

Background: Lupus Nephritis (LN) affects approximately 40% of patients with systemic lupus erythematosus (SLE) and may result in end-stage kidney disease (ESKD) in up to one-third of them 10 years after diagnosis. Most randomized controlled trials (RCTs) have failed to demonstrate the superiority of biologic drugs over standard-of-care (SoC).
Objectives: The aim of this systematic review was to quantify the response to placebo (and SoC) in patients with active LN.
Methods: A systematic review was conducted according to the 2020 PRISMA statement. The PubMed database was searched (2000 to September 2024) with MeSH term ‘lupus nephritis’, for phase II/III RCTs assessing the efficacy and safety of biologics in LN. The primary end-point was complete renal response (CRR) defined as patients with proteinuria <0.5 g/day and serum creatinine <120% from baseline at 48 to 52 weeks. Secondary end-points were partial renal response (PRR, 50% improvement in proteinuria and serum creatinine <120% from baseline at 48 to 52 weeks) and overall renal response (CRR + PRR). The expected and actual differential responses were also recorded. Descriptive statistics were used.
Results: A total of 10 RCTs (n=2318 in total) were included. Placebo-treated patients (n=1006) were mostly females (86.2%), with a mean age of 32.1 years, and a mean disease duration of 29 months. LN histologic classes were class III (26.1%), IV (46.6%), V (7.2%) and mixed (20.0%). Baseline eGFR was 90.5mL/min/1.73m 2 and baseline UPCR was 3.73mg/mg. Elevated anti-dsDNA titers were detected in 70.1%, low C3 and low C4 in 62.9% and 35.7% respectively. Mean SLEDAI-2K score was 11.5. All patients were treated at baseline with glucocorticoids at a dose of 0.5-1 mg/kgBW with a maximum daily dose of 60mg. Intravenous glucocorticoids (varying doses) were administered in 91.7 %. Mycophenolate mofetil (targeted daily dose of 2-3g) was administered to 83.8% and cyclophosphamide (500 mg biweekly for 6 infusions) to 16.2% of the placebo-treated patients respectively. Antimalarials (mainly hydroxychloroquine) were administered to 64.4%; angiotensin-converting enzyme inhibitors or angiotensin receptor blockers (ACEIs/ARBs) to 65.6% respectively. CRR was achieved by 176/660 patients (26.7%), and PRR was achieved by 146/527 patients (27.7%) (Figure 1A). The expected differential response rate was 20%; the actual differential response rate was 7.5% (Figure 1B). Regarding safety, infections were observed in 46.4%, serious infections (requiring hospitalization) in 12%, malignancies in 0.2% and 29 patients died (2.88%).
A. Complete (CRR), partial (PRR) and overall (ORR) renal response at 48 to 52 weeks in patients who were treated with placebo and SoC. The fourth bar represents the percentage of placebo-treated patients who achieved the primary end-point as defined by each study. B. Expected and actual differential treatment effect.
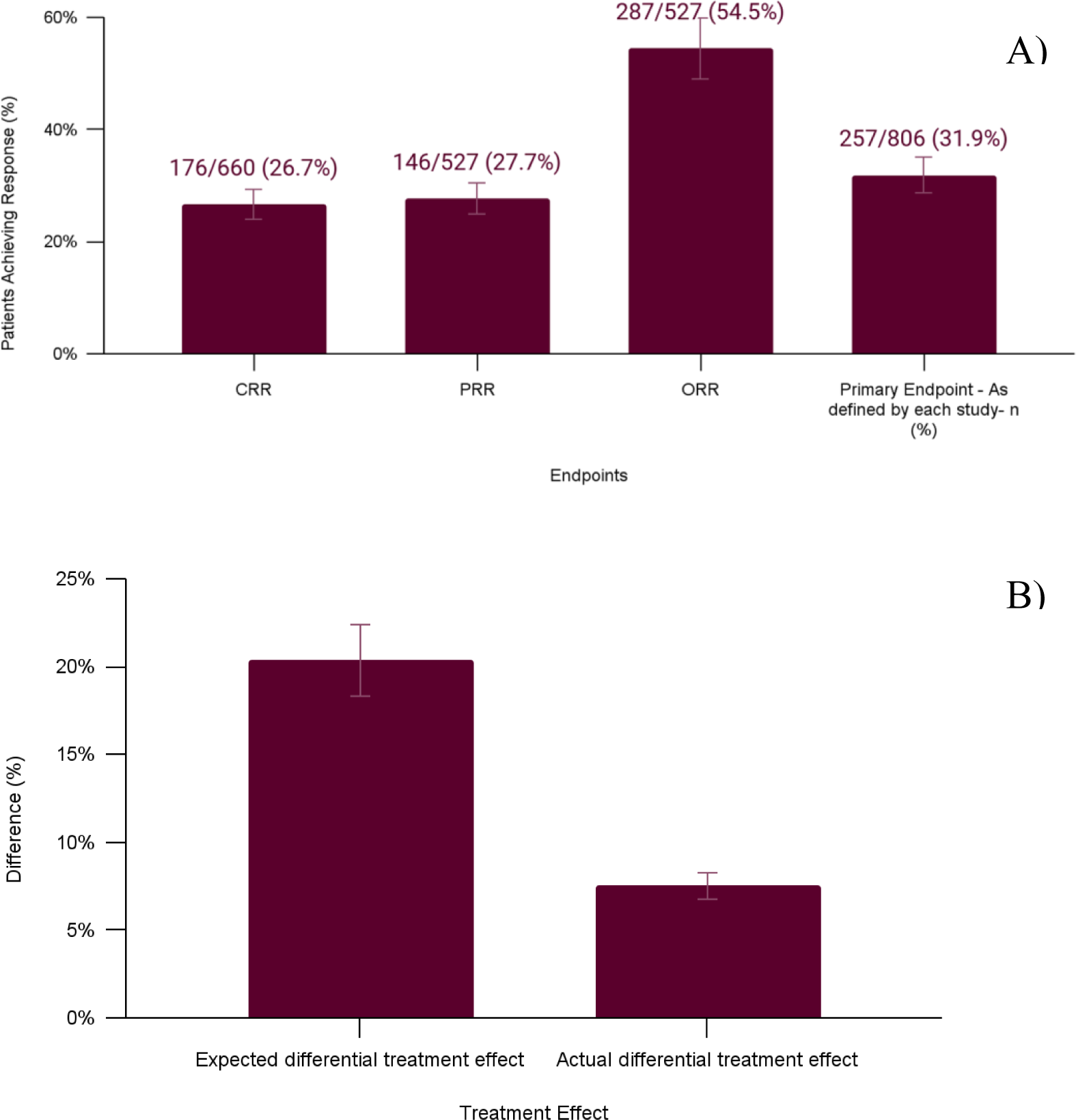
Conclusion: Approximately 27% of the placebo (+SoC)-treated patients achieved complete renal response while more than half of them achieved an overall (CRR+PRR) renal response at 48 to 52 weeks in RCTs with biologics in LN. There was a significant discrepancy between the expected and actual differential treatment effect that raises significant points to consider for the design of future studies.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Ayesha Nomaan: None declared, Mahnoor Moin: None declared, Philippe Bilodeau: None declared, Konstantinos Tselios AstraZeneca, GSK, Roche, UCB, Novartis, AstraZeneca, GSK, Fresenius Kabi.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (