

Background: Antinuclear antibodies (ANA) testing is crucial for identifying patients with systemic lupus erythematosus (SLE). However, its specificity is suboptimal and may result in unnecessary referrals to Rheumatology. Triage tools that enable early differentiation between SLE and non-SLE among ANA-positive patients could expedite initial assessment and improve patient outcomes [1], making them particularly valuable for lupus referral centers. The SLE Risk Probability Index (SLERPI) is a simple-to-use, machine-based model utilizing 8 clinical and 6 laboratory variables, designed to assist SLE diagnosis, with potential clinical utility for this purpose [2].
Objectives: The objective of this study was to evaluate the value of SLERPI in identifying patients with a higher probability of SLE among ANA positive patients.
Methods: We retrospectively reviewed consecutive referrals that were received between January 1st and October 31st, 2024 for suspected SLE. Referrals for patients younger than 16 years old or without documented positive ANA (≥1:80 by immunofluorescence) or with a prior diagnosis of connective tissue disease were excluded. A single reviewer analyzed each consultation request from the referring clinicians and scored SLERPI items individually, using only the information included in the referral. Based on this scoring, the total SLERPI score was classified as positive or negative, using a cut-off of >7 points, as previously described. Patients were classified as either non-SLE or diagnosed with SLE based on clinical documentation from their charts. Additional collected data included initial triage priority (Urgent, Semi-Urgent, Routine) assigned by the referral center, and a retrospective triage reassessment by the principal reviewer. Descriptive statistics were used.
Results: Fifty referrals meeting the selection criteria were identified. Of these, 44 were from general practitioners and 6 from specialists, with 90% showing positive ANA by immunofluorescence at titers ≥1/160. Initial triage classified 20% of cases as Semi-Urgent and 80% as Routine. On average, 8.7 out of 14 SLERPI items were undocumented by the referring provider. Only four items (arthritis, platelet numbers, leukocyte numbers and proteinuria) were documented in ≥50% of referrals, while 8 out of 14 items were documented in ≤20% of referrals. Among the 50 charts reviewed, 5 had a positive SLERPI score (>7), while 45 were negative. Four patients were diagnosed with SLE, and 46 were deemed non-SLE. Preliminary analysis demonstrated a positive predictive value of 60% for a SLERPI score >7 to identify SLE patients, with a negative predictive value of 98%, for an 8% SLE prevalence (Figure 1). SLERPI score >7 showed a positive likelihood ratio of 17.3 and a negative likelihood ratio of 0.26 for SLE identification. A SLERPI score >7 would have reclassified 6 non-SLE patients as Routine and prioritized one SLE case as Semi Urgent (Figure 2). The SLERPI score’s negative likelihood ratio was 0.13 for identifying patients ultimately classified as Routine, including those initially marked as Routine who remained so after review and those reclassified from Semi-Urgent to Routine.
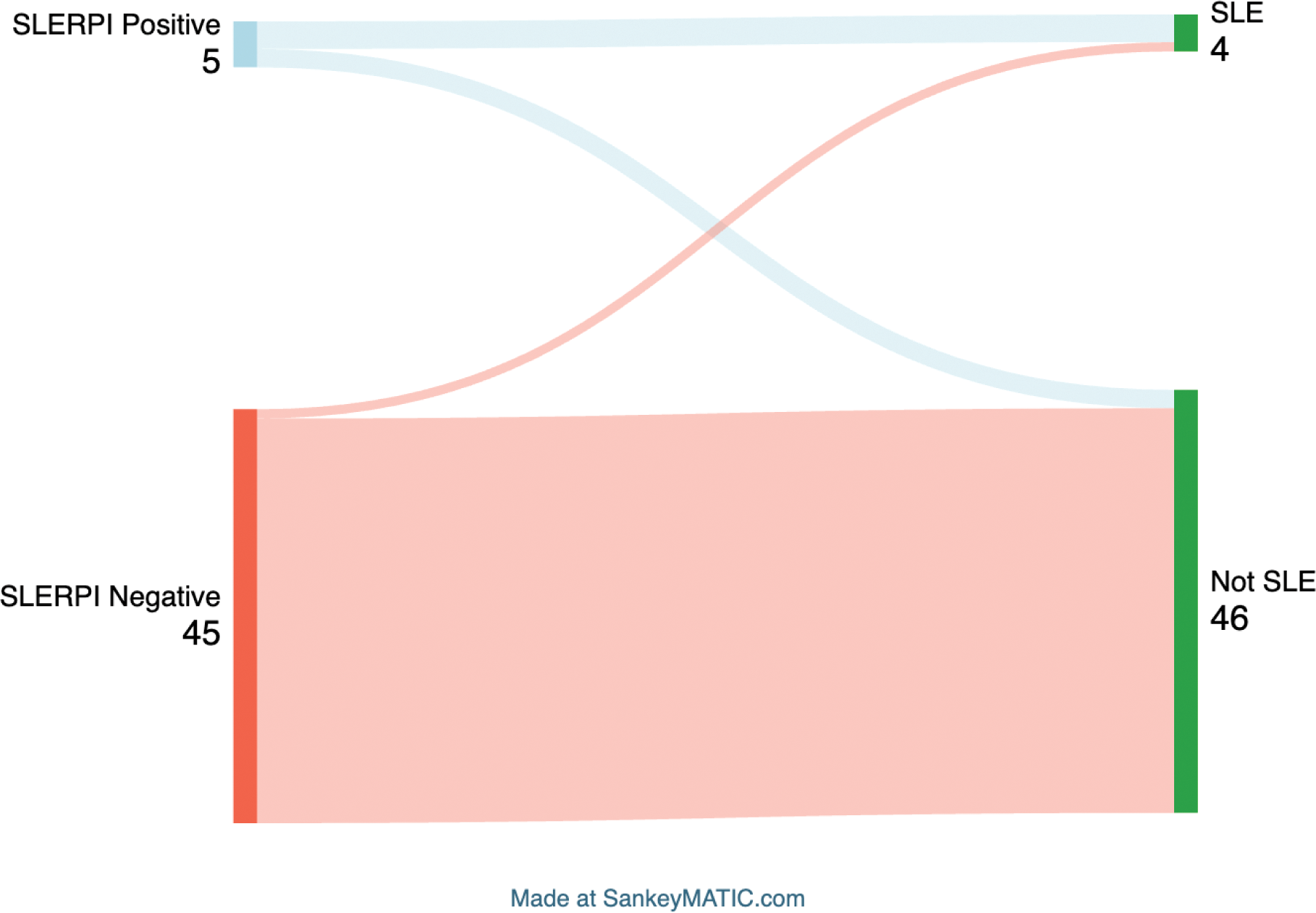
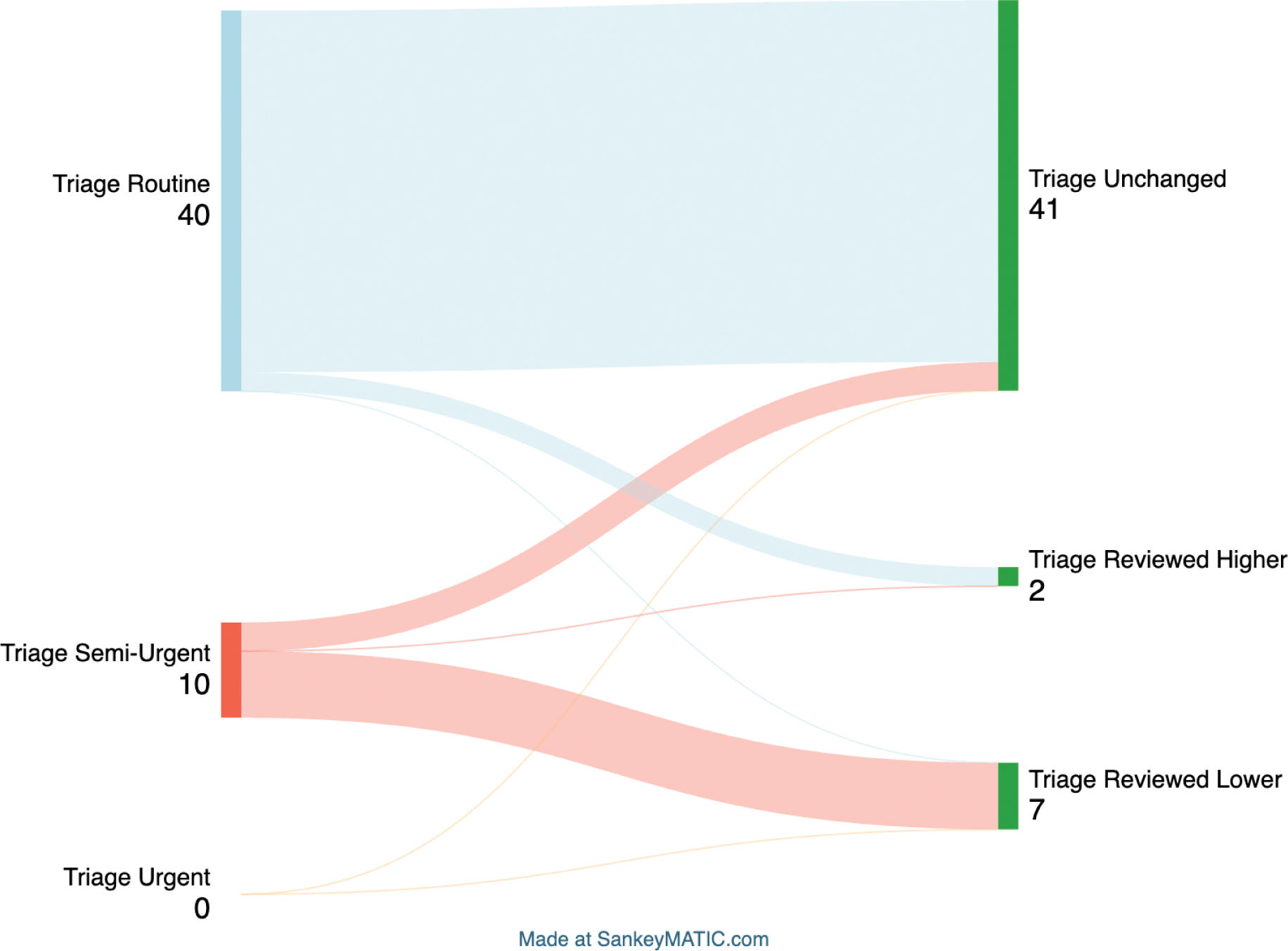
Conclusion: SLERPI shows potential as an effective triage tool for distinguishing SLE from non-SLE among ANA-positive patients. With a high negative predictive value of 98% for SLE diagnosis and a negative likelihood ratio of 0.13, the SLERPI score could help prioritize referrals. This may, in turn, accelerate early rheumatology assessment in newly diagnosed SLE patients, thus improving outcomes. This approach might also allow for more efficient resource allocation in lupus referral centers.
REFERENCES: [1] Adamichou C. Ann Rheum Dis 2021;80(6):758-766.
[2] Floris A. Arthritis Care Res 2020;72:1794-9.
Acknowledgements: NIL.
Disclosure of Interests: Philippe Bilodeau: None declared, Konstantinos Tselios AstraZeneca, GSK, Roche, UCB, Novartis, AstraZeneca, GSK, Fresenius Kabi.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (