

Background: With the advances in therapeutics for idiopathic inflammatory myopathies (IIM), achieving remission has become an increasingly important goal. However, data on remission, particularly across IIM subtypes, remain limited.
Objectives: This study aimed to assess remission rates and identify predictive factors for remission among IIM patients using a prospective myositis registry at a large academic center.
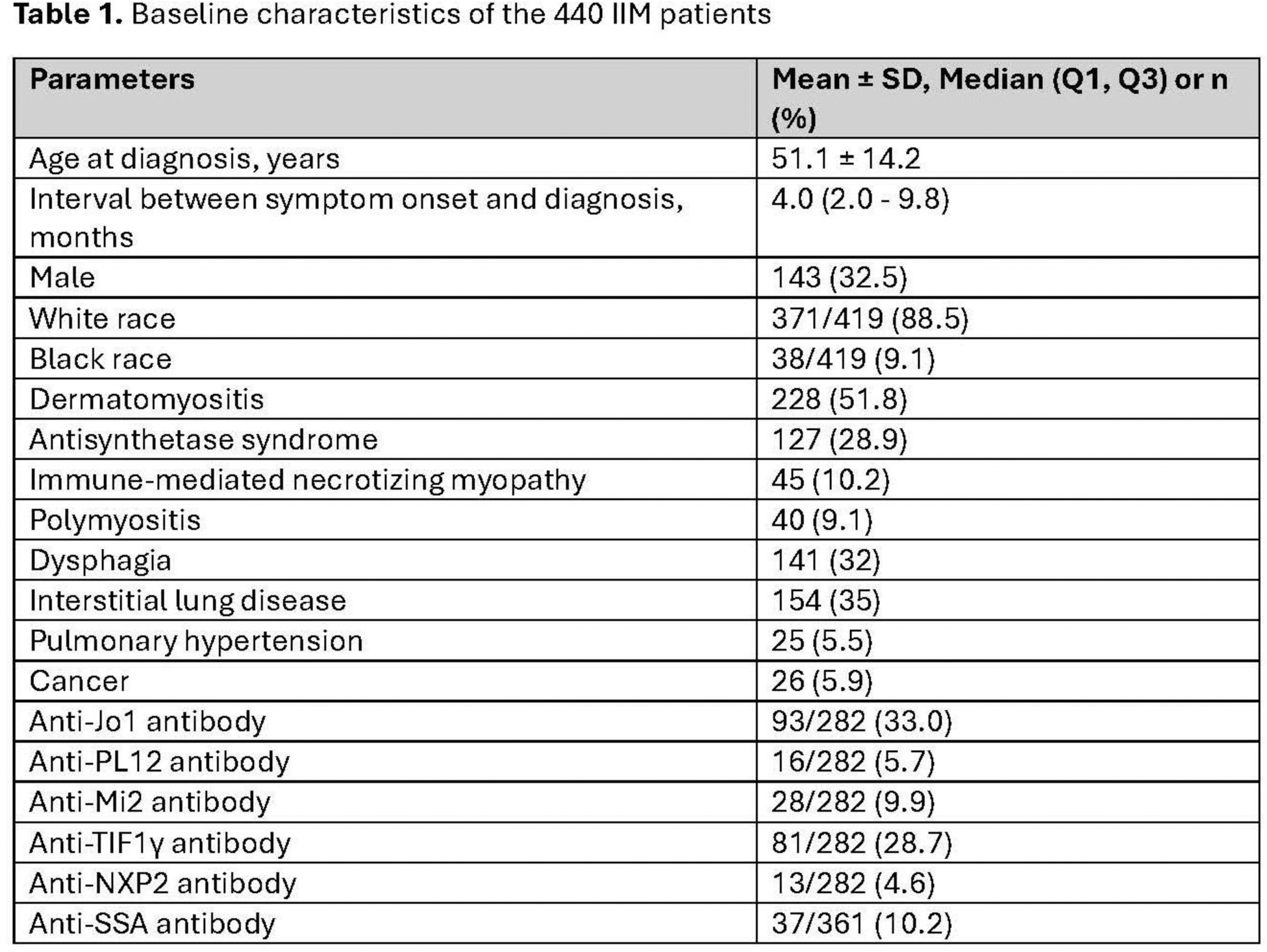
Methods: We enrolled and prospectively followed all IIM patients evaluated at the University of Pittsburgh Myositis Center between December 1981 and December 2024, utilizing a standardized database in the myositis registry. The remission study cohort included 440 adult IIM patients with disease duration > 1 year. We focused on four IIM subtypes according to the physician’s diagnosis: dermatomyositis (DM), antisynthetase syndrome (ASyS), immune-mediated necrotizing myopathy (IMNM), and polymyositis (PM). The diagnosis was based on the 2017 European League Against Rheumatism/American College of Rheumatology (EULAR/ACR) classification criteria for IIMs and Sontheimer criteria. ASyS was further defined by clinical symptoms and the presence of ASyS antibodies, while IMNM was further defined by clinical symptoms and either anti-HMGCR antibodies, anti-SRP antibodies, or muscle biopsy findings consistent with IMNM. Patients with overlap syndromes or inclusion body myositis were excluded. Remission at the last visit was defined as the absence of disease activity based on expert clinical assessment, either while receiving stable/de-escalating IIM therapy or off medication. Predictive factors for remission were evaluated using multivariate logistic regression analysis in a subpopulation of 353 patients with a minimum follow-up of 1 year post-enrollment in the myositis registry.
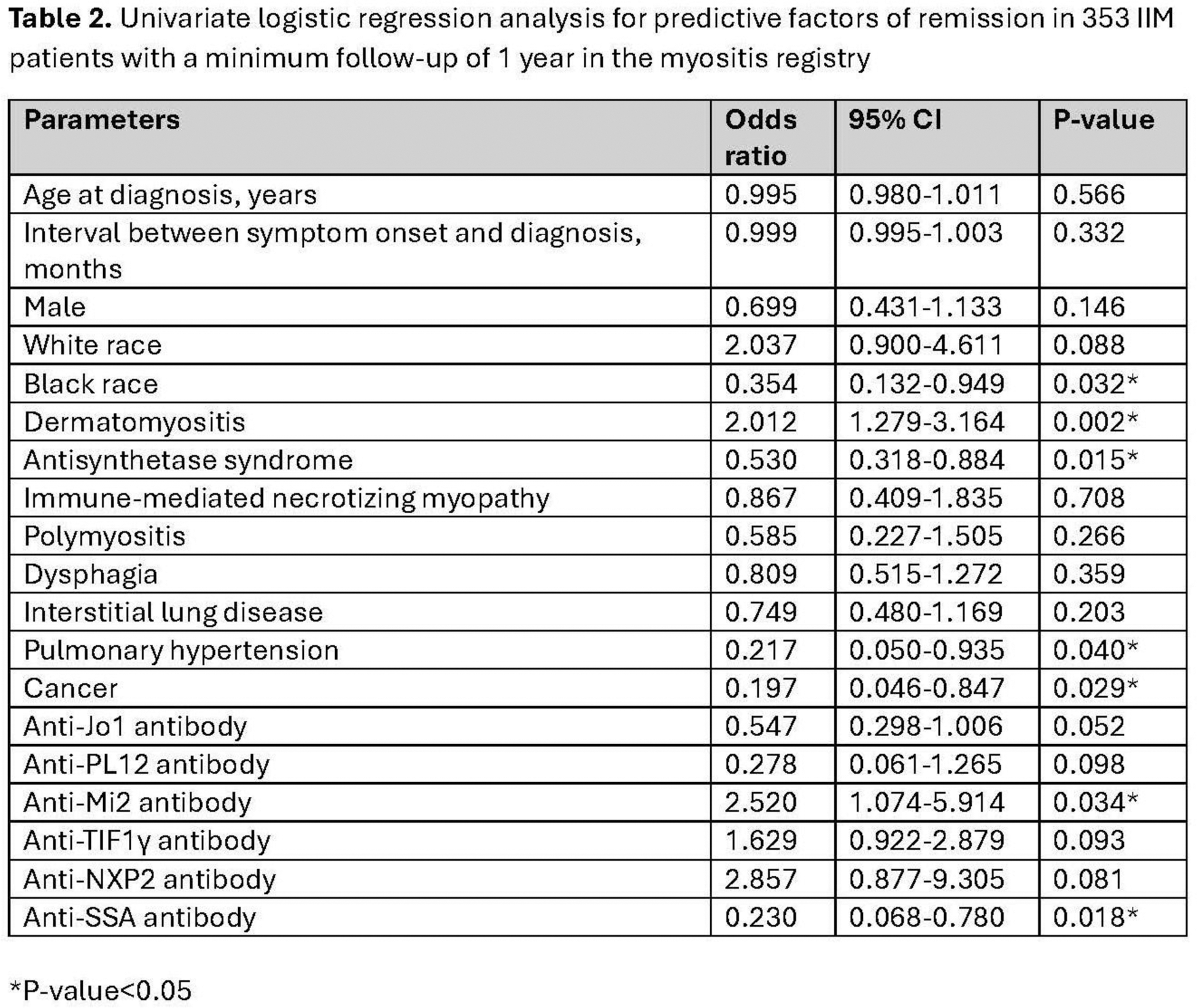
Results: Among 440 IIM patients, the majority were female (67.5%) and white (88.5%), with a mean age at diagnosis of 51.1 ± 14.2 years. The median disease duration was 7.6 years, and the overall remission rate was 28.4%. Remission rates by subtype were 35.5% (81/228) for DM, 20.5% (26/127) for ASyS, 26.7% (12/45) for IMNM, and 15.0% (6/40) for PM. The median follow-up time for all patients was 5.3 years. Univariate analysis identified DM subtype (OR 2.012, 95% CI 1.279–3.164, P=0.002) and anti-Mi2 antibodies (OR 2.520, 95% CI 1.074–5.914, P=0.034) as positive predictors, while black race (OR 0.354, 95% CI 0.132–0.949, P=0.032), ASyS subtype (OR 0.530, 95% CI 0.318–0.884, P=0.015), pulmonary hypertension (OR 0.217, 95% CI 0.050–0.935, P=0.040), cancer (OR 0.197, 95% CI 0.046–0.847, P=0.029), and anti-SSA antibodies (OR 0.230, 95% CI 0.068–0.780, P=0.018) were negative predictors of remission. In multivariate analysis, ASyS subtype (OR 0.497, 95%CI 0.265-0.934, P=0.040) and anti-SSA antibodies (OR 0.277, 95%CI 0.080-0.953, P=0.042) were the only independent factors associated with a lower likelihood of remission.
Conclusion: Remission rates vary across IIM subtypes, with DM patients achieving the highest remission rates and PM the lowest. ASyS subtype and anti-SSA were associated with a reduced likelihood of remission. These findings highlight the importance of tailored management strategies based on IIM subtypes and serological profiles.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of interests: Nantakarn Pongtarakulpanit: None declared, Eugenia Gkiaouraki: None declared, Shiri Keret: None declared, Vaidehi Kothari: None declared, Siamak Moghadam-Kia: None declared, Dana Ascherman: None declared, Rohit Aggarwal Octapharma, CSL Behring, Bristol-Myers Squibb, EMD Serono, Kezar, Pfizer Inc, AstraZeneca, Alexion, Argenx, Boehringer Ingelheim, Corbus, Janssen, Kyverna, Roivant, Merck, Galapagos, ActiGraph, Horizon Therapeutics, Teva, ANI Pharmaceutical, Nuvig, Capella, Cabaletta Bio, I-Cell, Sanofi, and Boehringer Ingelheim, Mallinckrodt, Pfizer Inc., Bristol Myers-Squibb, Q32, EMD Serono, Janssen, and Boehringer Ingelheim.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (