

Background: Posterior reversible encephalopathy syndrome (PRES) is a well-known but rare complication in patients with systemic lupus erythematosus (SLE), observed in <1% of patients.
Objectives: To describe the clinical features and presentation patterns, potential predictors and precipitating factors and outcomes of PRES in SLE.
Methods: We reviewed Electronic Medical Records (EMR) of patients with SLE diagnosed as PRES between January 2005 and July 2024 and admitted to the wards for management. We enetered data of all 28 patient in 4 different time points- before PRES event at base line visit and during admission with PRES, 6 months after discharge following PRES and at their latest visit in hospital. Clinical variables, disease activity, serological profile, investigation and MRI variables were noted from the EMR and picture archiving and communication system (PACS), respectively.
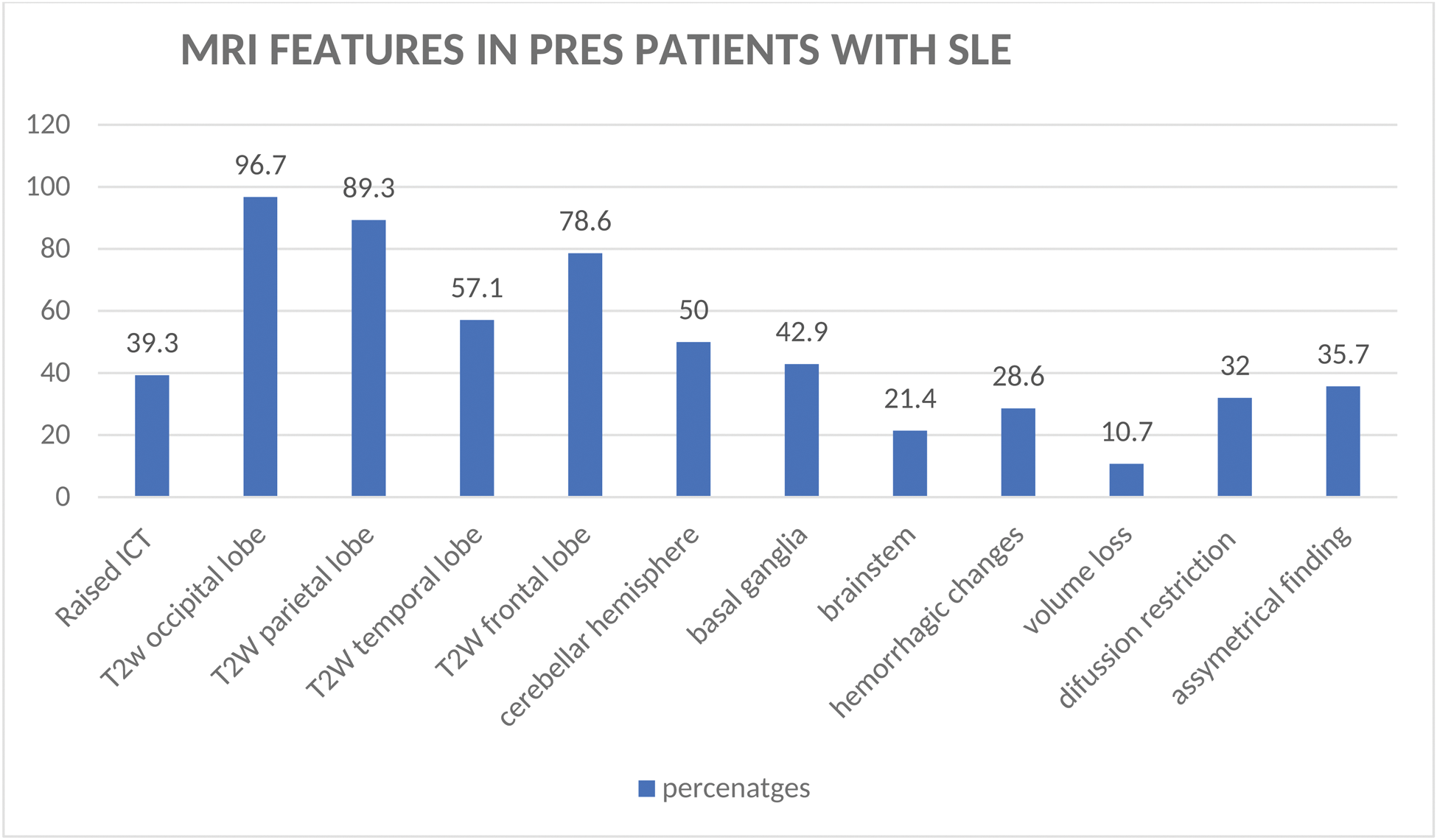
Results: We identified around 28 female patients over the last 19 years. Among the comorbidities, diabetes was present in 5 patients (17.9%), hypertension in 9 patients (32.1%), and hypothyroidism in 5 patients (17.9). PRES occurred at initial SLE diagnosis in 10.7% of cases. 32% had pre-existing hypertension, and one patient developed PRES in the postpartum period. New-onset hypertension was noted in 24 (72 %) patients, while 5 (15%) were normotensive at presentation. Seizures (92%) and headaches (100%) were the most common symptoms, with focal neurological deficits in 10.7%, visual impairment in 42%, and altered sensorium in 35%. Significant proteinuria and active urinary sediments were observed in 78% of patients, while 60.7% had concurrent infections, most commonly urinary tract infections (UTIs). The mean systolic blood pressure at the time of PRES was 153 mmHg (SD ± 20), while the mean diastolic blood pressure was 92 mmHg (SD ± 11.2). The median SLEDAI score of patients admitted with PRES was 20 (IQR: 14–22). The median CRP was 13, albumin was 2.4, and creatinine was 1.3 at the time of PRES in these patients, which normalized on subsequent visits. The median dsDNA was 447(67-897) during PRES. The antibody profile showed 39% SSA+, 25% APLA+, 39% DCT+, and 2 patients were ANA negative. The MRI features during PRES are detailed in Figure 1. The treatments given for patients with PRES are outlined in Table 1. In terms of treatment during admission with PRES, 39% of patients received cyclophosphamide, 42% received mycophenolate mofetil, and 7.2% received rituximab. Additionally, 64% of patients were treated with intravenous corticosteroids, with a median prednisolone dose of 50 mg/day (IQR: 26–57 mg/day). Two patients (3%) with SLE experienced a PRES relapse within six months. Of the 28 patients, 3 (10.7%) died following PRES: All three fatalities were associated with atypical features in MRI imaging of the brain. Among the SLE patients who died, two received 2g/day of mycophenolic acid and one received Rituximab, along with 1 mg/kg oral prednisolone and mycophenolic acid for 1 year before the PRES. All three of these patients received adequate immunosuppression during the PRES event.
Conclusion: Over a median 33 months follow up 10.7% mortality, 7.1% relapse; 89.2% improved clinically, 83% had radiological resolution, showing favorable outcomes in long term for PRES patients with SLE.Atypical MRI findings were common in PRES patients with SLE. PRES may present with normal blood pressure and can be a presenting feature in SLE. Thus, it could be considered as a neuropsychiatric manifestation of SLE.
Demographics, clinical and treatment details and outcomes in patients with PRES
| variable | Baseline visit | PRES admission
| 6months follow up after PRES
| Last visit in hospital
|
|---|---|---|---|---|
| Age, +SD (years) | 24±9.1 | 26±8.1 | 26±8.1 | 29.38±9.1 |
| Disease duration, Median (IQR) | 1(1-3) | 2(1-5) | 2(1-5) | 7(2-9) |
| Inflammatory arthritis, n (%) | 22(78.6%) | 13(46.4%) | 0 | 0 |
| Hematology manifestation, n(%) | 5(17.8%) | 7(24.9%) | 3(10.8%) | 1(4.3%) |
| Lupus nephritis related proteinuria, n (%) | 17(60.7%) | 22(78%) | 4(14.2%) | 4 |
| Cardiac involvement, n (%) | 5(17.9%) | 7 (25%) | 0 | 0 |
| C3, Median (IQR) | 43(30-75) | 43(31-70) | 99±29 | 102(82-116) |
| C4, Median (IQR) | 7(6-19) | 10(6-20) | 22916-32) | 29(11-36) |
| SLEDAI, Median (IQR) | 10(8-15) | 20(14-22) | 2(0-6) | 0(0-5.7) |
| dsDNA, Median (IQR) | 650 (271-947) | 447(67-897) | 127(3-413) | 119(11-258) |
| Prednisolone dose given, Median (IQR) | N=22
| N=28
| N=23
| N=23
|
| IV steroid, n (%) | 4(14.2%) | 18(64%) | 0 | 1(4.3%) |
| Cyclophosphamide, n (%) | 4(14.2%) | 10(35%) | 6(26%) | 4(17.3%) |
| Mycophenolate mofetil, n (%) | 11(39%) | 12(42.9%) | 18(78.2%) | 13(56%) |
| Azoran, n (%) | 2(7.1%) | 0 | 0 | 2(8.6%) |
| Rituximab, n (%) | 3(13.6%) | 4(14.3%) | 0 | 3(13%) |
| Tacrolimus, n (%) | 1(3.6%) | 0 | 0 | 2(8.6%) |
| Death, n (%) | 1 (3%) | 2(6%) | ||
| Cause of death | Patient 1- died due to raised intracranial tension, tonsilar herniation, PRES, SLE | Patient 2 (SLE): Cardiac failure and pulmonary nocardiosis
|
PRES -Posterior reversible encephalopathy, SLE -Sytemic lupus erythematosis, mean±SD, Median (IQR), frequency (%) SD-standard deviation, IQR interquartile range, IV -intravenous, SLEDAI -systemic lupus erythematosis activity index
MRI features of PRES in SLE patients
T2 hyperintensities were observed in the above areas with percentages mentioned, ICT- intracranial tension
REFERENCES: [1] Ando Y, Ono Y, Sano A, Fujita N, Ono S. Posterior Reversible Encephalopathy Syndrome: A Review of the Literature. Intern Med. 2022 Jan 15;61(2):135–41.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (