

Background: Baricitinib (BARI) is an oral JAK 1/2 inhibitor approved for the treatment of moderate to severely active rheumatoid arthritis (RA) [1]. RA-BE-REAL is a multinational, prospective, observational study of adult patients with RA with a 36-month (36M) and 24-month (24M) follow-up in European (EU) and non-European regions (OEU), respectively. In the EU population, a higher proportion of patients treated with BARI compared with other targeted synthetic (ts) or biologic (b) disease-modifying antirheumatic drugs (DMARDs), achieved remission (REM) or low disease activity (LDA) as previously reported [1].
Objectives: To provide descriptive baseline characteristics and effectiveness outcomes of BARI and other ts/bDMARDs at 24M in both EU and OEU regions, in addition to the effectiveness of BARI in EU countries at 36M.
Methods: Patients (N=1322) with RA from five EU countries (France, Germany, Italy, Spain, and UK) and three OEU countries (Australia, Canada, and Saudi Arabia) initiated BARI 2 mg or 4 mg (Cohort A) or any ts/bDMARDs (Cohort B) as per label requirements for the first time. Response rates for REM and LDA were determined by the Clinical Disease Activity Index (CDAI), and the patient’s assessment of pain using the 100 mm pain visual analog scale (VAS).
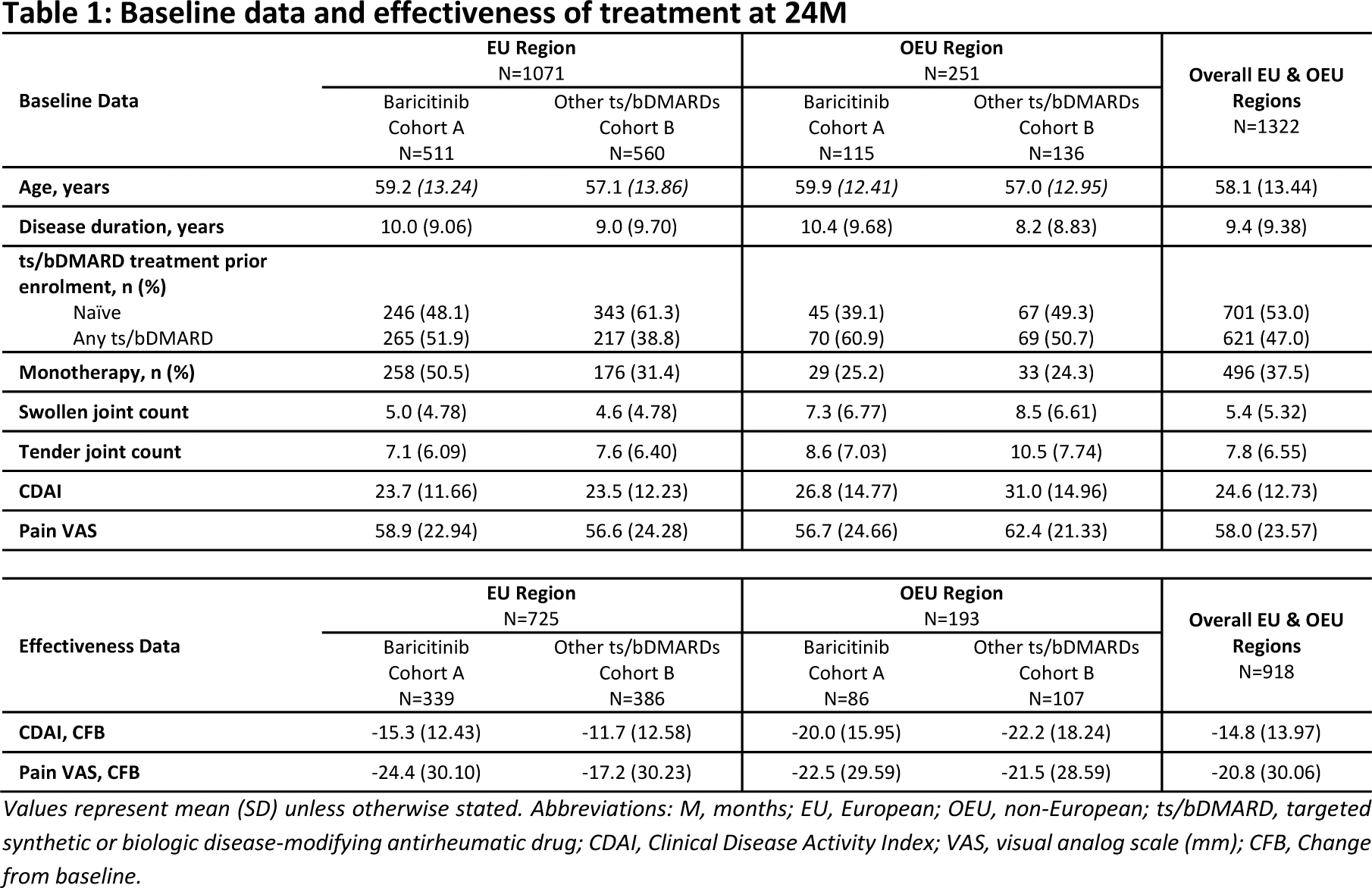
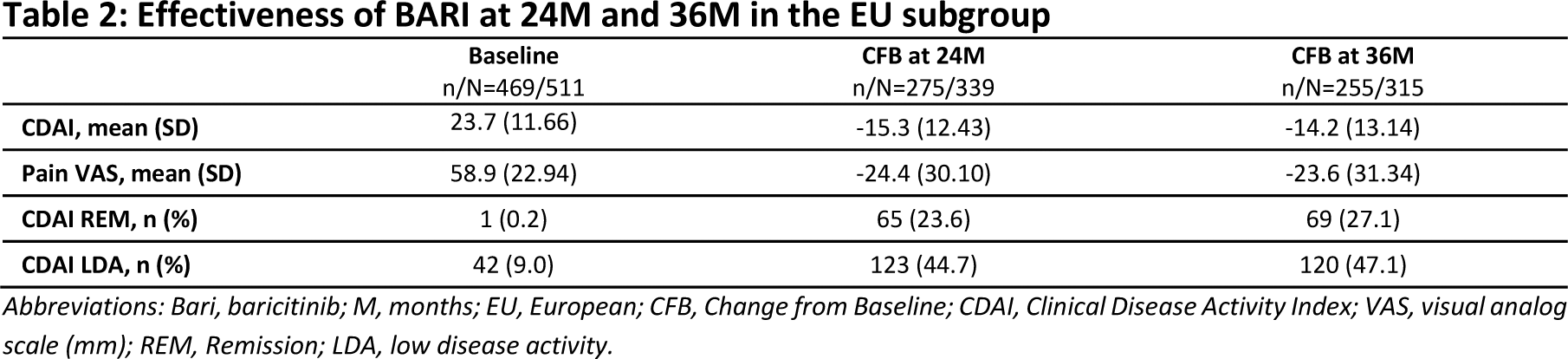
Results: A summary of patient characteristics identified higher baseline disease activity in OEU countries however, improvements in disease activity and pain were observed in both regions at 24M, Table 1. In Cohort B, 83.8% received bDMARDs [57.5% TNFi, 26.3% non-TNFi], 16.2% tsDMARDs, and 30% were on monotherapy. The overall CDAI mean change from baseline (CFB) at 24M was -14.8 (13.97) (Cohort A: -16.2 [13.31], Cohort B: -13.7 [14.41]) and the overall mean change of pain VAS from baseline was -20.8 (30.06) (Cohort A: -24.0 [29.96], Cohort B: -18.1 [29.91]). Most patients achieved CDAI REM or LDA by 24M (68.8% Cohort A [24.4% REM, 44.4% LDA], 62.1% Cohort B [23.3% REM, 38.8% LDA]), with results numerically favoring BARI. Effectiveness results at 24M within the EU and OEU subgroups mirrored the overall effectiveness data. The overall CDAI mean CFB at 24M was -13.3 (12.63) (Cohort A: -15.3 [12.43], Cohort B: -11.7 [12.58]) in the EU and -21.2 (17.19) (Cohort A: -20.0 [15.95], Cohort B: -22.2 [18.24]) in the OEU regions. Additionally, the mean CFB in pain VAS was -20.6 (30.36) (Cohort A: -24.4 [30.10], Cohort B: -17.2 [30.23]) and -21.9 (28.97) (Cohort A: -22.5 [29.59], Cohort B: -21.5 [28.59]), in EU and OEU regions, respectively. Most patients within the EU and OEU subgroups achieved CDAI REM or LDA by 24M (EU: 68.3% Cohort A [23.6% REM, 44.7% LDA], 61.4% Cohort B [23.1% REM, 38.3% LDA]; OEU: 70.8% Cohort A [27.7% REM, 43.1% LDA], 65.4% Cohort B [24.4% REM, 41.0% LDA]), with results numerically favoring BARI. Patients treated with BARI in the EU subgroup were followed up for 36M, and improvements in disease activity and pain were maintained from baseline, Table 2. CDAI mean CFB was -14.2 (13.14), and mean change of pain VAS was -23.6 (31.34). By 36M, 74.2% of patients treated with BARI achieved CDAI REM or LDA (27.1% and 47.1%, respectively).
Conclusion: Baseline clinical characteristics indicated that OEU patients exhibited higher disease activity, with similar improvements observed in EU and OEU patients treated with BARI. Effectiveness outcomes within EU and OEU subgroups reflected overall results, with results numerically favoring BARI. Effectiveness outcomes in the EU population of patients treated with BARI demonstrated a sustained response over 36M.
REFERENCES: [1] Alten et al., Rheumatol Ther. 2023; 10:1575-1595.
Acknowledgements: Medical writing services were provided by John Downey, an employee of Eli Lilly and Company.
Disclosure of Interests: Rieke Alten Speakers bureau: Abbvie, BMS, Celltrion, Chugai, Galapagos, Janssen, Eli Lilly and Company, Novartis, Pfizer, Roche, UCB, and Viatris, Consultant of: Abbvie, BMS, Celltrion, Chugai, Galapagos, Janssen, Eli Lilly and Company, Novartis, Pfizer, Roche, UCB, and Viatris, Gerd R. Burmester Speakers bureau: Alfasigma, Abbvie, Galapagos, Eli Lilly and Company, and Pfizer, Consultant of: Abbvie, Galapagos, and Alfasigma, Marco Matucci-Cerinic Speakers bureau: BI, and MSD, Grant/research support from: ARGENX, and BI, Jens Gerwien Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Walid Fakhouri Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Samuel Ogwu Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Ewa Haladyj Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Inmaculada De La Torre Shareholder of: Eli Lilly and Company, Employee of: Eli Lilly and Company, Bruno Fautrel Consultant of: AbbVie, Alfasigma, Amgen, Biogen, BMS, Celltrion, Chugai, Fresenius Kabi, Eli Lilly and Company, Medac, Nordic Pharma, Novartis, Owkin, Pfizer, ReMeds, Sandoz, SOBI, and UCB, Grant/research support from: AbbVie.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (