

Background: Data on cardiac involvement in idiopathic inflammatory myopathies (IIM) remain limited. Autopsy studies have identified acute inflammatory processes in the myocardium of patients with IIM, followed by fibrotic changes that lead to conduction defects [1, 2]. However, the available information on cardiac involvement is limited, primarily due to the rarity and heterogeneity of the disease, as well as the rare occurrence of overt cardiac complications in these patients. The myositis-associated antibody (MAA) Ro52 is among the most frequent autoantibodies in IIM patients, with a prevalence ranging from 10% to 40% [3]. It is known that congenital heart block is associated with maternal positivity for anti-Ro52 [4], but the role of this antibody in adult patients with systemic lupus erythematosus and IIM remains poorly defined. Growing evidence from clinical and experimental studies suggests that anti-Ro/SSA antibodies may significantly affect ventricular repolarization through a direct inhibitory reaction with the extracellular pore region of the cardiac hERG-K+ channel, thus increasing the predisposition to the development of long QT syndrome and ventricular arrhythmias in patients positive for these antibodies [5]. A recent Japanese study has indeed shown QTc interval prolongation in patients with dermatomyositis positive for anti-Ro52 compared to those with DM negative for Ro52, as well as in DM patients versus healthy controls [6].
Objectives: This study aims to evaluate whether anti-Ro-52 positivity is closely correlated with electrocardiographic alterations in patients with IIM.
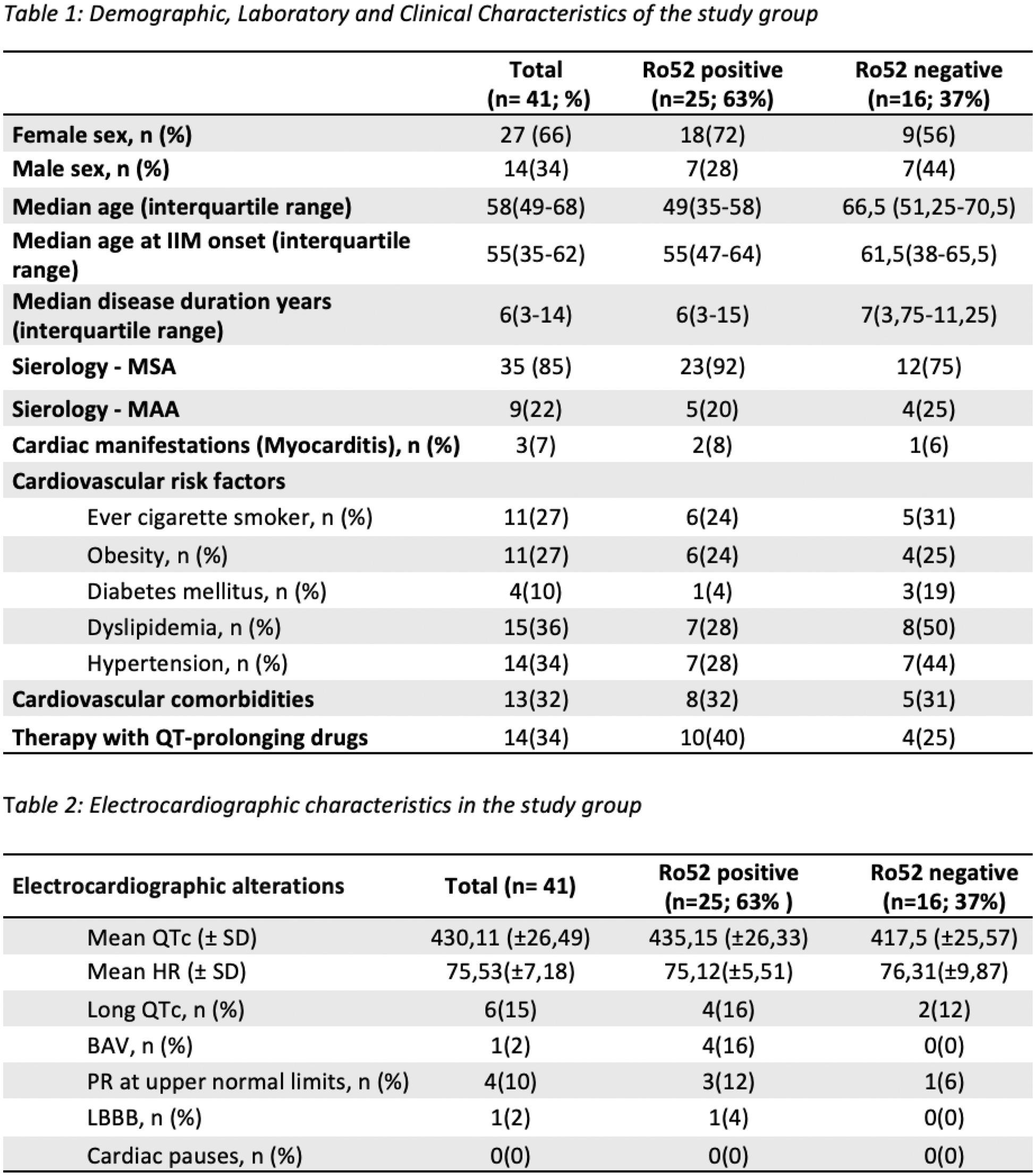
Methods: Forty-one patients with IIM were enrolled [11 polymyositis, 11 dermatomyositis, 5 clinically amyopathic dermatomyositis, 12 antisynthetase syndrome, and 2 immune-mediated necrotizing myopathy; 14 males and 27 females]. The median disease duration at enrollment was 6 years (3-14), the median age at diagnosis was 55 years (35-62), and the median age at enrollment was 58 years (49-68). All patients were positive for myositis-specific antibodies (MSA) or myositis-associated antibodies (MAA). Only three patients had a known clinical history of cardiac involvement, diagnosed as myocarditis (2 positive and 1 negative for anti-Ro52). The population was divided into two groups based on anti-Ro52 positivity (n = 25; M = 7; F = 18) or negativity (n = 16; M = 7; F = 9). All patients underwent 24-hour Holter ECG monitoring, with subsequent analysis using the Cubeholter WS software, trace review by a cardiology specialist, and QTc interval calculation using the Bazett formula. A QTc value of 440 ms for men and 460 ms for women was considered the upper normal limit. Fourteen patients (10 positive and 4 negative for anti-Ro52) were receiving treatment with drugs that are known to potentially prolong the QTc interval at the time of the 24-hour Holter ECG monitoring.
Results: All tracings show sinus rhythm with an average heart rate (HR) of 75,53 (± 7,18 SD) without difference between anti-Ro52 negative (mean average HR 75,12 ±5,51) and anti-Ro52 positive group (mean average HR 76,31 ±9,87). No pathological pauses were recorded in any patients. A first-degree atrioventricular block was recorded in one patient with anti-Ro52. In 4 patients the PR interval was at the upper limits of normal (3 anti-Ro52 positive and 1 anti-Ro52 negative). One case of left bundle branch block in an anti-Ro52 positive patient. The QTc was prolonged in four anti-Ro52 positive patients and two anti-Ro52 negative patient. The QTc length showed no significant difference between the groups (P= 0.1294). Age, disease duration, gender, cardiovascular comorbidities and cardiovascular risk factors (smoking, blood pressure, obesity, diabetes mellitus and dyslipidemia) were similar between the two groups (P>0.05).
Conclusion: This study has provided important insights into the electrocardiographic alterations in patients with IIM, with particular focus on the influence of anti-Ro52 antibody positivity. Although no statistically significant differences were observed in QTc interval prolongation between anti-Ro52 positive and negative patients, a prolonged QTc was observed in both groups, suggesting potential cardiovascular implications in patients with IIM. Additionally, some electrocardiographic abnormalities, such as first-degree atrioventricular block and left bundle branch block, were more frequent in anti-Ro52 positive patients, indicating a potential role of this antibody in conduction disturbances. Currently, our patient sample is relatively small, which may explain the discrepancy between the results obtained in our study and those reported in the literature, where a stronger correlation between anti-Ro52 positivity and QTc prolongation is often observed. Therefore, it is crucial to conduct future studies with larger sample sizes and longer follow-up periods to confirm these preliminary findings and further explore the role of anti-Ro52 antibodies in cardiac involvement in IIM, thus improving the clinical management of these patients.
REFERENCES: [1] Gupta, Raghav et al. International journal of cardiology vol. 148,3 (2011): 261-70.
[2] Schwartz, Thomas et al. RMD open vol. 2,2 e000291. 27 Sep. 2016.
[3] Lundberg, Ingrid E et al. Nature reviews. Disease primers vol. 7,1 86. 2 Dec. 2021.
[4] Buyon, J P et al. Journal of the American College of Cardiology vol. 31,7 (1998): 1658-66.
[5] Lazzerini, Pietro Enea et al. Circulation. Arrhythmia and electrophysiology vol. 9,4 (2016): e003419.
[6] Wang, Hui, and Ping Lin. Heart and vessels vol. 39,11 (2024): 979-987.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (