuation of hydroxychloroquine are risk factors for relapse in remitted patients after discontinuing immunosuppressants. By combining BlyS levels with other variables easily available in clinical practice, we obtained the BlyS lupus Flare Index, a score to predict the flare risk in remitted patients, candidates for immunosuppressant withdrawal.
REFERENCES: NIL.
Clinical and demographic features presented in the cohort examined. Legend: MMF=mycophenolate mofetil, AZA=azathioprine, MTX=methotrexate, CsA=cyclosporin A, HCQ=hydroxychloroquine, GC=glucocorticoid. *Some data are missing.
| Items | N | % |
|---|---|---|
| Duration of remission*>3 years | 20 | 35.1% |
| ISs discontinued due to prolonged remission* | ||
| MMF | 29 | 50.9% |
| AZA | 17 | 29.8% |
| MTX | 5 | 8.8% |
| CsA | 3 | 5.3% |
| Therapy after IS discontinuation* | ||
| HCQ | 22 | 38.6% |
| HCQ plus GC | 23 | 40.4% |
| Items | Mean (± sd ) | Median (IQR ) |
| Time to achieve remission (months) | 37.7 (± 48.5) | 13 (6 – 63) |
| Length of remission at IS stop (months) | 41 (± 30) | 36 (22 – 60) |
| Length of disease at IS stop (years) | 11 (± 8) | 9 (5-13) |
| Anti-DNA (UI/mL) | 40.75 (± 20) | 33.00 (27.50 – 50.00) |
| BLyS (ng/mL) | 1.62 (±1.98) | 0.869 (0.669 – 1.262) |
Formula and ROC curve of the BlyS Lupus Flare Index. The red dot marks the selected cutoff of -3.43. Legend: HCQ=hydroxychloroquine.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (