

Background: Hydroxychloroquine (HCQ) is the mainstay of treatment for systemic lupus erythematosus (SLE). However, HCQ can lead to risk of QT prolongation. The incidence of QT interval prolongation with HCQ use in SLE patients is not clear, and risk factors for QT prolongation are also unknown.
Objectives: The primary objective of this study was to determine the incidence of long QT syndrome with HCQ use in SLE patients. The secondary objective was to identify risk factors for long QT syndrome with HCQ use.
Methods: SLE patients treated with HCQ at our hospital between 2016 and 2024 were included in the analysis of this study. The QT interval (QTc: calculated using the Bazett formula) was measured from the 12-lead electrocardiogram (ECG) before and after HCQ administration (defined as ECG1 before HCQ administration and ECG2 after). SLE patients were divided into two groups according to the ECG2 QT interval results.: ECG2-QTc ≥ 440 ms (PG: prolonged group) and ECG2-QTc < 440 ms (non-PG: non-extended group). To identify risk factors for QT prolongation, age, gender, weight, estimated glomerular filtration rate (eGFR), comorbidities (cardiac disease, hypertension), concomitant medications, duration of HCQ treatment, daily HCQ dose, actual weight/day HCQ dose, and cumulative HCQ dose were retrospectively extracted from the medical records. Fischer’s exact test and Wilcoxon’s rank sum test were used for risk factor analysis. Additionally, the association between changes in serum S100A8 and S100A9, previously reported biomarkers of HCQ effectiveness, and QTc was analysed.
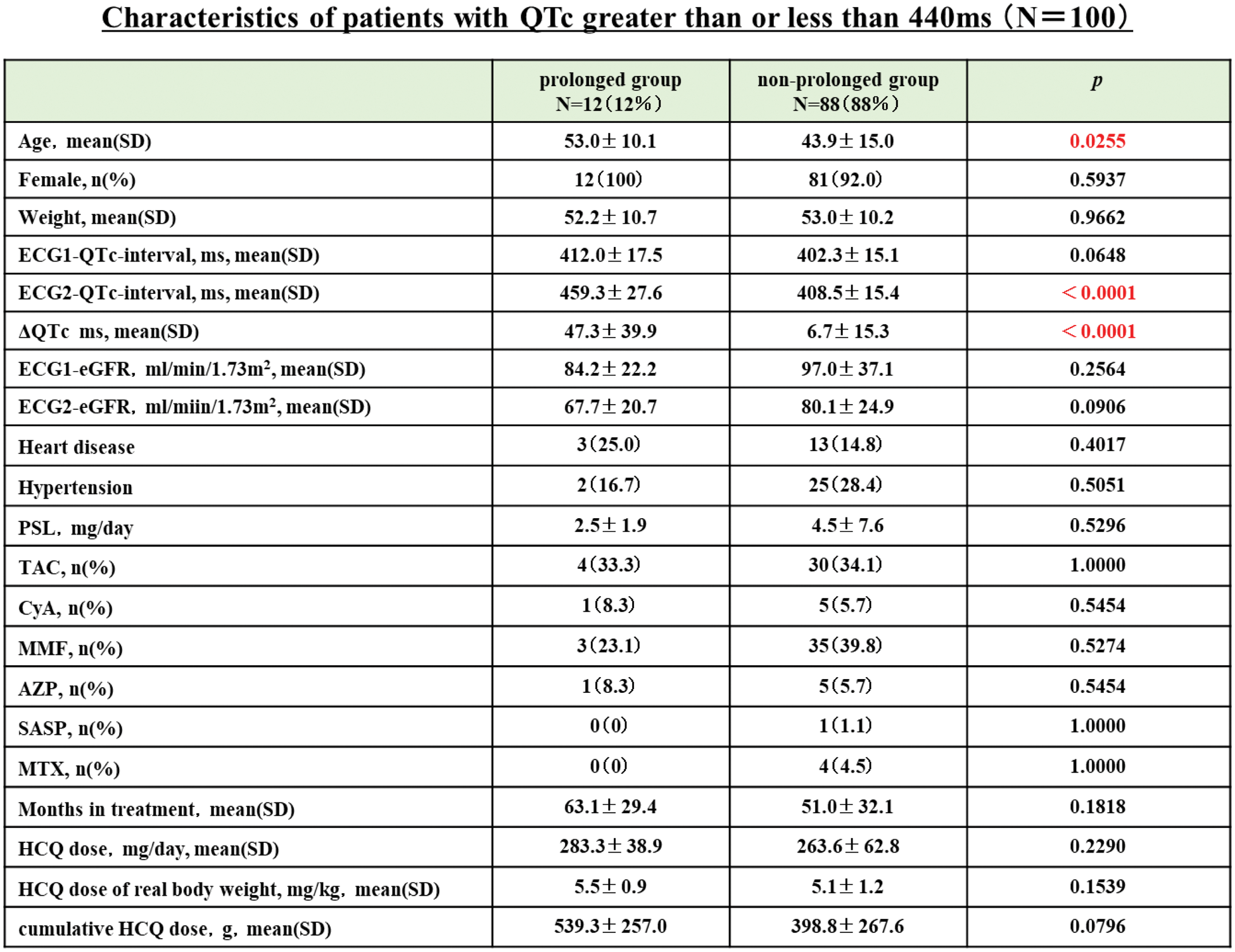
Results: One hundred SLE patients were included in the study. According to ECG2-QTc values, 12 (12.0%) patients were divided into the PG group and 88 (88.0%) into the non-PG group. Both groups showed significantly prolonged QTc after HCQ administration (p < 0.001, p = 0.006, respectively) (Figure 1A,1B), however, the difference in QTc (⊿QTc) was significantly greater in the PG than in the non-PG (47.3 ± 39.9 vs. 6.7 ± 15.3, p < 0.001). In the PG, QT prolongation with ⊿QTc ≥ 60 ms, known to be a risk factor for the development of TdP (2), was observed in 4 cases (33.3%), whereas it was not observed in the non-PG. The mean age was 53.0 ± 10.1 years in PG and 43.9 ± 15.0 years in non-PG, which was significantly higher in PG (p = 0.0255). Logistic regression analysis for long QT syndrome revealed the cut-off value of mean age as 40.0 years old. The mean eGFR in the PG was 67.7±20.7 ml/min/1.73m2 and 80.1±24.9 ml/min/1.73m2 in the non-PG, but there was no significant difference. There were no differences between the two groups in gender, body weight, comorbidities, concomitant medications, duration of HCQ treatment, daily HCQ dose, actual weight/day HCQ dose, and cumulative HCQ dose. In 70 cases where serum S100A8 and S100A9 could be analysed, the effect of HCQ on S100A8 and S100A9 was associated with QT prolongation.
Conclusion: HCQ treatment prolonged QT in almost all SLE patients, with 12% developing long QT syndrome and 4% at risk of developing TdP. Age has been identified as a risk factor for long QT syndrome, and it was found that age over 40 needs to be considered. Potential biomarkers for predicting QT prolongation include S100A8 and S100A9.
REFERENCES: [1] R Wakiya, et al. Hydroxychloroquine modulates elevated expression of S100 proteins in systemic lupus erythematosus. Lupus 2019 Jun;28(7):826-833.
[2] Woosley RL, et al. Mechanism of the cardiotoxic actions of terfenadine. JAMA 1993; 269: 1532-1536.
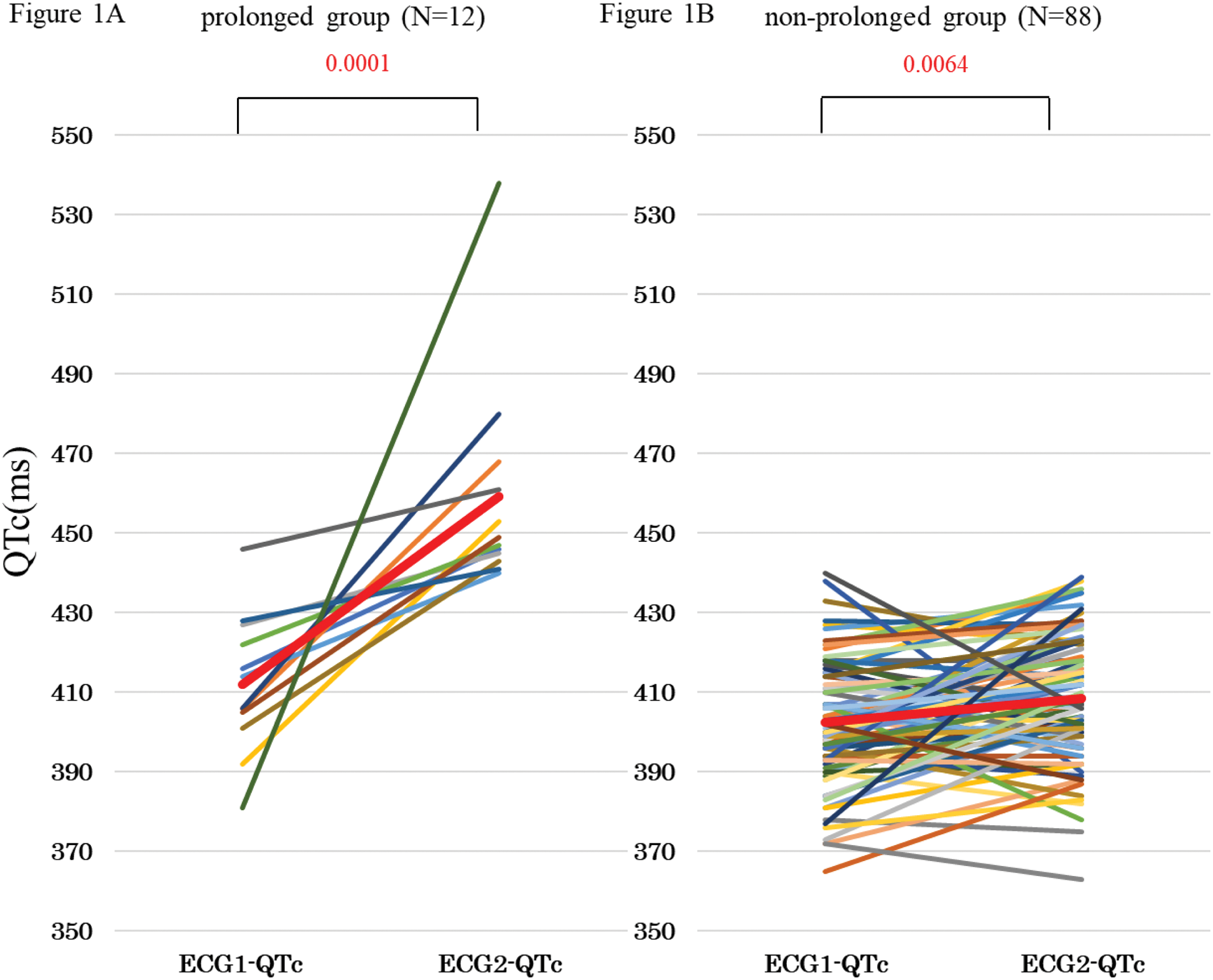
Table 1.
Acknowledgements: NIL.
Disclosure of Interests: Mao Mizusaki: None declared , Hiromi Shimada: None declared , Yusuke Ushio: None declared , Taichi Miyagi: None declared , Rina Mino: None declared , Kanako Chujo: None declared , Ryoko Kagawa: None declared , Naoto Manabe: None declared , Shusaku Nakashima: None declared , Koichi Sugihara: None declared , Risa Wakiya: None declared , Hiroaki Dobashi honoraria from Abbvie, Eli Lilly, Astellas, Ayumi Pharmaceutical, Chugai Pharmaceutical, Eisai Pharmaceutical, Asahi Kasei Pharma, UCB Pharmaceutica, and KISSEI Pharmaceutical, research grant from Asahi Kasei Pharma, Chugai Pharmaceutical, and AbbVie Japan.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (