

Background: Evaluating activity in Systemic Lupus Erythematosus (SLE) is essential to optimize patient management, for which different indices such as SLEDAI-2K and SLE-DAS are used. To define low activity and remission states, we use LLDAS and DORIS, respectively.
Objectives: To assess the utility of activity and remission indices in SLE through their analysis and comparison, and to correlate them with analytical and immunological biomarkers.
Methods: A retrospective, descriptive, cross-sectional study was conducted in a cohort of SLE patients treated between September 2022 and November 2023 in a tertiary hospital. Patients had to meet the 2019 EULAR/ACR Classification Criteria. Demographic, clinical, analytical, and index-related variables were collected. Variable analysis was performed using Kappa and Spearman correlation coefficients and the Mann-Whitney U test.
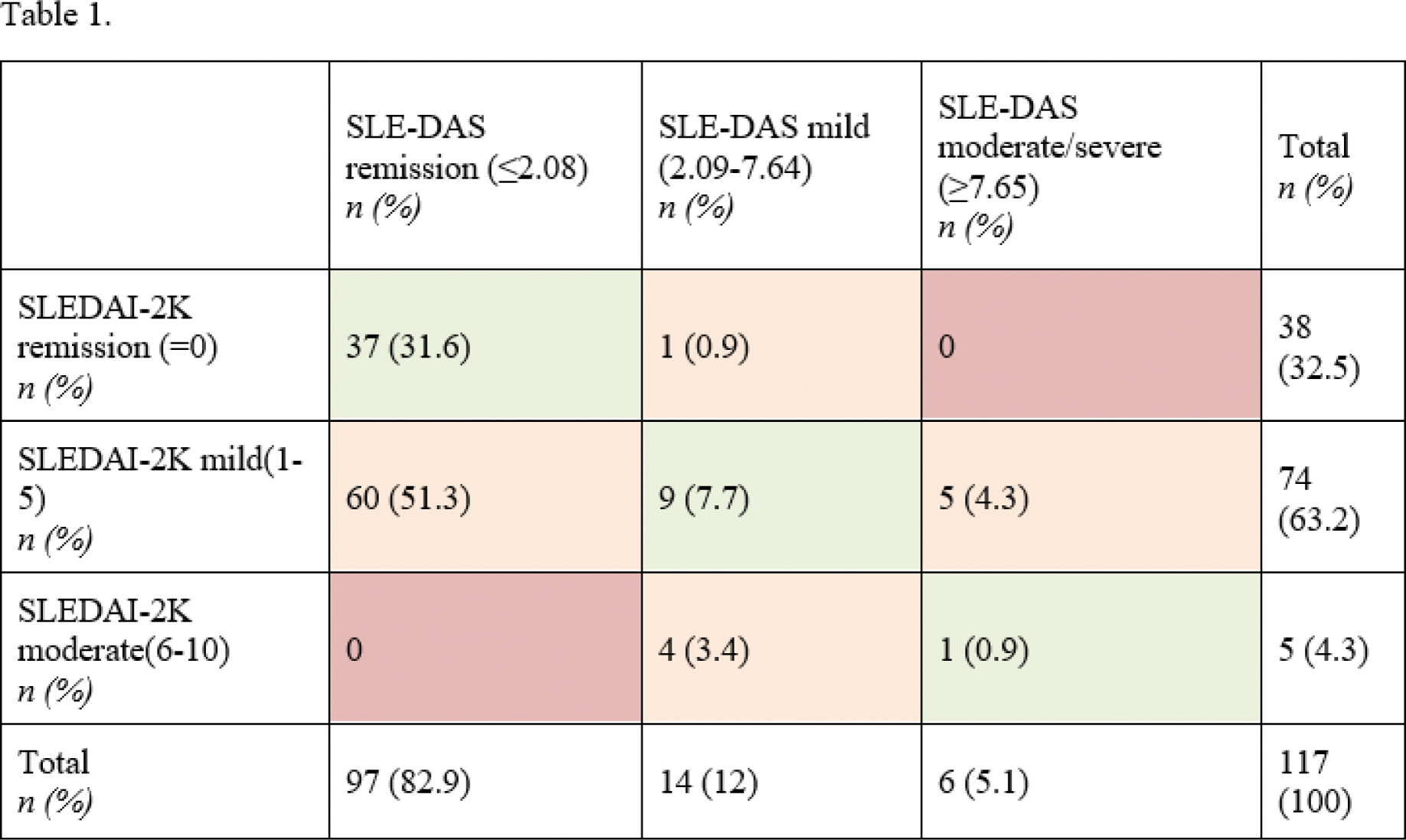
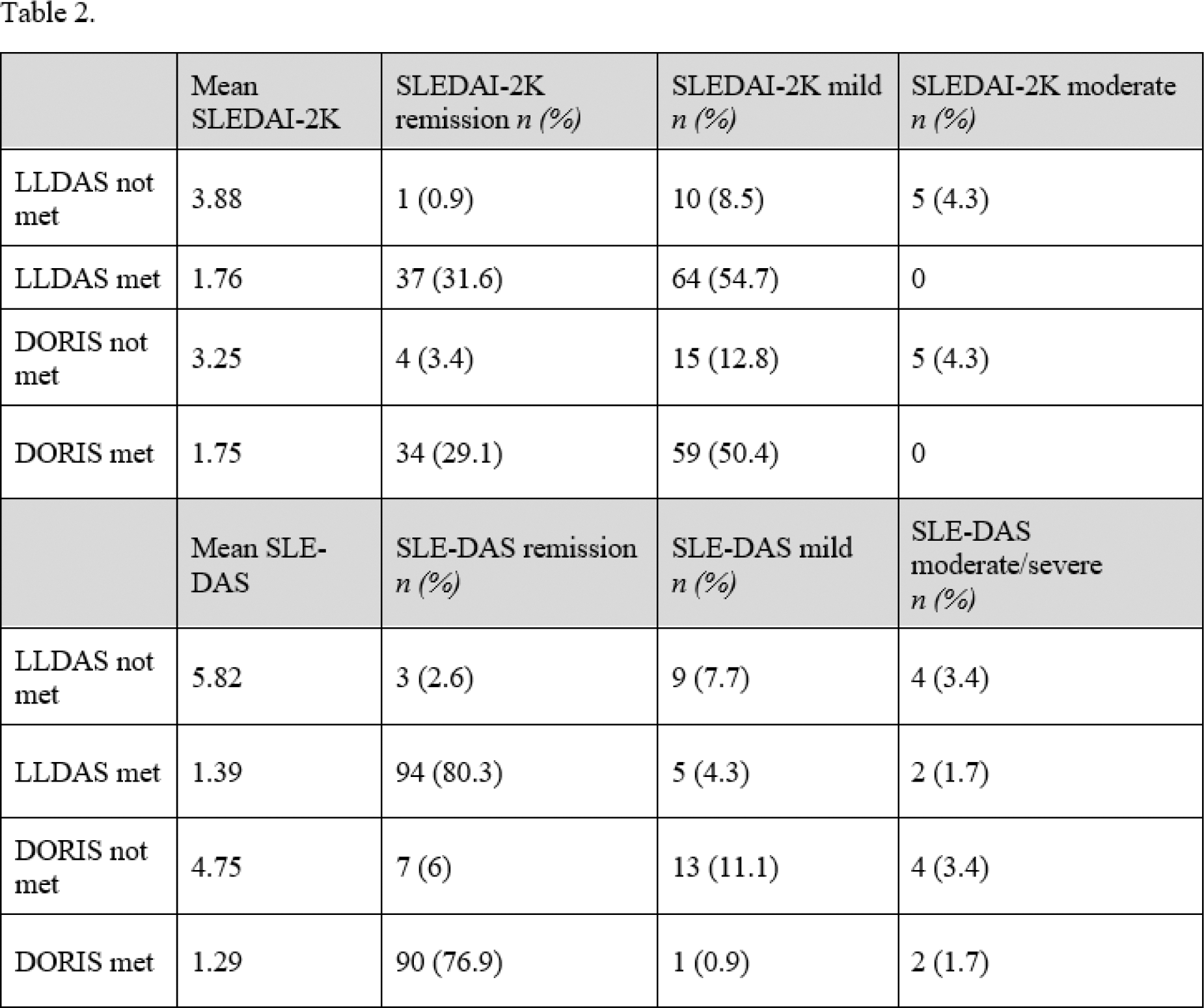
Results: A total of 117 patients were included (94% females, mean age 50.58 years, mean disease duration 15.8 years). The most common manifestation was arthralgia (33.3%), and less frequent were arthritis (5.1%), cutaneous involvement (4.3%) and oral mucosal ulceration (1.7%). No cases of serositis, fever, or myositis were observed. Only 1 patient experienced a renal flare, and 4 patients had significant proteinuria. Thrombocytopenia (<100000) was present in 4 patients (3.4%), and 16.2% of patients had lymphopenia <1000. Only 2 patients had leukopenia (<3000). Serologically, 38.5% had elevated DNA levels, 39.3% low C4 levels and 31.6% low C3 levels. Table 1 presents values and relationships between SLEDAI-2K and SLE-DAS according to activity categories. There weren’t any patients with severe SLEDAI-2K (>10). The correlation between the two indices was strong (Spearman coefficient 0.869, p<0.001 ). However, when variables were evaluated by categories (remission, mild, moderate, severe), this relationship disappeared (Kappa coefficient 0.084, p=0.062). According to SLE-DAS, 82.9% of patients were in remission, compared to 32.5% according to SLEDAI-2K. Of the patients, 86.3% met LLDAS criteria, and 79.5% were in remission per DORIS. Table 2 shows the relationships between activity indices, LLDAS and DORIS. To evaluate the relationship between the different activity and remission indices, the mean differences were calculated. The difference in the LLDAS group vs the group with activity was statistically significant (SS) for both SLEDAI-2K (1.76 vs 3.88; p<0.001 ) and SLE-DAS (1.39 vs 5.82; p<0.001 ). The mean difference in the DORIS remission group vs non-remission group was SS for both SLEDAI-2K (1.75 vs 3.25; p<0.001 ) and SLE-DAS (1.29 vs 4.75; p<0.002 ). The relationship between SLEDAI-2K and clinical-serological manifestations was evaluated using median differences, which were SS for arthritis ( p=0.047 ), cutaneous involvement ( p=0.014 ), hypocomplementemia ( p<0.001 ), and DNA ( p<0.001 ). For SLE-DAS, the median difference was SS for all variables (lymphopenia p=0.015 , leukopenia p=0.019 , thrombocytopenia p<0.001 , proteinuria p<0.001 , arthritis p<0.001 , cutaneous involvement p=0.003 , hypocomplementemia p<0.001 and DNA p<0.001 ).
Conclusion: Both SLEDAI-2K and SLE-DAS are useful for assessing SLE activity, although differences may arise when categorizing them. Both indices show SS associations with LLDAS and DORIS, despite SLE-DAS not being part of their definitions, and the proportion of patients in remission according to SLE-DAS is closer to the proportions of LLDAS and DORIS. The data obtained suggest that SLE-DAS could be a better indicator for evaluating clinical activity in patients with hematological and renal involvement. Our study highlights the potential relevance of lymphopenia as a surrogate marker of SLE activity. The effective disease control reflected in our population according to LLDAS and DORIS suggests that current disease management is effective.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of interests: Julia Bernardez Otsuka, Berta Paula Magallares GSK. Astrazeneca, Helena Codes Abbvie, Laura Martínez-Martínez: None declared, Ignasi Gich: None declared, Ivan Castellví Boehringer-Ingelheim, GSK, Novartis, Boehringher-Ingelheim, GSK, Johnson & Johnson, Boehringer-Ingelheim, Novartis, Sanofi, Johnson & Johnson, Kern, Patricia Moya: None declared, Jose Luis Tandaipan Johnson & Johnson, Hye Sang Park Lilly, Pfizer, Alfa Sigma, Janssen, Novartis, MSD, Boehringer-Ingelheim and Amgen, Cesar Díaz-Torné: None declared, Luís Sainz Comas: None declared, Asier García-Alija: None declared, Ana Laiz Abbvie, Janssen, UCB, Novartis, Susana P. Fernandez-Sanchez GSK, Astrazeneca, Sandra Ros: None declared, Hèctor Corominas: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (