

Background: Thrombocytopenia that develops during the course of SLE is associated with a poor prognosis and its treatment can sometimes be challenging.
Objectives: We aimed to identify clinical, laboratory characteristics and prognostic features of SLE patients with thrombocytopenia.
Methods: This retrospective single center study included 102 patients SLE patients with chronic (>12 months) or acute (<3 months) thrombocytopenia. All data regarding clinical and laboratory characteristics, disease duration, treatments, disease activity (SLEDAI) and damage, and bleeding history were reviewed. Thrombocytopenia was defined as a platelet count <100 × 10 9 /L and was confirmed by a peripheral blood smear. “Early”, “initial”, and “durable” treatment responses were defined as platelet count >30 × 10 9 /L and at least doubling baseline at first week, first month and 6th month, respectively. Remission was defined as platelet count >100 × 10 9 /L at 12 months after treatment. Relapse was defined as a platelet count <100× 10 9 /L or a >50% decrease in a patient in remission. Bleedings that were life-threatening and required transfusion were considered major bleeding, and those that did not meet these criteria were considered minor bleeding.
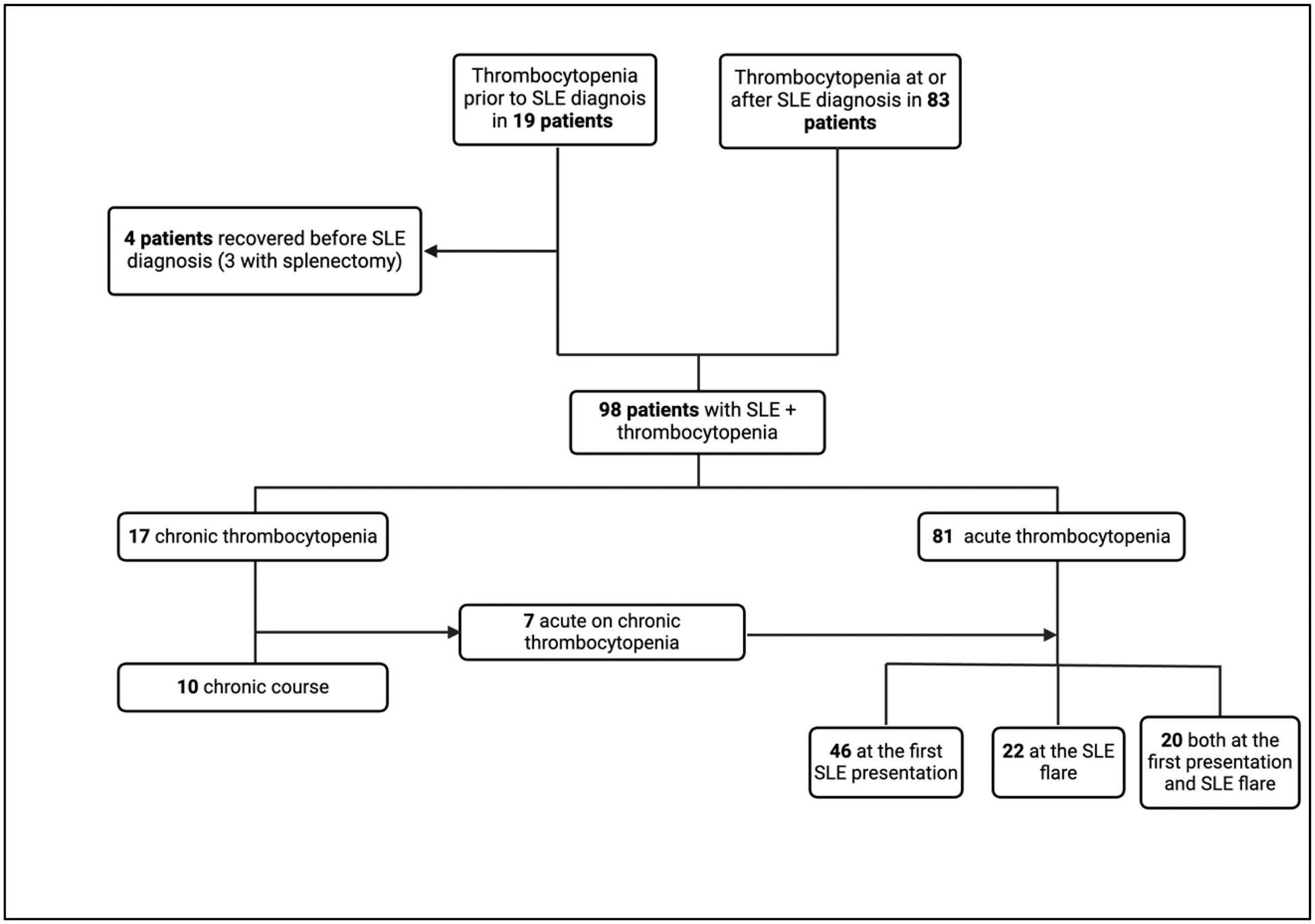
Results: Demographic, serological and laboratory characteristics of 102 patients are shown in Table 1. Four patients (3 with splenectomy) whose thrombocytopenia did not recur after SLE diagnosis were excluded and 98 patients were included in the analysis (Figure 1). The mean SLEDAI of the 66 patients with acute thrombocytopenia at SLE diagnosis was 16.8 (± 9.9), and 40 (60.2%) had a SLEDAI >12. Platelet count was ≤20 × 10 9 /L in 17 patients (25.7%) and >50 × 10 9 /L in 30 (45.5%). Sixty-five patients (98.5%) were managed with glucocorticoids, initiated with intravenous pulse in 32 (48.5%). IVIG was required in 4 (6.1%) patients, CYC was used in 21 (31.8%), AZA in 29 (43.9%) and MMF in 9 (13.6%). Early response was achieved in 21 (31.8%) patients, initial response in 50 (75.8%), and durable response in 60 (90.9%). There was at least one relapse in 38 patients. At the first relapse, rituximab was administered to 9 of 25 patients, and 7 of them did not relapse again. Total 5 patients underwent splenectomy due to refractory disease. Binary logistic regression analysis showed that aPL positivity was a risk factor for relapse [OR (95% CI): 3.8 (1.4-10.3), P =0.009]. Minor bleeding occurred in 24 patients and major bleeding in 13. Median SLICC damage index (SDI) was 1 (IQR:0-3) and 65 patients (63.7%) had SDI ≥1. Patients with at least one relapse had higher SDI than those with no relapse (mean SDI: 2.7 (± 2.4) vs 1.4 (±2.0), P =0.008).
Conclusion: Thrombocytopenia can be detected at the time of SLE diagnosis or during the course of the disease and even it may precede the SLE diagnosis. Patients with aPL positivity have higher risk for relapse. Prevention of relapses should be aimed as relapse is associated with higher damage.
Demographic, clinical and laboratory characteristics of the cohort
| All ( n =102) | All ( n =102) | ||
|---|---|---|---|
| Age at diagnosis (years), median (IQR ) | 28 (21-38.25) | Hemolytic anemia | 27 (26.5) |
| Duration of disease (months), median (IQR ) | 143.5 (81.75-240.25) | Leukopenia | 52 (51) |
| Female, n (% ) | 90 (88.2) | Neuropsychiatric | 23 (22.5) |
| SLE + aPL, n (% ) | 31 (30.4) | Laboratory findings, n (% ) | |
| SLE + APS, n (% ) | ANA | 99 (97.1) | |
| Thrombotic | 12 (11.8) | Anti-dsDNA | 75 (73.5) |
| Obstetric | 5 (4.9) (71.6 ) | Anti-Sm | 21 (20.6) |
| Thrombotic + obstetric | 8 (7.8) | Hypocomplementemia | 79 (77.5) |
| Clinical manifestations, n (% ) | 73 (71.6 ) | Positive Direct Antiglobulin test | 16 (15.7) |
| Malar rash | 51 (50) | Lupus anticoagulant | 39 (38.2) |
| Alopecia | 64 (62.7) | aCL IgG >40 | 18 (17.6) |
| Discoid rash | 4 (3.9) | aCL IgG 20-40 | 15 (14.7) |
| Oral/ nasal ulcers | 33 (32.4) | aCL IgM >40 | 19 (18.6) |
| Arthritis | 58 (56.9) | aCL IgM 20-40 | 16 (15.7) |
| Pleuritis | 12 (11.8) | aβ2GPI IgG | 11 (10.8) |
| Pericarditis | 12 (11.8) | aβ2GPI IgM | 11 (10.8) |
| Lupus nephritis | 42 (41.2) |
Study Flow Chart
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (