

Background: Systemic Lupus Erythematosus (SLE) is a complex autoimmune disease characterised by multisystem involvement, where neuropsychiatric manifestations, including depression, are prevalent. Dysregulated activation of the type I interferon (IFN) pathway contributes to SLE pathogenesis and has been implicated in depressive behaviour through mechanisms such as serotonin modulation and neurogenesis inhibition [1]. Anifrolumab, a monoclonal antibody targeting the Type I IFN receptor, is effective in reducing SLE disease activity and glucocorticoid burden [2]. However, its potential impact on depressive symptoms remains underexplored.
Objectives: To assess the impact of intravenous anifrolumab 300 mg on depressive symptoms in adult patients with moderately to severely active SLE, alongside secondary outcomes evaluating patient-reported quality of life.
Methods: This single-centre, prospective, observational study enrolled adult SLE patients meeting 2019 EULAR/ACR classification criteria with autoantibody positivity and active disease (SLEDAI-2K ≥6). Participants received 300 mg of anifrolumab intravenously every 4 weeks in combination with background SLE-treatment as per clinician’s practice. Depression severity was assessed using Beck’s Depression Inventory (BDI) and Patient Health Questionnaire-9 (PHQ-9) at baseline, 3, 6, and 12 months. Patient-reported outcomes included FACIT score and Lupus Quality of Life (Lupus QoL). Wilcoxon signed-rank test was used to compare with baseline median values of all variables at different timepoints. Due to lacking numerosity in this preliminary phase, Cohen’s effect size was calculated to outline clinical impact of treatment. Data are expressed as mean ± SD unless otherwise specified.
Results: Twenty-six (26) patients have been enrolled to date. Female sex is predominant (n=22; 85%), with a mean age of 46 ± 15.0 and disease duration of 11 ± 9 years. The commonest disease manifestations were cutaneous and musculoskeletal, observed in 93% of the sample. Four patients (14%) had a history neuropsychiatric SLE. A moderate/strong reduction in median BDI scores was observed at 12 months compared with baseline (-0.43 effect size) (Figure 1). Similarly, PHQ-9 scores showed sustained improvement (effect size -0.28 at 12 months). FACIT and LupusQoL scores exhibited comparable positive trends (effect size -0.40 and +0.40 respectively at 12 months). Lupus QoL improvement also resulted statistically significant at 6 months compared to baseline overall (84.0 vs. 63.5, p=0.049), and in different sub-domains, namely pain (11.0 vs. 8.0, p=0.017), burden to others (9.0 vs 6.5, p=0.039), and emotional health (21.0 vs. 18.5, p=0.048) (Table 1). Moreover, anifrolumab improved systemic and cutaneous disease activity at 12 months (median SLEDAI-2k 4.0 vs. 8.5 at baseline, p=0.002; median CLASI-A 0.0 vs. 3.0 at baseline, p=0.015). All 7 patients treated for at least 12 months were glucocorticoid-free at last follow-up and 4 (57%) met DORIS remission criteria.
Conclusion: In this preliminary report, anifrolumab seems to alleviate depressive symptoms in SLE patients, with concurrent improvement in their quality-of-life. These findings highlight a potential dual role of anifrolumab in modulating SLE disease activity and depressive symptoms, mediated through the inhibition of IFN signalling. However, mood improvement apparently mirrors disease activity and may also be secondary to a general healing effect of the drug instead. Comparative studies with other effective treatments for SLE, longer follow-up, larger samples and disease biomarkers are warranted to confirm these effects and elucidate underlying mechanisms.
REFERENCES: [1] Capuron L, Miller A. Cytokines and Psychopathology: Lessons from Interferon-α. Biol Psychiatry 2004;56:819–824; doi:10.1016/j.biopsych.2004.02.009.
[2] Morand EF, et al. Trial of Anifrolumab in Active Systemic Lupus Erythematosus. N Engl J Med 2020;382:211-21; doi: 10.1056/NEJMoa1912196.
Effect size heatmap of depression and quality of life scores over time during anifrolumab treatment.
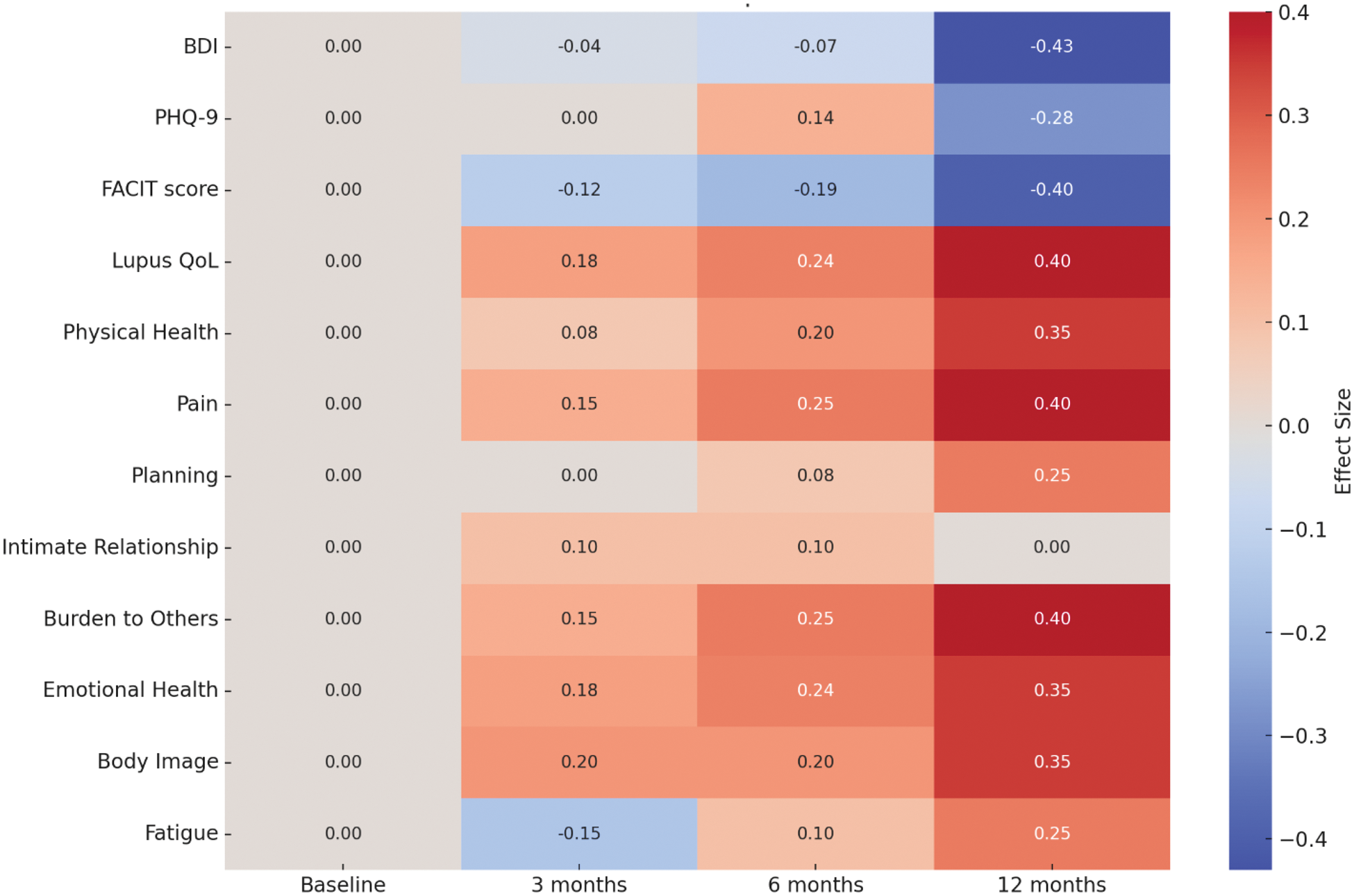
Median changes in patient-reported depression measures and quality of life during treatment period.
| Baseline (n=26) | 3 months (n=23) | 6 months (n=14) | 12 months (n=7) | ||||
|---|---|---|---|---|---|---|---|
| Median ± IQR | Median ± IQR | P value* | Median ± IQR | P value* | Median ± IQR | P value* | |
| BDI | 9.5 ± 14 | 9.0 ± 10 | 0.597 | 10.0 ± 9 | 0.599 | 3.0 ± 5 | 0.156 |
| PHQ-9 | 6.5 ± 8 | 5.0 ± 7 | 0.888 | 4.0 ± 6 | 0.878 | 2.0 ± 4 | 0.269 |
| FACIT score | 16.5 ± 14 | 13.0 ± 11 | 0.161 | 10.5 ± 9 | 0.195 | 7.0 ± 6 | 0.345 |
| Lupus QoL | 63.5 ± 25 | 80.0 ± 30 | 0.064 | 84.0 ± 20 | 0.049 | 95.0 ± 15 | 0.173 |
| Physical Health | 22.0 ± 10 | 24.0 ± 12 | 0.441 | 27.0 ± 8 | 0.059 | 29.0 ± 6 | 0.172 |
| Pain | 8.0 ± 6 | 10.0 ± 5 | 0.161 | 11.0 ± 4 | 0.017 | 12.0 ± 3 | 0.176 |
| Planning | 9.0 ± 6 | 9.0 ± 5 | 0.349 | 10.5 ± 4 | 0.439 | 12.0 ± 3 | 0.593 |
| Intimate Relationship | 6.0 ± 5 | 8.0 ± 4 | 0.26 | 8.0 ± 4 | 0.34 | 7.0 ± 3 | 0.892 |
| Burden to Others | 6.5 ± 6 | 8.0 ± 5 | 0.2 | 9.0 ± 4 | 0.039 | 12.0 ± 3 | 0.141 |
| Emotional Health | 18.5 ± 8 | 21.0 ± 7 | 0.048 | 23.0 ± 6 | 0.247 | 23.0 ± 5 | 0.673 |
| Body Image | 13.0 ± 7 | 16.0 ± 6 | 0.4 | 16.0 ± 5 | 0.079 | 20.0 ± 4 | 0.077 |
| Fatigue | 11.0 ± 6 | 9.0 ± 5 | 0.33 | 12.0 ± 4 | 0.831 | 16.0 ± 3 | 0.414 |
*P values of Wilcoxon signed-rank test compared with baseline
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (