

Background: Sjögren’s disease (SjD) is a systemic, chronic, and progressive autoimmune disease that presents with a wide spectrum of clinical manifestations. SjD is mostly characterized by dry mouth and eyes, but many patients experience fatigue, musculoskeletal pain, and other extraglandular manifestations affecting multiple organs and systems [1]. Due to its complex and variable presentation and lack of definitive diagnostic criteria, SjD can be difficult to diagnose and is poorly understood in rheumatology. Patients can be diagnosed with SjD in the absence of other diseases (formerly primary SjD), or they may have overlapping autoimmune diseases (formerly secondary SjD) such as systemic lupus erythematosus (SLE) or rheumatoid arthritis (RA) [2]. Treatment of patients with SjD is limited to symptomatic control with local and systemic therapies, as no curative or disease-modifying treatments are available [3]. Due to limited treatment options, patients with SjD experience significant clinical and subclinical burden.
Objectives: To characterize the clinical and demographic characteristics of patients with SjD treated in a rheumatology setting, and to understand the disease features and comorbidities in this population 12 months post-diagnosis.
Methods: Adult patients in the Excellence Network in RheumatoloGY (ENRGY) rheumatology network with ≥2 ICD-9 and ICD-10-CM codes for SjD diagnoses ≥28 to ≤365 days apart from January 2017 through February 2023 were included in this retrospective analysis of electronic health record (EHR) data. Prior to diagnosis by a rheumatologist, patients were required to have ≥12 months without an SjD diagnosis. Patient EHR data were linked to administrative claims from the Komodo Health database. The index date was defined as the date of the first SjD diagnosis. Demographic and clinical characteristics were captured at index. This analysis included a post-index follow-up period of ≥12 months. Further clinical manifestations and patient-reported outcomes (PROs) were reported up to 12 months post-index.
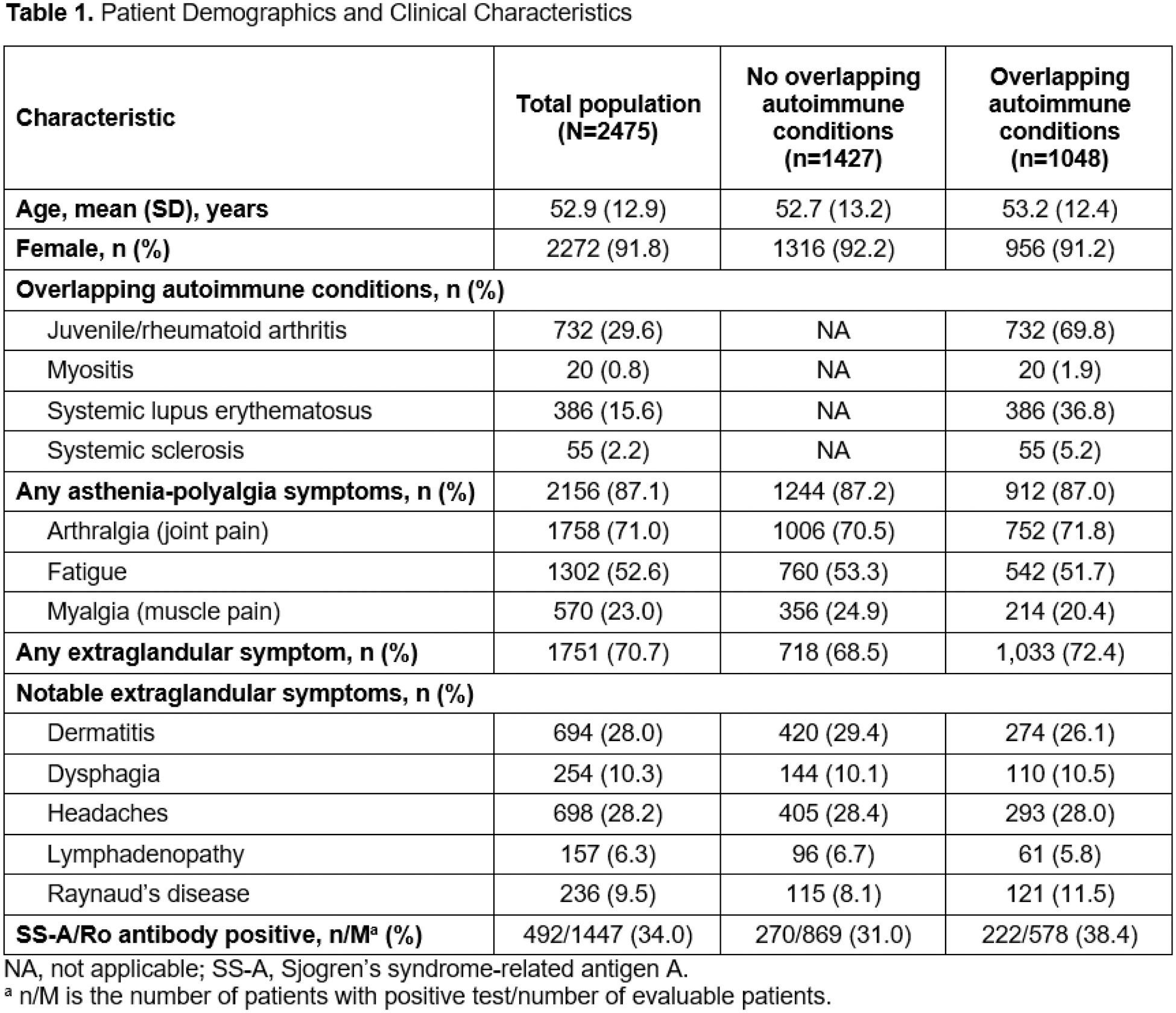
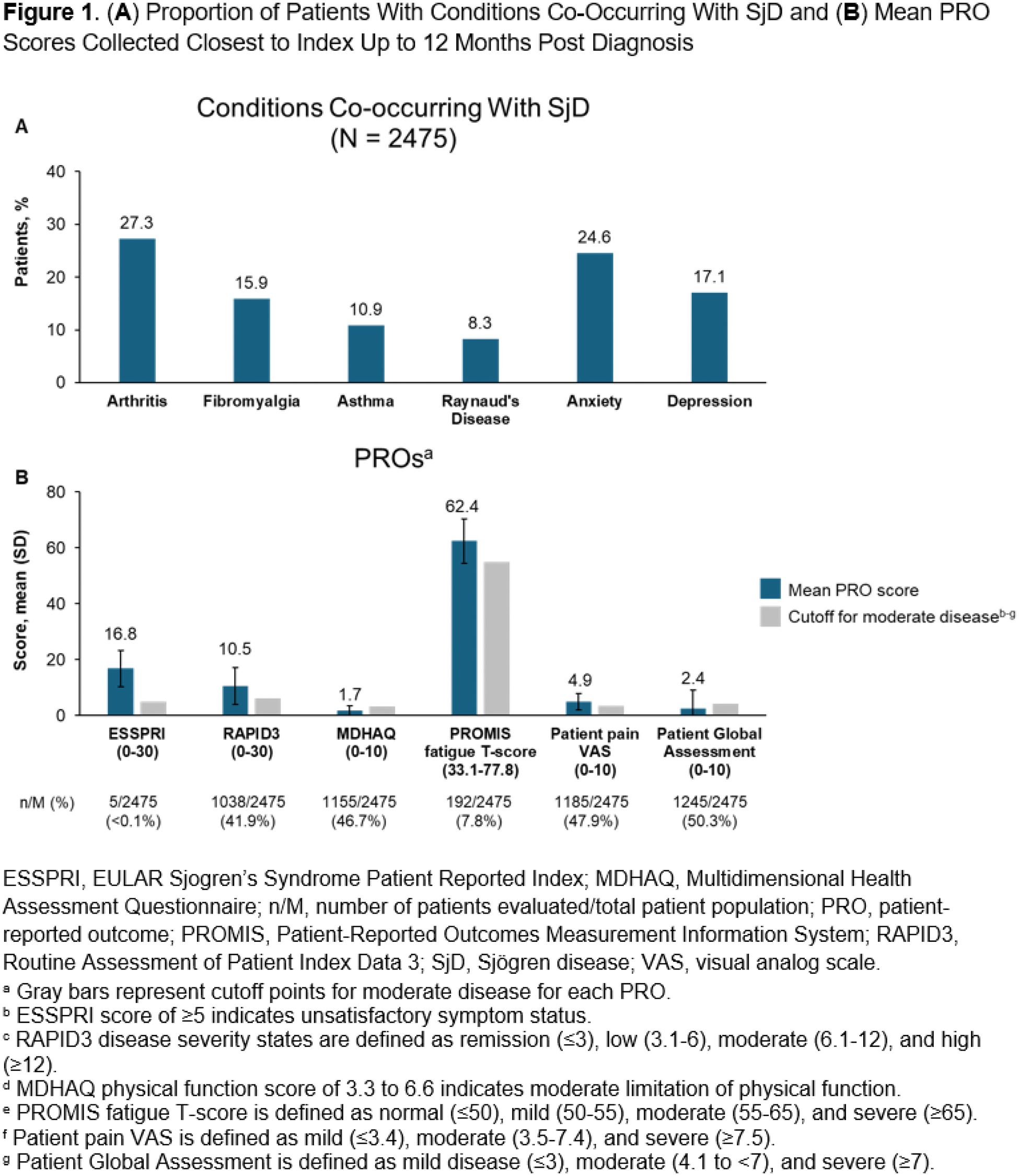
Results: Among 2475 patients, the mean (SD) age was 52.9 (12.9) years and 91.8% were female. Approximately half (55.8%) presented with dryness symptoms prior to diagnosis, which were mainly characterized by dry eyes (22.4%) and dry mouth (10.8 %). Additionally, 87.9% of patients had ≥1 asthenia-polyalgia symptom, which was most often arthralgia (71.0%), fatigue (52.6%), or myalgia (23.0%). A high proportion of patients (70.7%) had ≥1 comorbidity, which included headaches (28.2%), dermatitis (28.0%), dysphagia (10.3%), Raynaud’s disease (9.5%), and lymphadenopathy (6.3%). At index, 58.5% of patients underwent testing for SS-A antibodies, of whom 34.0% had positive results. Use of diagnostic procedures was infrequent; procedures included salivary gland ultrasound (15.4%), magnetic resonance sialography (<0.1%, n=1), an ocular surface staining test (<0.1%; n=1), and an unstimulated whole saliva flow test (<0.1%, n=1). Anxiety (35.9%) and depression (27.1%) were common in patients. A total of 1048 patients (42.3%) had overlapping autoimmune diseases prior to index or up to 1 year post-index, including RA (29.1%), SLE (15.6%), systemic sclerosis (2.2%), and myositis (0.8%). Patient characteristics were similar among those with or without overlapping autoimmune diseases (Table 1). Co-occurring conditions at 12 months post-diagnosis included arthritis (27.3%), anxiety (24.6%), gastroesophageal reflux disease (24.6%), fibromyalgia (15.9%), depression (17.1%), and Raynaud’s disease (8.4%) (Figure 1). Mean (SD) PRO scores collected closest to the index date up to 12 months before or after index included EULAR Sjögren’s Syndrome Patient Reported Index (ESSPRI; 16.8 [6.5]; n=5), Routine Assessment of Patient Index Data 3 (10.5 [6.6]; n=1038), Pain 0-10 (4.5 [2.9]; n=1185), and Patient Global Assessment (2.4 [6.6]; n=1245).
Conclusion: In this retrospective analysis of EHR data with linked claims, patients diagnosed with SjD had a broad spectrum of extraglandular manifestations and comorbidities. The infrequent use of standard laboratory assessments, diagnostic procedures, and the ESSPRI PRO may reflect a lack of disease awareness and an absence of understanding of the superior tools for improved diagnostic coding and optimization of disease management. Furthermore, healthcare professionals may under-document some of the most burdensome SjD-related symptoms, such as dryness, in EMR notes or insurance claims submissions. Arthritis was the most common co-occurring condition at 1 year post-diagnosis. High rates of anxiety and depression were indicative of the substantial humanistic burden faced by patients with SjD. At 12 months post-diagnosis, co-occurring diagnoses and conditions other than dryness were common, and high PRO scores highlighted moderate to severe disease burden in these patients. These real-world data highlight the diverse and complex clinical challenges faced throughout the patient journey by those with SjD who are treated by rheumatologists.
REFERENCES: [1] Baldini C, et al. Nat Rev Rheumatol . 2024;20(8):473-491.
[2] Sebastian A, et al. Reumatologia . 2019;57(5):277-280.
[3] Maleki-Fischbach M, et al. Arthritis Res Ther . 2024;26(1):43.
Acknowledgements: This study was funded by Novartis Pharmaceuticals Corporation. Medical writing support was provided by Coles Keeter, PhD, of Nucleus Global, an Inizio company, and was funded by Novartis Pharmaceuticals Corporation. This abstract was developed in accordance with Good Publication Practice (GPP 2022) guidelines. Authors had full control of the content and made the final decision on all aspects of this publication.
Disclosure of Interests: Jeffrey R Curtis AbbVie, Amgen, Aqtual, Bendcare, BMS, CorEvitas, GSK, Janssen, Lilly, Moderna, Novartis, Pfizer, Sanofi, Scipher, UCB, SetPoint, FASTER, and TNacity Blue Ocean, Emily E Holladay: None declared , Antton Egana Novartis, Novartis, Dhaval Patil Novartis, Novartis, Courtney McDermott Novartis, Novartis, Yujie Su: None declared , Fenglong Xie: None declared , Amy Mudano: None declared , Shanette Daigle: None declared , Sara S. McCoy Novartis, Otsuka, Visterra, Amgen, Kiniksa, iCell, Aurinia, and TearSolutions.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (