

Background: The control of the Systemic Lupus Erythematosus (SLE) faces many challenges. According to EULAR recommendations, achieving remission or low disease activity should be the main ambition [1]. However, the chronicity and complexity of the disease, characterized by unexpected flares and asymptomatic periods, hinders the chances to accomplish low disease activity and to prevent organ damage. The accumulated organ damage and the adverse effects of the available treatments increase the risk of infections and premature death [2]. Importantly, the consequences are also perceptible in the quality of life and mental health impacting the daily life of patients dealing with the disease [3].
Objectives: Primarily, this study aimed to describe organ damage, remission achievement and SLE treatment in a sub-cohort of Spanish patients with moderate to severe SLE included in the SPOCS (SLE Prospective Observational Cohort Study). Secondly, we aimed to describe the quality of life and depression in the same cohort.
Methods: Patients with moderate and severe SLE patients were recruited between 2017 to 2019 in 18 sites in Spain and followed-up biannually for a 36-month period, until 2021. The routine visit, at the time consent is signed, will be treated as the baseline visit. Moderate and severe criteria were SLEDAI-2K score ≥6 points or modified SLEDAI-2K score ≥4. Organ damage was reported using SLICC/ACR Damage Index (SDI). Remission was defined using 2021 DORIS criteria and low disease activity by LLDAS. Quality of life, mental health and fatigue were measured using EQ-5L, PHQ-8 and FACIT-F respectively. Data was reported by absolute and relative frequencies for categorical variables and mean and standard deviation (SD) for continuous variables.
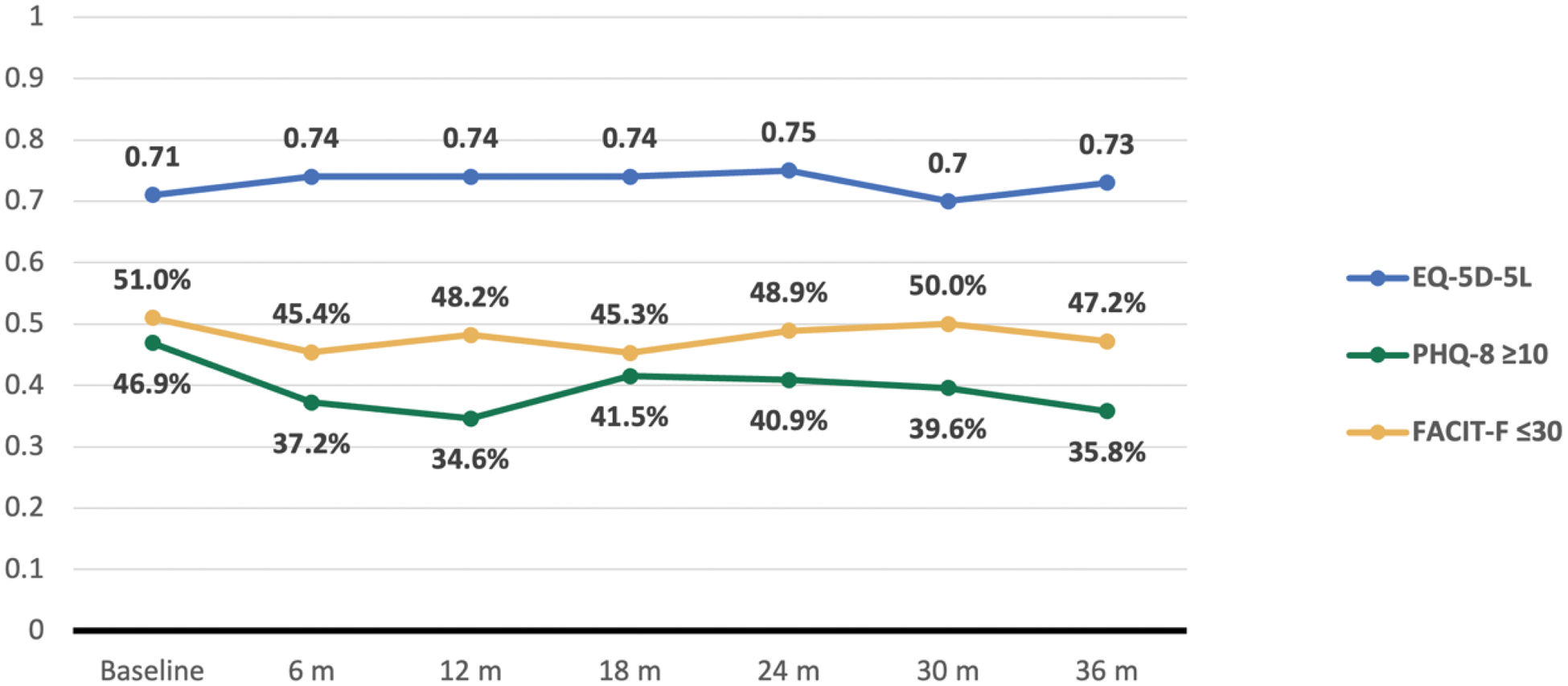
Results: Among 99 patients at baseline, mean (SD) age was 45 (12.0) years and 91 (92%) were female. Mean (SD) baseline oral corticosteroids dose was 5.4 (4.6) mg/day of prednisone. A total of 45% of the patients presented any organ damage at baseline and more than 10% accrued new organ damage after 12 months. At all-time points during follow-up, less than 15% and 25% of patients achieved remission and LLDAS, respectively. SLEDAI-2K indicated that at least 30% of patients had moderate to severe disease activity during any time of follow-up (SLEDAI-2K≥6). Hydroxychloroquine was the most frequent medication, followed by corticosteroids and immunosuppressants (Table 1). Regarding quality of life (QoL), more than 50% of the patients reported impact on the domain of pain and discomfort, usual activities and anxiety and depression during the study. Mean (SD) PHQ-8 were 9.3 (5.9), and 74% presented depression (PHQ-8 ≥5), 46.9% were moderate-severe (PHQ-8 ≥10). Severe fatigue (FACIT-F<30) was reported at baseline in 48.9% of the patients reaching 52.8% at the end of the follow-up (Figure 1).
Disease characteristics of the Spanish SPOCS cohort over time.
| Baseline
| 6 months (N=89) | 12 months (N=57) | 18 months (N=54) | 24 months (N=60) | 30 months (N=56) | 36 months (N=58) | |
|---|---|---|---|---|---|---|---|
| Any organ damage a , n (% ) | 44 (45) | 27 (51) | 23 (62) | 26 (62) | |||
| LLDAS, n (% ) | 19 (23.5) | 4 (8.0) | 8 (15.4) | 7 (14.3) | 3 (6.1) | 11 (22.0) | |
| Remission, n (% ) | 11 (12.6) | 3 (5.3) | 3 (5.8) | 7 (12.7) | 3 (5.8) | 5 (9.8) | |
| SLEDAI-2K ≥6, n (% ) | 81 (98.8) | 21 (35.6) | 15 (50) | 17 (42.5) | 12 (34.3) | 14 (38.9) | 11 (29.7) |
| Medication use, n (% ) | |||||||
| Antimalarials | 87 (87.9) | 77 (86.5) | 50 (87.7) | 47 (87.0) | 53 (88.3) | 48 (85.7) | 52 (89.7) |
| Glucocorticosteroids | 30 (42.3) | 33 (51.6) | 19 (47.5) | 11 (42.3) | 11 (39.3) | 11 (44.0) | 14 (53.8) |
| Immunosupressants | 50 (50.5) | 48 (53.9) | 33 (57.9) | 30 (55.6) | 34 (56.7) | 28 (50.0) | 33 (56.9) |
| Biologics | 17 (17.2) | 19 (21.3) | 13 (22.8) | 11 (20.4) | 14 (23.3) | 14 (25.0) | 13 (22.4) |
Organ damage was measured annually.
Between 0 and ≤ 5 mg/day, considering the 71 patients that were taking glucocorticosteroids
Patients’ self-reported outcomes related to quality of life (EQ-5D-5L), depression (PHQ-8) and fatigue (FACIT-F).
PHQ-8 and FACIT are reported using percentages while EQ-5D-5L is reported using the mean for the whole sample available.
EQ-5D-5L index value ranges from 0 (dead) to 1 (full health). PHQ-8 score ≥5 indicates depression, ≥10 indicates severe depression. FACIT-F total scores: range from 0 to 52, with lower scores indicating greater fatigue and higher scores indicating less fatigue. A score less than 30 indicates severe fatigue.
Abbreviations: FACIT-F: Functional Assessment of Chronic Illness Therapy – Fatigue; PHQ-8: Personal Health Questionnaire Depression Scale.
Conclusion: In a Spanish cohort of moderate-severe SLE achieving the EULAR objectives (namely, remission and LLDAS) and reducing the progression of organ damage, including corticosteroid use, remained the main challenges during a three-year period, reflecting an important burden of disease accompanied by negative impact in QoL, not only concerning the physical impact but also considering the mental health and quality of life. New therapeutic and preventive strategies to face the current challenges of the disease are necessary for SLE patients.
REFERENCES: [1] Fanouriakis A, Kostopoulou M, Andersen J, Aringer M, Arnaud L, Bae SC, et al. EULAR recommendations for the management of systemic lupus erythematosus: 2023 update. Ann Rheum Dis. 2024 Jan;83(1):15–29.
[2] Ajeganova S, Hafström I, Frostegård J. Patients with SLE have higher risk of cardiovascular events and mortality in comparison with controls with the same levels of traditional risk factors and intima-media measures, which is related to accumulated disease damage and antiphospholipid syndrome: a case-control study over 10 years. Lupus Sci Med [Internet]. 2021 Feb 5;8(1).
[3] Schmeding A, Schneider M. Fatigue, health-related quality of life and other patient-reported outcomes in systemic lupus erythematosus. Best Pract Res Clin Rheumatol. 2013 Jun;27(3):363–75.
Acknowledgements: NIL.
Disclosure of Interests: Íñigo Jesus Rúa-Figueroa: None declared, Mercedes Freire González: None declared, Tarek Carlos Salman-Monte: None declared, Ricard Cervera Segura: None declared, Ana Isabel Turrión: None declared, Ivan Castellví: None declared, Jaime Calvo Alén: None declared, Julia Barbado Ajo: None declared, Montserrat Díaz: None declared, Norberto Ortego-Centeno: None declared, Nuria Lozano Rivas: None declared, Josefina Cortés-Hernández: None declared, Paula Álvarez-Hernández Current employee of AstraZeneca, Marta Galvez-Fernandez Current employee of AstraZeneca, Noemí Bahamontes Current employee of AstraZeneca, Bo Ding Current employee of AstraZeneca, José M. Pego-Reigosa: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (