

Background: Monoclonal proteins of a heterogeneous type appear to occur more frequently in patients with various autoimmune disorders, including systemic lupus erythematosus (SLE), with respect to the general population. In general population, monoclonal gammopathy of undetermined significance (MGUS) prevalence is estimated to be 3% among individuals aged >50 [1] and MGUS carries a 1% per year risk of progression to multiple myeloma [2]. However, their clinical significance remains unclear, particularly in the context of SLE, necessitating further investigation.
Objectives: This study aimed to evaluate the prevalence and clinical characteristics of monoclonal gammopathy (MG) in SLE patients in a real-life setting, to assess the long-term outcome in term of progression to multiple myeloma, and to investigate SLE features more likely associated with MG.
Methods: This study is a retrospective analysis of prospectively collected data from a monocentric SLE cohort. The following variables were retrieved from clinical records: demographic data, cumulative organ involvement, treatments with conventional or biological DMARDs at any time during the disease course, concurrent diagnosis of Sjögren’s syndrome, and the presence of MG. For patients with MG, additional data were collected on follow-up duration since MG detection.
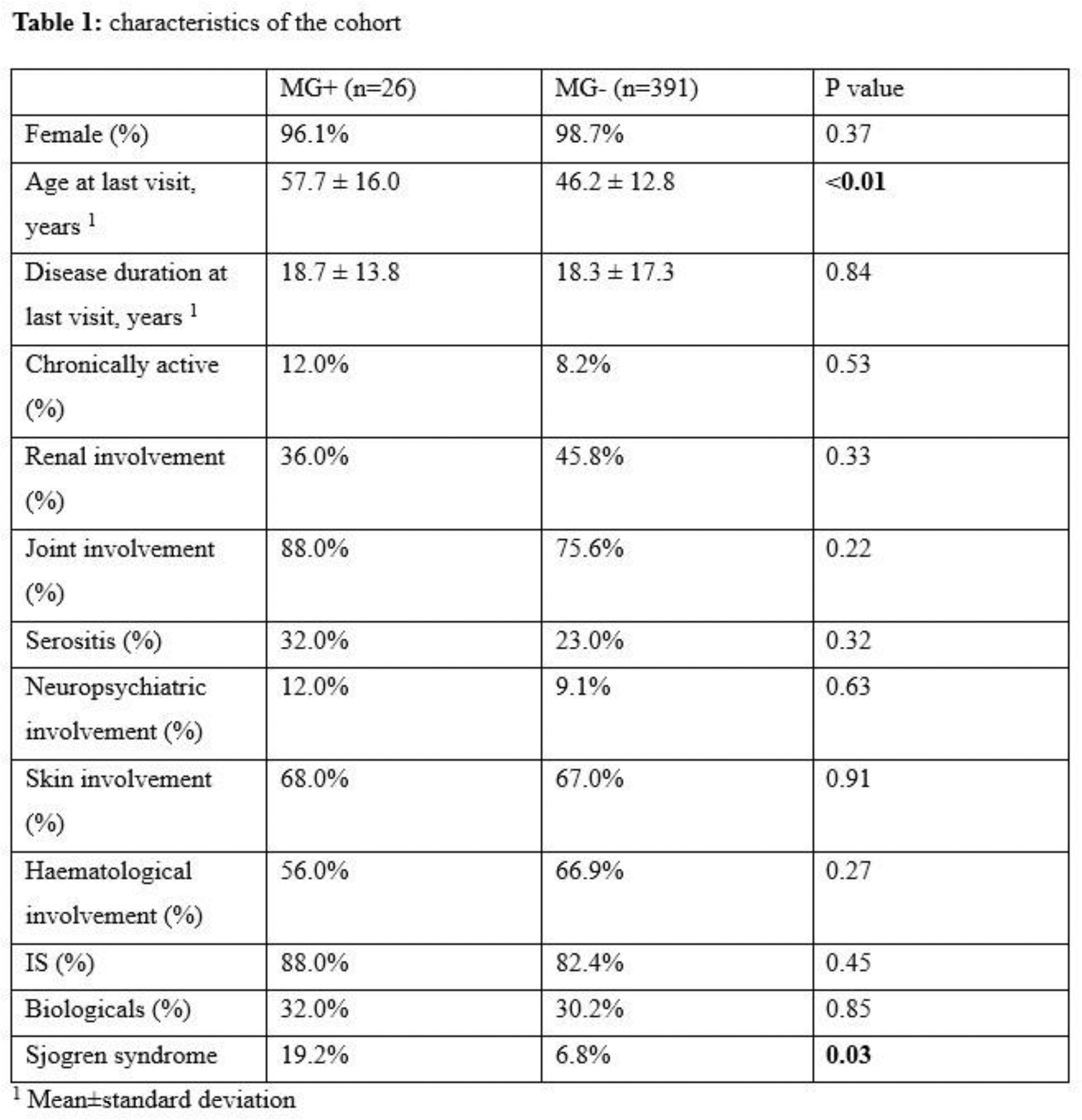
Results: From our cohort of 417 SLE patients with a mean disease duration of 18.3 ± 10 years at last evaluation, 26 (6.2%) were found to have documented monoclonal gammopathy (MG). The most common type of gammopathy was IgG (72.2%), followed by IgM (22.2%) and IgA (5.6%). The mean follow-up period since MG detection was 3.8 ± 2.6 years (range 1–10 years). During follow-up, 2 patients progressed to smouldering multiple myeloma, and the evolution occurred 3 and 4 years after MG detection, respectively. Therefore, in our cohort, MG carries an estimated 2.02% per year risk of progression to smouldering multiple myeloma. Any MGUS progressed to symptomatic myeloma. The characteristics of patients with MG, as well as comparisons with SLE patients without MG, are summarized in Table 1. Patients with MG were older at the time of the last visit (mean age 57.7 ± 16 years vs. 46.2 ± 12.8 years, p<0.01) and were more likely to have a concurrent diagnosis of Sjögren’s syndrome (19.2% vs. 6.8%, p = 0.03). No significant differences were observed between the two groups in terms of disease duration, organ involvement, or history of treatment with conventional or biological DMARDs.
Conclusion: Our findings highlight that MG is more frequently observed in SLE patients, particularly those with Sjögren’s syndrome, compared to the general population. In this cohort, the gammopathy usually exhibits a benign course, but the estimated annual progression risk to smouldering multiple myeloma underscores the need for vigilant monitoring. The small sample size limits the generalizability of these results, emphasizing the value of larger studies to validate and expand upon these findings.
REFERENCES: [1] Kyle RA et al. A Long-Term Study of Prognosis in Monoclonal Gammopathy of Undetermined Significance. N Engl J Med, 2002.
[2] Kyle RA et al. Prevalence of Monoclonal Gammopathy of Undetermined Significance. N Engl J Med, 2006.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (