

Background: Tacrolimus is a pregnancy-compatible immunosuppressive increasingly used in systemic lupus erythematosus (SLE) pregnancies. Physiological changes throughout pregnancy impact tacrolimus pharmacokinetics, altering the drug’s whole blood concentrations during gestation. However, data is very limited to guide clinicians caring for pregnant women receiving tacrolimus in interpreting tacrolimus trough levels and adjusting the dosage.
Objectives: By completing a systematic literature review, we evaluated the variation of maternal tacrolimus trough levels and dosage during pregnancy in SLE and non-SLE women.
Methods: Using a combination of relevant search terms and keywords, we systematically searched Embase, Ovid, PubMed, Web of Science and Cochrane Library up to September 2024. All observational studies which measured tacrolimus trough levels during pregnancy were included without language or date restriction. Records which were reviews, case reports, abstracts only, non-human, and had no tacrolimus trough levels during pregnancy were excluded from the review. We used the random-effects model where mean trough levels and doses of tacrolimus before pregnancy, during pregnancy and in the postpartum were analyzed and expressed as standardized mean difference (SMD) or mean difference (MD), with 95% confidence intervals (CI).
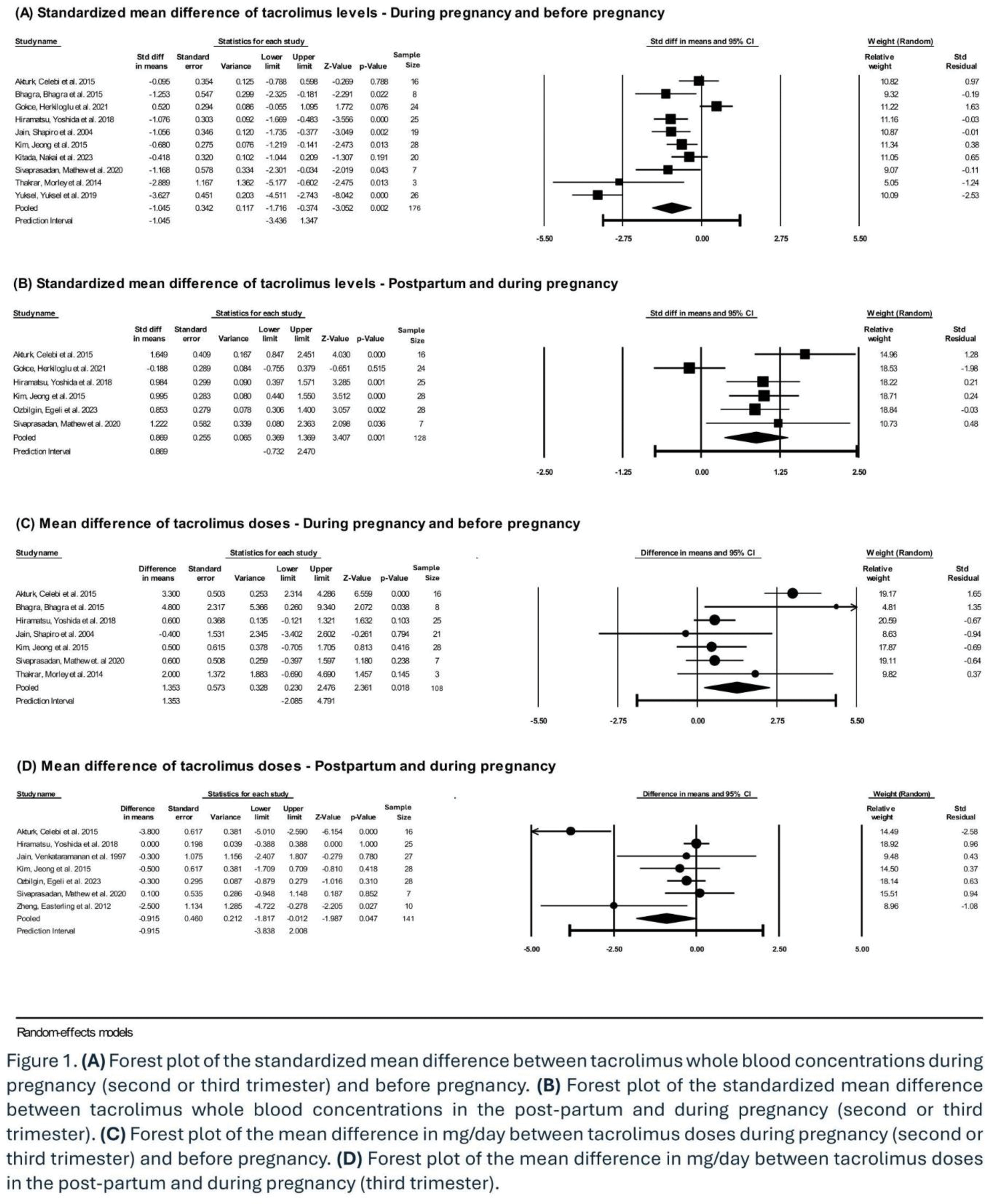
Results: Of 404 records identified, 282 were screened based on title and abstract, of which 53 full-text articles were assessed for eligibility. Eighteen articles were included in the systematic review and 13 in the meta-analysis, of which only 2 specifically assessed tacrolimus blood levels in SLE pregnancies, while the remainder addressed tacrolimus monitoring in pregnant post-transplant recipients. Whole blood concentrations of tacrolimus significantly decreased during pregnancy compared to pre-pregnancy levels (SMD -1.05; 95% CI -1.72, -0.37) and significantly increased in the postpartum compared to the levels during gestation (SMD 0.87; 95% CI 0.37, 1.37) (Figure 1A and B). The mean differences in tacrolimus trough levels were -1.56 ng/ml (95%CI -2.82, -0.31) between first trimester and before pregnancy, -0.49 ng/ml (95% CI -1.04, -0.07) between second and first trimesters, 0.63 ng/ml (95% CI 0.30, 0.96) between third and second trimesters, and 1.28 ng/ml (95% CI 0.60, 1.96) between the postpartum and third trimester. The variation in tacrolimus levels during pregnancy was usually addressed by increasing the drug dose during pregnancy vs pre-pregnancy (MD 1.35 mg/dayl 95% CI 0.23, 2.48) and decreasing the dose in the postpartum vs pregnancy (MD -0.92 mg/day; 95% CI -1.8, -0.01) (Figure 1C and D).
Conclusion: Tacrolimus whole-blood concentrations decrease in the first and second trimesters, then increase back to pre-pregnancy levels in the postpartum. Tacrolimus dosage in pregnancy is increased to maintain tacrolimus trough levels within the usual therapeutic ranges. Increasing drug dosage could elevate the bio-effective fraction of tacrolimus (not measured by trough levels), raising concerns for safety and efficacy of dose augmentation during pregnancy. Further research is needed to guide clinicians in adjusting tacrolimus dosage in SLE and post-transplant pregnancies to optimize therapeutic drug monitoring in these high-risk populations.
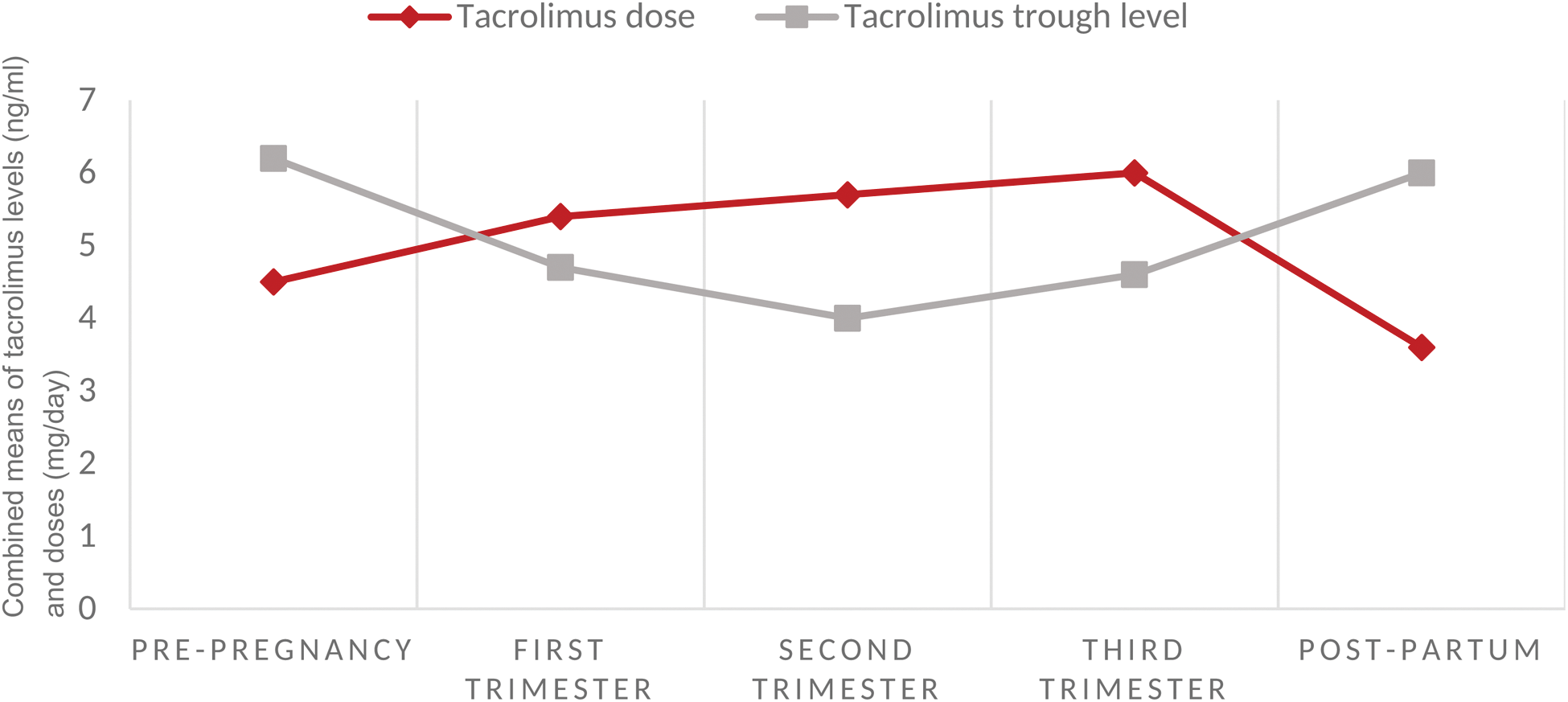
Combined means of tacrolimus doses (mg/day) and whole-blood trough levels (ng/ml) before pregnancy, at each trimester and in the postpartum (10 studies, n=182).
REFERENCES: NIL.
Acknowledgements: This work was supported by a student bursary from the Canadian Mother-Child Cohort (CAMCCO) and by a funding award from the Canadian Rheumatology Association (CRA) - Canadian Initiative for Outcomes in Rheumatology Care (CIORA).
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (