

Background: Systemic Lupus Erythematosus (SLE) is a complex autoimmune disease characterized by multisystem involvement. Anifrolumab, a monoclonal antibody targeting the type I interferon receptor, represents a new target therapy for SLE. In Italy, the drug was approved at the end of March 2023 and is indicated for patients with refractory or moderately severe SLE, despite ongoing therapy, with a SLEDAI-2K score greater than 6. Limited real-world data are available.
Objectives: To evaluate the efficacy and safety of anifrolumab in a cohort of SLE patients.
Methods: From a cohort of 446 active SLE patients at our center, those who started anifrolumab therapy by December 31, 2024, were selected. Demographic, clinical, therapeutic, and disease activity data (SLEDAI-2K and SLE-DAS) were collected. Adverse events, including infections and therapy discontinuations, were monitored. In patients with a follow-up of at least 6 months, disease activity (SLEDAI-2K and SLE-DAS), serology (C3 and C4 levels, and anti-dsDNA positivity), daily glucocorticoid dose, hemoglobin levels, and platelet counts were also evaluated longitudinally. To achieve this, data were collected three months before starting the drug (T-3), at treatment initiation (T0), after one month (T1), after three months (T2), and six months after starting the drug (T3). Continuous variables were presented as means ± SD (for normally distributed data). The comparison between T-3 and T0 was performed using Student’s t-test for paired samples. To analyze trends at T0, T1, T2, and T3, general linear models for repeated measures were applied to continuous variables, with significance assessed using Pillai’s trace test. The analysis was complemented by pairwise comparisons. A two-sided alpha level of 0.05 was used for all inferential tests.
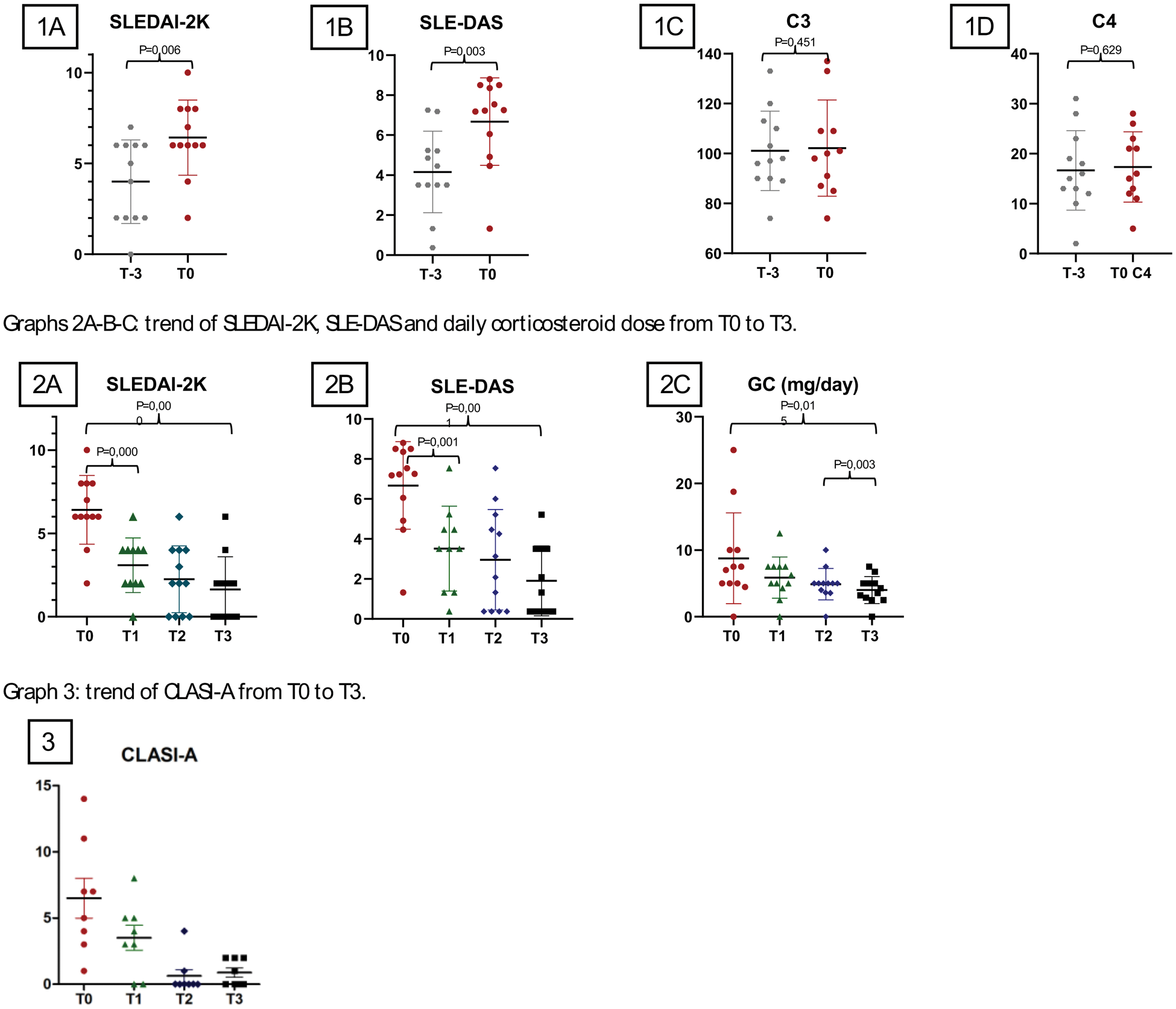
Results: From the cohort of 446 active SLE patients at our center, 22 patients (4,93%) met the inclusion criteria. Of these 22 patients on anifrolumab, the majority were female (19; F:M ratio of 6,3:1) and of Caucasian ethnicity (17; 77,27%). The mean age at treatment initiation was 39,14±14,64 years, with a mean disease duration of 13,45±12,24 years. Cumulative disease manifestations were primarily mucocutaneous (21; 95,45%) and articular (19; 86,36%), with less frequent renal (3; 13,64%) and hematological (12; 54,55%) involvement. Anifrolumab was primarily started for mucocutaneous (20; 90,91%) and articular (13; 59,09%) involvement. For 4 patients (18,18%), it was their first biologic therapy, while the remaining 18 patients (81,82%) had failed other biologic therapies, with an average of 2,55±1,44 DMARDs used before starting anifrolumab. At baseline, the mean corticosteroid dose was 8,56±7,73 mg/day, with moderate disease activity indicated by mean SLEDAI-2K (6,22±2,25) and SLE-DAS (6,86±2,35) scores. The mean duration of therapy was 6,77±5,00 months. There were 10 (45.45%) patients vaccinated for herpes zoster (HZ) before the start of anifrolumab. Ten infectious events were reported, including 6 respiratory infections (specifically 4 pneumonias), affecting 9 patients (40,91%). Three cases required hospitalization (13,64%): two pneumonias with respiratory failure and one pyelonephritis. No patients developed HZ. There were five temporary suspensions of anifrolumab (22.73%), while three were permanently discontinued (13.64%): two for inefficacy and one for unplanned pregnancy. In patients with a follow-up of at least six months (12; 54,55%), disease activity, C3 and C4 levels in the three months prior to drug start were assessed, and it was found that disease activity estimated by SLEDAI-2K and SLE-DAS was significantly higher at drug initiation (T0, P=0,006 and P=0,003); no differences emerged related to complement levels (Graphs 1A-B-C-D) and anti-dsDNA positivity. A significant reduction in disease activity, as assessed by SLEDAI-2K and SLE-DAS scores (P=0,000 and P=0,001), was observed after drug initiation, and the significant difference was already evident after one month of treatment (P=0,000 and P=0,001). Daily corticosteroid doses also decreased significantly over six months (P=0,015), particularly from three months after the start of treatment (T2 vs T3; P=0,03) (Graphs 2A-B-C). No significant changes were observed in hemoglobin levels, leukocyte or platelet counts, or serologically (complement levels and anti-dsDNA positivity). Due to the limited number of patients with skin involvement and a follow-up of more than six months, it was not possible to assess significant differences in skin disease (CLASI scores). However, the mean CLASI-A score at the beginning of treatment was 6,5±4,28, which decreased significantly to 0,88±0,99 after six months (Graph 3).
Conclusion: In this preliminary analysis of a single center cohort, anifrolumab demonstrated significant efficacy in reducing disease activity in patients with (SLE), as measured by SLEDAI-2K and SLE-DAS scores, with notable improvements observed as early as one month after treatment initiation. Furthermore, the therapy allowed a significant reduction in daily corticosteroid use, particularly from the third month of treatment onward. While the safety profile was generally consistent with expectations, infections, including severe cases requiring hospitalization, were observed in a notable proportion of patients. This highlights the importance of close monitoring during treatment. Due to the limited sample size and the short duration of follow-up, further studies with larger cohorts and longer observation periods are needed to confirm these findings and to assess the long-term efficacy and safety of anifrolumab.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (