

Background: There is an unmet need to accurately assess disease activity and establish treatment goals for patients with systemic lupus erythematosus (SLE). Recently, the SLE Disease Activity Score (SLE-DAS) has been introduced and validated in Italian, Mexican and Indian populations [1]. Although SLE-DAS showed higher sensitivity than SLEDAI 2000 (SLEDAI-2K), little is known about the performance of this novel instrument in Chinese patients with SLE, especially those with high disease activity.
Objectives: We aimed to evaluate the validity of the SLE-DAS in Chinese population with SLE and determine the most appropriate tool for evaluating clinical changes.
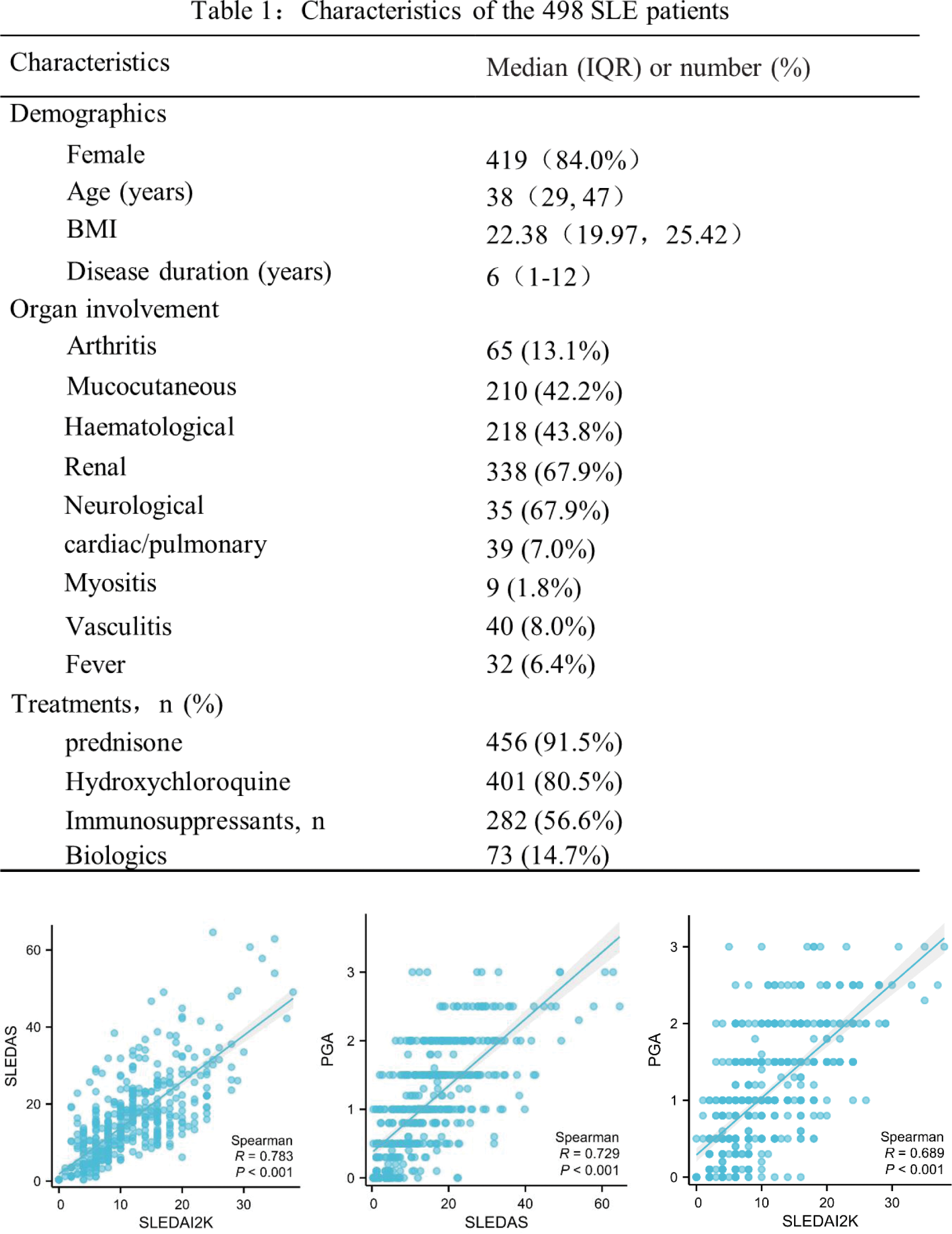
Methods: We retrospectively studied 498 patients with SLE (fulfilling the classification criteria of ACR 1997 and/or SLICC 2012 and/or EULAR/ACR 2019) from Peking University First Hospital. Disease activity was quantified using Physician Global Assessment (PGA), SLEDAI-2K, and SLE-DAS. Demographic, clinical and laboratory parameters were also collected. To assess the performance of various instruments, we compared the correlations and agreements between SLE-DAS, PGA and SLEDAI-2K. We also we evaluated whether SLE-DAS was associated with serum complement levels.
Results: There were 68.9% patients with moderate to severe activity in our study. SLE-DAS score had a moderate positive correlation with PGA (r=0.729, p<0.001), and with SLEDAI-2K (r=0.783, p<0.001). SLE-DAS also showed a weak but evident association with C3 complement levels and with C4 complement levels. The total agreement between SLE-DAS and SLEDAI-2K was 84.3% (κ = 0.667).
Conclusion: SLE-DAS was a novel and useful tool to measure activity in SLE. However, the performance of SLE-DAS in our population was not robust, partly due to the high disease activity of enrolled patients. We need to evaluate the validity of the SLE-DAS separately in patients with different disease activity.
REFERENCES: [1] Jesus D, Matos A, Henriques C,et al. Derivation and validation of the SLE Disease Activity Score (SLE-DAS): a new SLE continuous measure with high sensitivity for changes in disease activity[J]. Ann Rheum Dis. 2019 Mar;78(3):365-371.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (