

Background: Belimumab (BEL) has shown efficacy in various aspects of systemic lupus erythematosus (SLE), but real-world data on its effects on hematological manifestations are limited. Anemia is common in SLE, with multiple contributing factors such as chronic inflammation, disease activity, and immunosuppressive treatments. Inflammatory cytokines, also play a key role in driving anemia of chronic disease by impairing iron utilization and erythropoiesis. Moreover, nutrient deficiencies, renal insufficiency, and bone marrow suppression further complicate the hematologic picture.
Objectives: This study aims to evaluate the efficacy and safety of BEL in managing SLE-associated anemia, in a national cohort.
Methods: BeRLiSS-newJS (cohort retrospectively analysed) study included patients with joint or skin involvement requiring therapy with BEL, according to physician judgment, in fourteen Italian SLE referral centers. BeRLiSS-newJS inclusion criteria were: 1) Fulfillment of ACR 1997 revised criteria or SLICC 2012 or EULAR/ACR 2019 classification criteria; 2) Treatment with intravenous (10 mg/kg on days 0, 14, and 28, and then every 28 days) or subcutaneous (200 mg/week) BEL for at least 6 months; 3) Active disease, i.e. clinical SLE Disease Activity Index-2000 (cSLEDAI-2K)>0 despite glucocorticoids (GC) and antimalarials (AM), with or without immunosuppressive agents (IS); 4) positive serology (anti-dsDNA antibodies and low C3 or C4 serum levels). For the present study, we considered patients who at baseline also had anemia, defined according to BILAG definition (grade C or higher) as hemoglobin (Hb)≤10.9 g/dl. The study period spans from June 2013 to May 2024. Clinical and laboratory variables were collected at baseline and every six months. They included SLEDAI-2K, fatigue (Visual Analogue Scale 0-10), daily GC intake, complete blood count, anti-dsDNA antibodies, C3, C4, and concomitant medications. Anemia responders were defined as patients achieving Hb≥10.9 g/dl. Comparisons of continuous data with parametric distribution were performed using t-test for paired data, and one-way analysis of covariance (ANCOVA) with Bonferroni’s post hoc analysis. Continuous data with non-parametric distribution were analyzed using Wilcoxon’s test for paired data. Comparisons of categorical data were performed using χ2 test (Pearson test if indicated). p-values less than 0.05 were considered significant. Backward stepwise logistic regression was employed to identify predictors of response at 12 months, with significance set at 5%. Statistical analyses were performed using the SPSS (version 29) software.
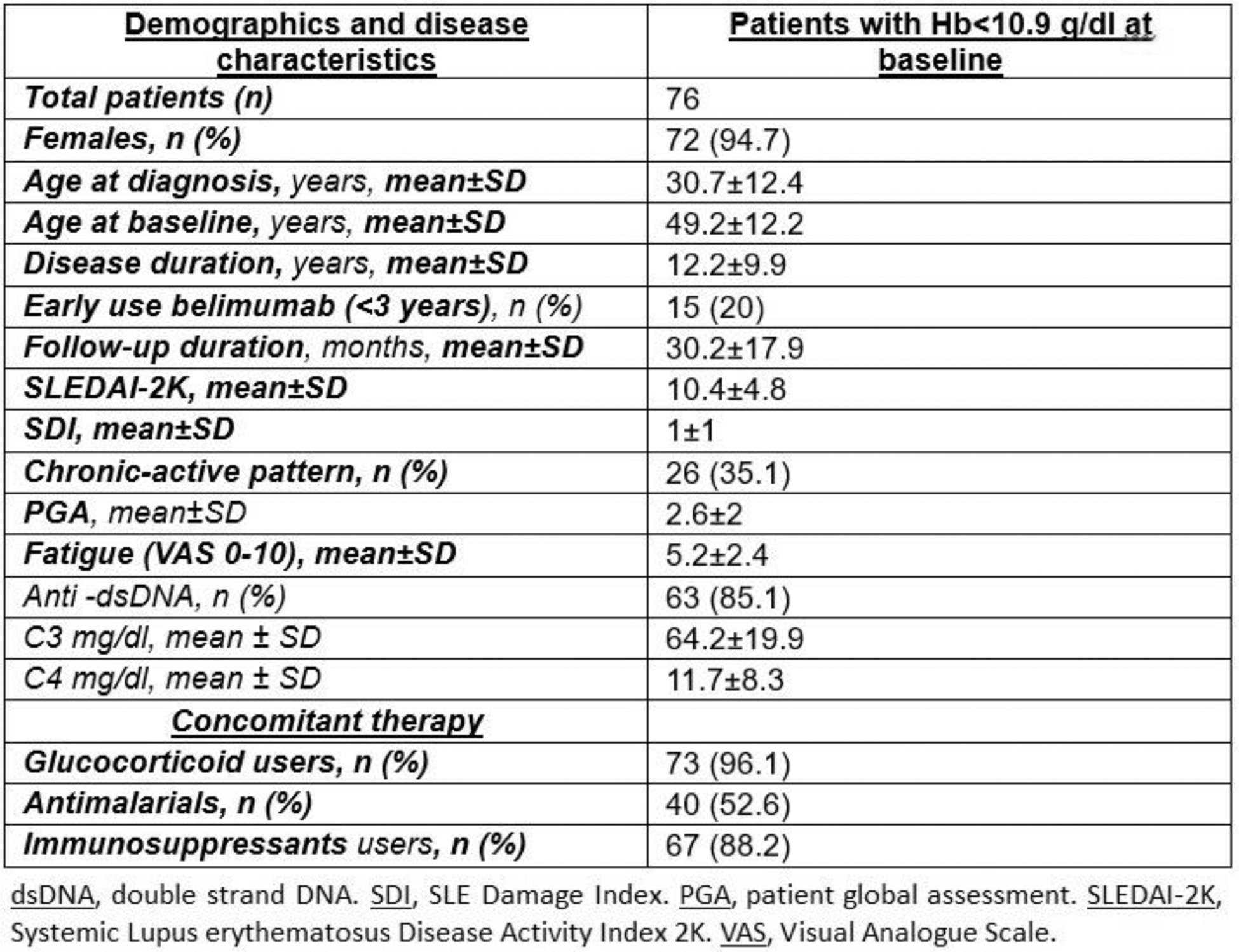
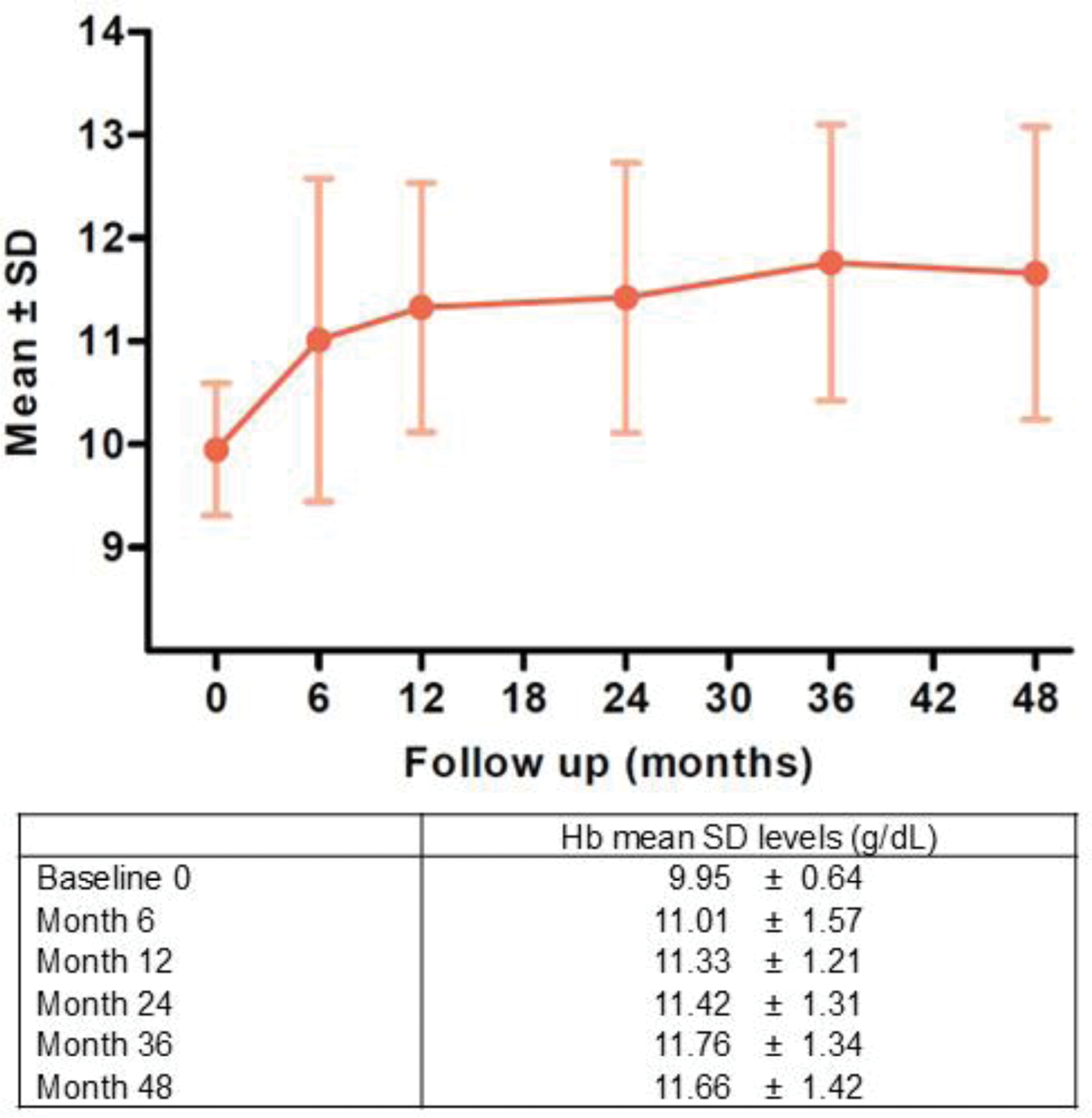
Results: Baseline characteristics of patients included in the study are reported in Figure 1. According to the inclusion criteria, 76 patients with anemia were considered; data up to month 48 of BEL therapy were available in 33 patients. Hb values increased significantly from a mean±SD baseline value of 9.9±0.6 g/dl to 11.7±1.4 g/dl at 48 months, as shown in Figure 2 (month 6 11.01±1.57 g/dl; month 12 11.33±1.21 g/dl; month 24 11.42±1.31 g/dl; month 36 11.76±1.34 g/dl, p<0.001); at 48 months, only 36.4% of patients still had anemia. Notably, we observed a significant reduction in VAS fatigue (baseline 5.2±2.4, month 12 3.0±2.4, month 24 2.5±2.3, month 48 2.6±2.6). Mean GC dose (expressed as PDN equivalents) significantly decreased from baseline (12.9±12.1 mg/d) during the follow-up (month 6 6.6±4.5, month 12 6.5±8.8, month 24 3.5±3.2, month 36 3.1±2.9, month 48 3.6±4.9, p=0.003). GC users decreased from baseline (73, 96.1%) to month 6 (64, 94.1%), month 12 (57, 90.5%), month 24 (32, 72.7%), month 36 (25, 65.8%) and month 48 (17, 70.7%). Hb values did not significantly differ during follow-up between patients on antimalarials compared to those on belimumab alone (p=0.27). At multivariate regression analysis for predictors of response at 12 months, baseline factors significantly associated with anemia resolution were a shorter disease duration (OR 0.93, 95%CI 0.87-0.99, p=0.022), and concomitant use of IS (OR 20.7, CI 1.45–287.26, p=0.024). Overall, 8 patients discontinued BEL for adverse events (AE), none due to severe AE.
Conclusion: Add-on therapy with BEL led to clinical improvement in a significant proportion of patients with anemia in a real-world setting, accompanied by a glucocorticoid-sparing effect. Although limited data exist in the literature on the use of BEL for SLE-related anemia, these findings suggest a potential role in addressing this hematologic manifestation of SLE. The observed improvement in anemia is likely attributable to better disease control, reduction of systemic inflammation, and the decreased need for corticosteroids. Larger, prospective studies are warranted to confirm these observations and further elucidate the mechanisms underlying BEL impact on anemia. The lack of laboratory variables for defining the specific nature of the anemia represents a limitation of this study.
REFERENCES: NIL.
Demographics and disease characteristics at baseline.
Hb levels (g/dL) in patients with anemia at belimumab initiation.
Acknowledgements: NIL.
Disclosure of Interests: Davide Ragno: None declared , Federico Arru: None declared , Alberto Cauli: None declared , Lorenzo Cavagna: None declared , Rossella De Angelis: None declared , Giacomo Emmi: None declared , Roberto Gerli: None declared , Marcello Govoni: None declared , Renato Lo Gullo: None declared , Simone Negrini: None declared , Luca Quartuccio: None declared , Maurizio Rossini: None declared , Carlo Salvarani: None declared , Paola Tomietto: None declared , Angelo Vacca: None declared , Andrea Doria GSK, AstraZeneca, GSK, AstraZeneca, Luca Iaccarino GSK, AstraZeneca, GSK, AstraZeneca, Margherita Zen GSK, AstraZeneca.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (