

Background: In rheumatoid arthritis (RA) management, five Janus kinase (JAK) inhibitors are available in Japan. However, there is limited evidence comparing the long-term retention rates and efficacy of these agents, considering the presence or absence of rheumatoid factor (RF) and anti-cyclic citrullinated peptide antibody (CCP).
Objectives: The purpose of this study was to examine the continuation rates of five JAK inhibitors in patients with and without autoantibodies. Reasons for discontinuation of each JAK inhibitors were also examined.
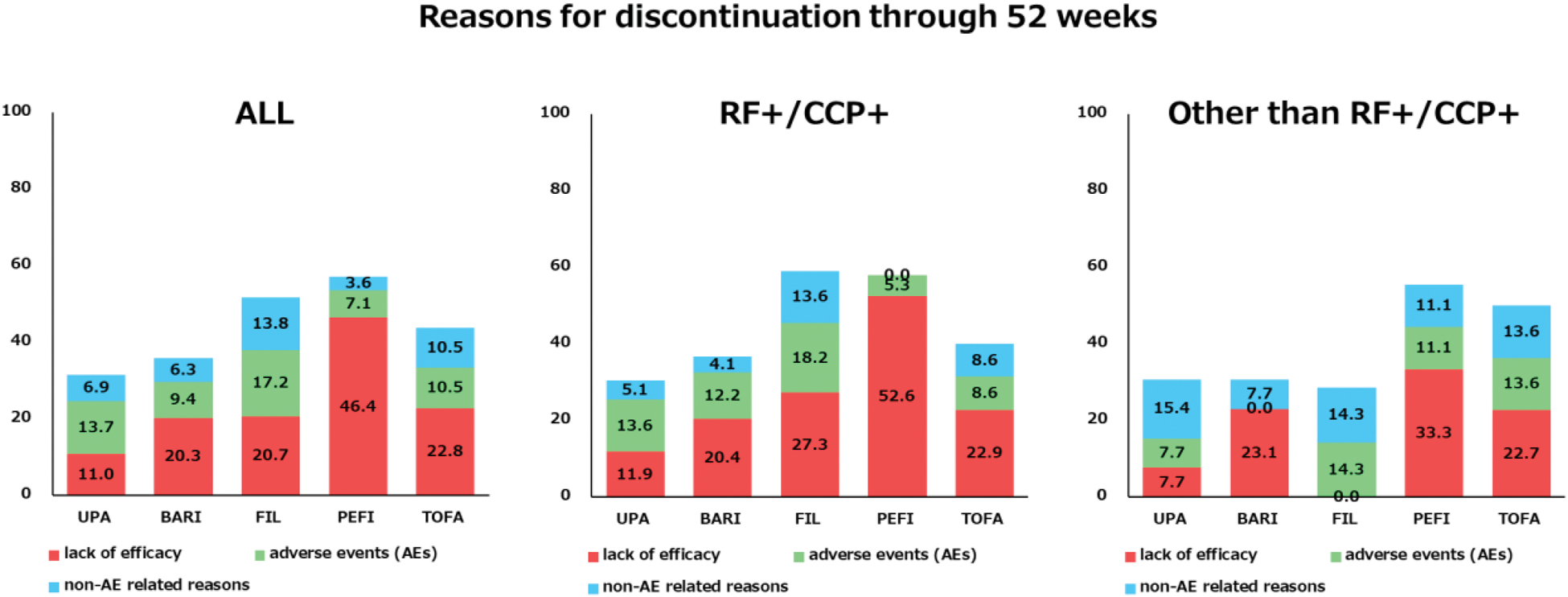
Methods: A total of 251 patients treated with JAK inhibitors were included from the Osaka Metropolitan University BIO/JAK database, which includes over 1,600 cases. Patients were categorized based on the JAK inhibitor: tofacitinib (TOFA, n=57), baricitinib (BARI, n=64), upadacitinib (UPA, n=73), peficitinib (PEFI, n=28), and filgotinib (FIL, n=29). Retention rates at 52 weeks and long-term retention rates were analyzed. Reasons for discontinuation were classified into three categories: lack of efficacy, adverse events (AEs), and non-AE related reasons. Patients were also stratified into RF+/CCP+ and other subgroups for further comparison.
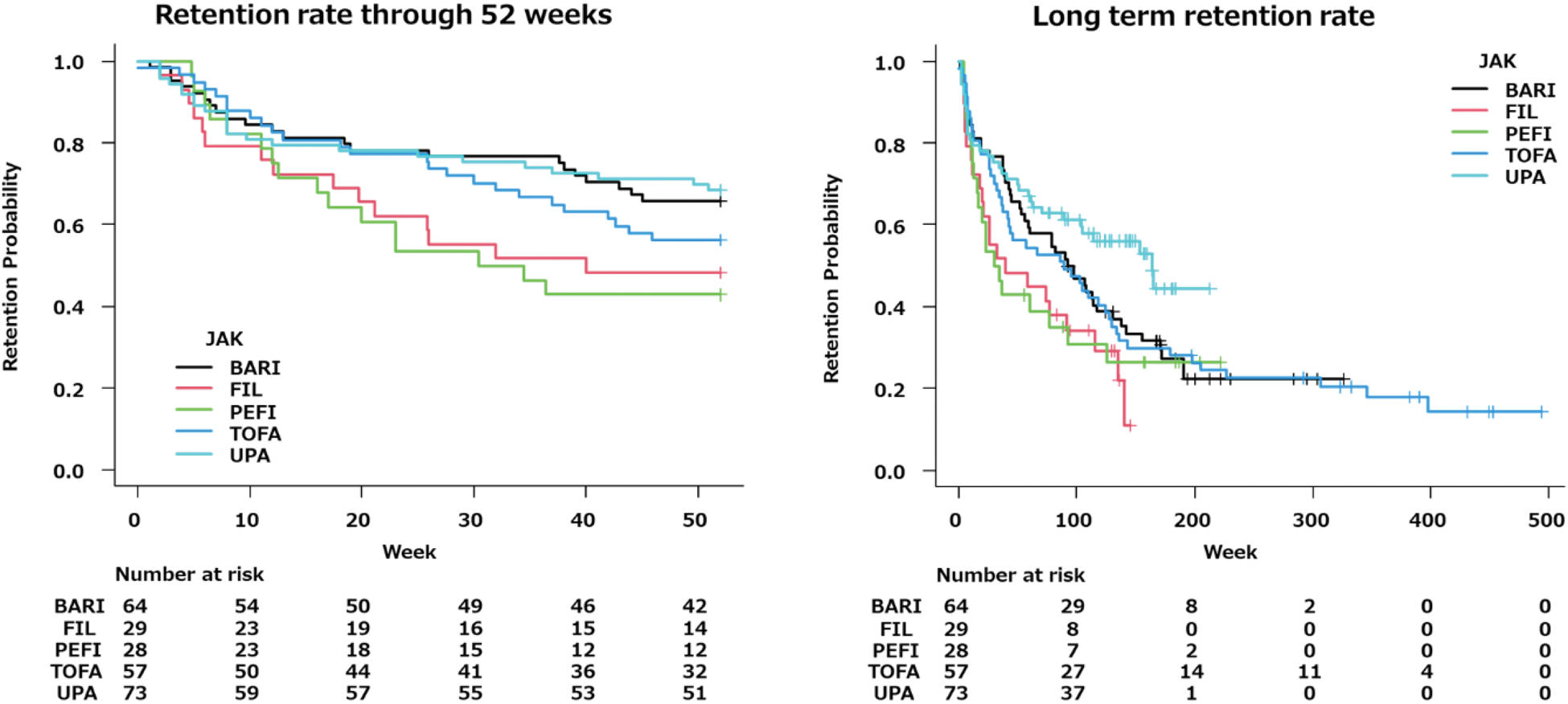
Results: The 52 weeks retention rates were as follows:
TOFA: 56.2%
BARI: 64.1%
UPA: 68.5%
PEFI: 42.9%
FIL: 48.3%
The proportion of patients achieving low disease activity (CDAI) at 52 weeks were as follows:
TOFA: 28.1%
BARI: 48.4%
UPA: 42.5%
PEFI: 32.1%
FIL: 13.8%
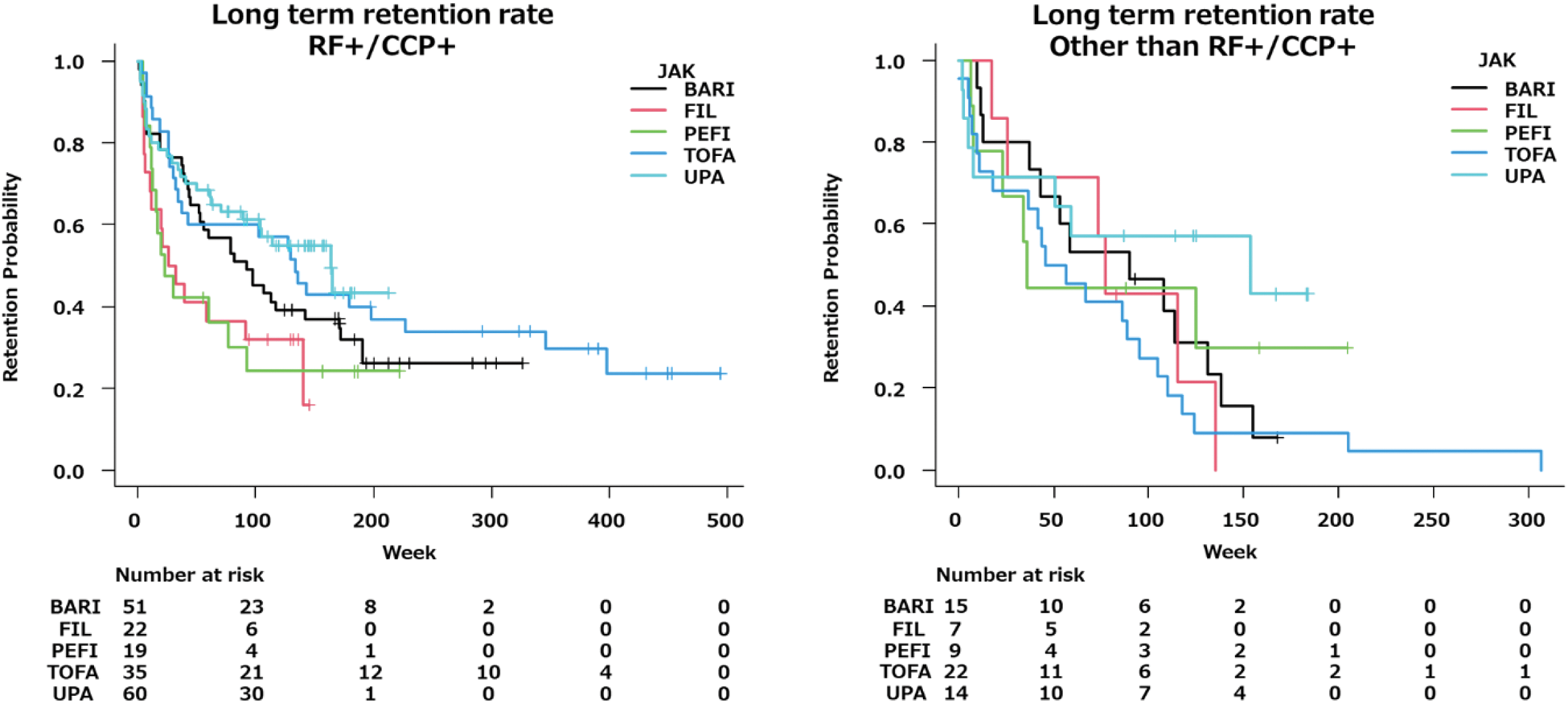
BARI and UPA demonstrated higher retention rates. However, long term retention rates were superior in UPA. In addition, UPA had superior long-term persistence rates in both seropositive and seronegative studies in the presence or absence of autoantibodies. UPA showed the lowest rate of discontinuation due to lack of efficacy at 52 weeks and the highest long-term retention rate. In the other than RF+/CCP+ subgroup, UPA and FIL showed lower rates of discontinuation due to lack of efficacy at 52 weeks. Over the long term, UPA demonstrated the lowest rate of discontinuation due to lack of efficacy and the highest retention rate, comparable to that of the RF+/CCP+ group.
The 52-week and long-term retention rates of five JAK inhibitors
The long-term retention rates of five JAK inhibitors with and without autoantibodies.
Reasons for discontinuation through 52 weeks.
Conclusion: This study highlights differences in efficacy and retention rates among the five JAK inhibitors, with a focus on autoantibody status. UPA showed superior retention rates and efficacy, particularly in seronegative patients, aligning with its indication for psoriatic arthritis. These findings underscore the importance of considering autoantibody status when selecting JAK inhibitors for RA management.
REFERENCES: [1] Sugawara M, Fujieda Y, Noguchi A, et al. Prediction of the intolerance or non-responder to Janus kinase inhibitors in patients with rheumatoid arthritis: a preliminary retrospective study with integrative cluster analysis. Clinical and Experimental Rheumatology 2022;40:1674-80.
Acknowledgements: NIL.
Disclosure of Interests: Tadashi Okano Abbvie, Tanabe Mitsubishi, Asahikasei, Janssen, Novartis, UCB, Eli Lilly, Astellas, Eisai, Kenji Mamoto: None declared, Yutaro Yamada: None declared, Takahito Kojima: None declared, Tomokazu Choshi: None declared, Masahiro Tada: None declared, Kentaro Inui: None declared, Tatsuya Koike: None declared., Hidetomi Terai: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (