

Background: Systemic lupus erythematosus (SLE) is a complex autoimmune disorder with multisystem involvement, often presenting with diverse clinical manifestations and high disease activity. The ACR/EULAR 2023 guidelines recommend Belimumab or Anifrolumab for moderate to severe activity, reserving Rituximab for refractory cases. However, clinical responses vary, necessitating close monitoring to optimize treatment outcomes.
Objectives: To evaluate the clinical and analytical outcomes of patients with SLE treated with Belimumab, Anifrolumab, or Rituximab over one year and analyze patient profiles within each treatment group.
Methods: This retrospective study analyzed SLE patients treated at a university hospital from 2010 to 2024, diagnosed using the 2019 ACR/EULAR criteria. Data included demographic variables, clinical manifestations, and laboratory parameters (hemoglobin, leukocytes, lymphocytes, platelets, C3, C4, and dsDNA). Treatment duration, discontinuation rates, and concomitant therapies were recorded.
Results: Forty-five patients were included (93.33% female; median age 34.33 years, IQR: 16–60). Among them, 25 received Belimumab, 14 Anifrolumab, and 6 Rituximab. Hydroxychloroquine was used by 95.56% (mean dose 355.81 mg/day), and 88% required concomitant corticosteroids (median dose 7.5 mg/day). Clinical manifestations included articular involvement (95.56%), cutaneous lesions (71.11%), hematologic abnormalities (84.44%), neurological symptoms (46.67%), and renal disease (33.33%).
Anifrolumab demonstrated the most consistent control of hematologic parameters, with hemoglobin levels above 12 g/dL, lymphocytes > 1,500/µL, and platelets > 150,000/µL. Complement levels (C3 and C4) remained stable, though dsDNA levels increased significantly, suggesting persistent immunological activity despite clinical improvements.
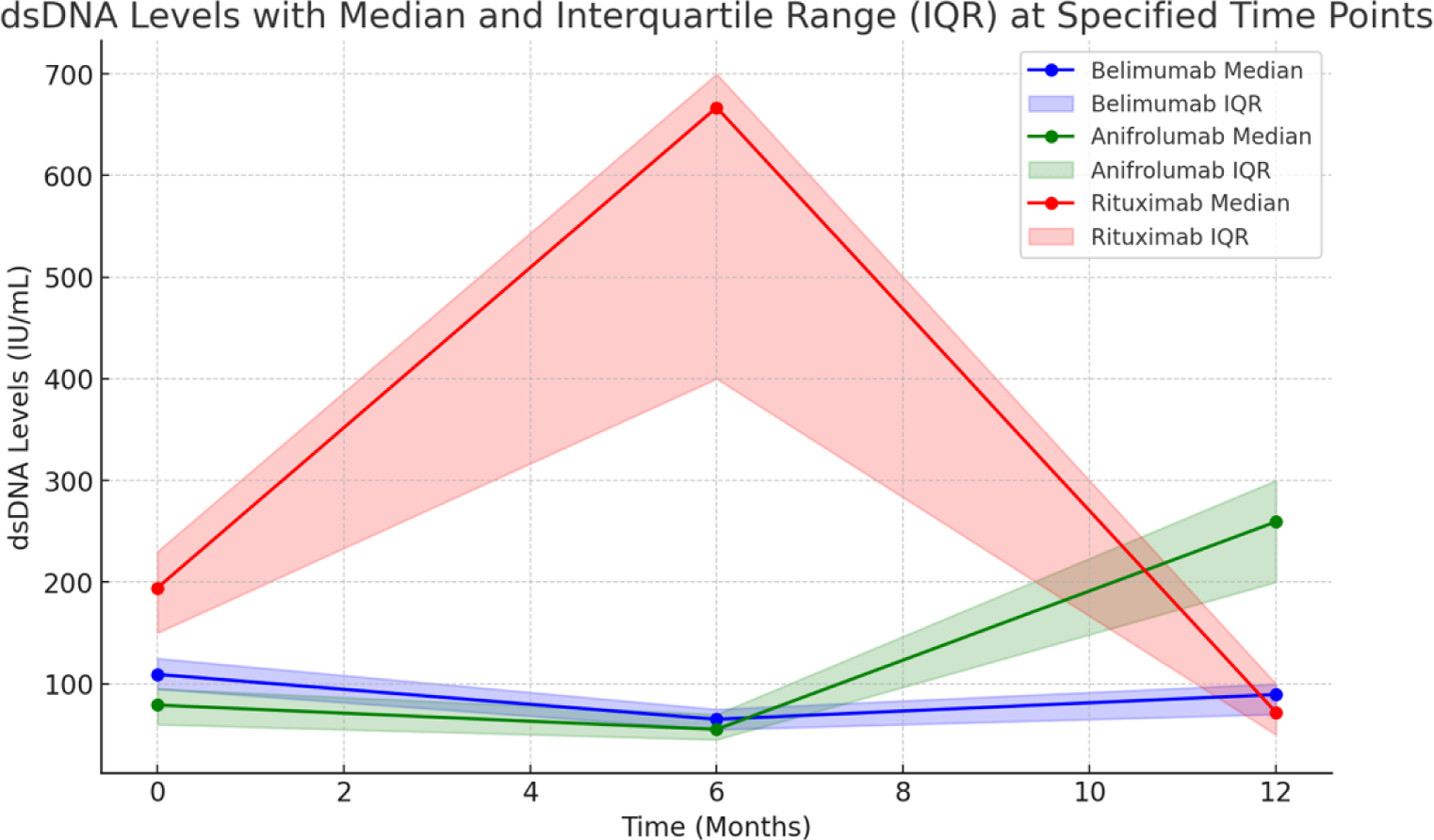
Belimumab showed moderate efficacy, maintaining stable hematologic parameters, but fluctuating complement levels and dsDNA suggested intermittent disease activity.
Rituximab provided less consistent control, with frequent leukocyte and lymphocyte drops, complement consumption, and persistently elevated dsDNA levels, reflecting ongoing disease activity in patients treated for more severe or refractory cases.
Treatment discontinuation rates were highest with Rituximab (50%), followed by Belimumab (40%) and Anifrolumab (35.71%). Causes of discontinuation included infections, lack of efficacy, and adverse events. Anifrolumab-treated patients required the lowest median corticosteroid dose (5 mg/day), compared to Belimumab (7.5 mg/day) and Rituximab (10 mg/day).
Conclusion: Anifrolumab demonstrated superior hematologic and complement stability, though its inability to reduce dsDNA levels warrants further investigation into its long-term efficacy in controlling immunological activity. Belimumab and Rituximab, while less consistent in hematologic control, provided greater dsDNA stability, particularly in patients with refractory disease. These findings highlight the need for individualized treatment strategies and regular monitoring of both clinical and laboratory parameters to optimize outcomes. Future studies should explore biomarkers that predict therapeutic response and evaluate long-term outcomes associated with these biologic therapies.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (