

Background: Biologic disease-modifying anti-rheumatic drugs (bDMARDs) are a common therapy for psoriatic arthritis (PsA). Although a wide range of bDMARDs is available, there is no established method for selecting the bDMARD most suitable for the respective patient. Precision medicine, which tailors treatment to individual patient characteristics, could provide an effective approach.
Objectives: We aim to develop an algorithm based on immunophenotyping that assists in identifying the most suitable bDMARD for each individual patient, thereby enhancing therapy persistence and treatment response in PsA. This approach has been described by Miyagawa, Nakayamada et al. (2019) for a limited number of PsA patients. Here we present the first results of our study, which is part of an ongoing multicentric project conducted at the Centers for Personalized Medicine in Baden-Württemberg, Germany, involving 573 patients so far with inflammatory diseases, including psoriatic arthritis, spondyloarthritis, psoriasis, and inflammatory bowel disease.
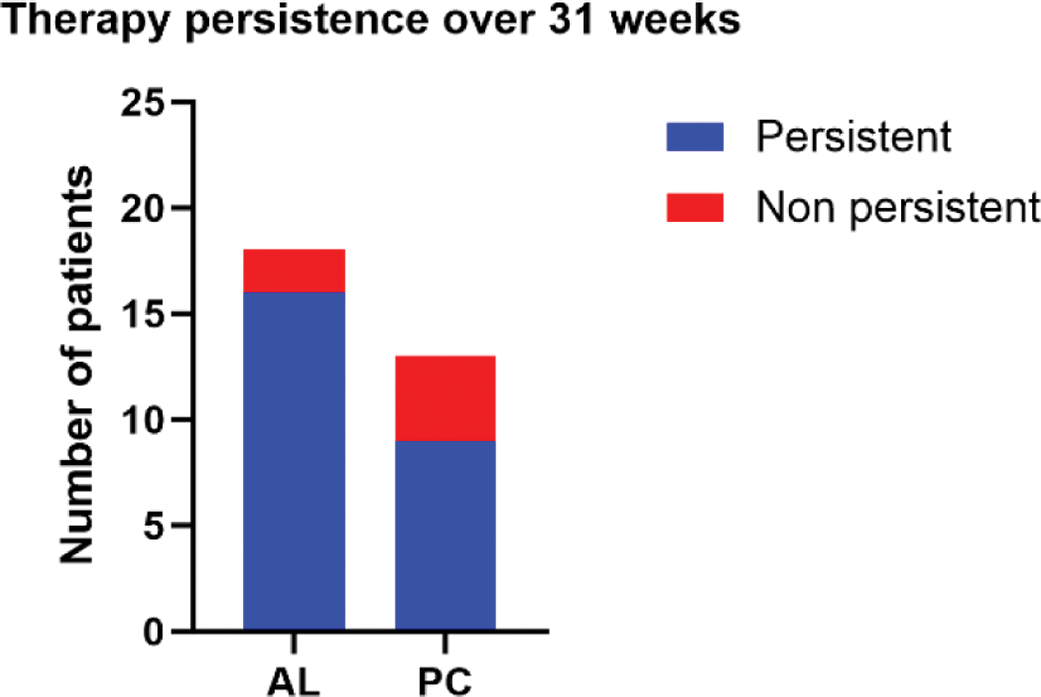
Methods: We performed immunophenotyping on 31 first-line PsA patients. Among them, 18 patients were treated according to an algorithm (AL) based on phenotypic differences in peripheral helper T cells analyzed by 8-color flow cytometry (Miyagawa, Nakayamada et al., 2019), while 13 received treatment according best physician’s choice (PC). Both groups were treated with bDMARDS. We evaluated therapy persistence between the two groups over 31 weeks.
Results: We categorized the 18 patients treated according to the algorithm into four immunophenotypes based on peripheral blood analysis: activated Th1 cell-predominant type, activated Th17 cell-predominant type, Th1/Th17-high type and Th1/Th17-low type (Miyagawa, Nakayamada et al., 2019). Ustekinumab was administered to patients in the activated Th1 cell-predominant group, interleukin-17 inhibitors or interleukin-23 inhibitors to those in the activated Th17 cell-predominant and Th1/Th17-high groups, and TNF inhibitor to patients in the Th1/Th17-low group. Out of 18 patients treated according to the algorithm, 16 (88.9%) remained therapy persistent for at least 31 weeks, while two (11.1%) were not persistent. In the physician’s choice group, nine out of 13 patients (69.2%) remained therapy persistent for 31 weeks, while four (30.8%) were not persistent (n.s. p = 0.2076, Fisher’s exact test).
Therapy persistence over 31 weeks
Source: Own representation.
Conclusion: Our study validated the approach described by Miyagawa et al. under real-world conditions. Although the difference in therapy persistence was statistically not significant, we hypothesize that the algorithm-based treatment according to Th1/Th17-activation represents one category in personalized therapy decisions for PsA patients. We suggest that personalized therapy recommendation needs to be based on a combination of multiple parameters involved in the complex interplay. Hence, optimization of the algorithm and successive integration of additional game-changing diagnostics (e.g., proteomics, metabolomics) could further improve long-term patient outcome prediction and quality of life. Ongoing development of the network of Centers for Personalized Medicine in Baden-Württemberg provides essential expertise in multi-centric translational approaches to validate new biomarkers and diagnostics under real-world conditions and within prospective studies and interdisciplinary, quality-assured infrastructures.
REFERENCES: [1] Miyagawa, I., S. Nakayamada, K. Nakano, S. Kubo, S. Iwata, Y. Miyazaki, M. Yoshikawa, H. Yoshinari and Y. Tanaka (2019). “Precision medicine using different biological DMARDs based on characteristic phenotypes of peripheral T helper cells in psoriatic arthritis.” Rheumatology 58 (2): 336-344.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (