

Background: Systemic Lupus Erythematosus (SLE) is a chronic autoimmune disease associated with substantial morbidity and mortality. Patient-reported outcome (PRO) measures are particularly valuable in SLE as they assess health-related quality of life (HRQoL) and capture disease aspects that may not be fully reflected by conventional disease activity measures, such as features of Type 2 SLE.
Objectives: This study aimed to examine the association between clinically meaningful improvements in HRQoL scores, assessed using the Short Form 36 (SF-36), and disease activity, measured by SLEDAI-2K, in patients with SLE in a real-world clinical setting.
Methods: A retrospective analysis was conducted using prospectively collected data from SLE patients followed at a single center in Toronto, Canada. Clinical and laboratory data were collected every 3 to 6 months, while SF-36 was administered annually. Patients with active disease (SLEDAI-2K ≥ 6) from 2005 (marking the advent of mycophenolate mofetil use) to 2024, with baseline and one-year follow-up SF-36 data, were included. Minimum clinically important differences (MCID) in SF-36 scores were defined as increases in SF-36 Physical (PCS) and Mental Component Summary (MCS) scores by ≥2.5, individual domain scores by ≥5, and minimum important difference (MID) for SLEDAI-2K as a decrease by ≥4. Associations between improvements in SF-36 (two summary and eight domain scores) and SLEDAI-2K responses at one year were analyzed using chi-square tests. To study the absolute change in SLEDAI-2K, two regression models examined least square mean differences (LSM) in SLEDAI-2K scores from baseline to one year for PCS and MCS score responders versus non-responders at one year. Impact of PCS and MCS score response vs. non-response on SLEDAI-2K over follow-up (one, two, three and five years) was studied using two separate Linear Mixed Models (LMM).
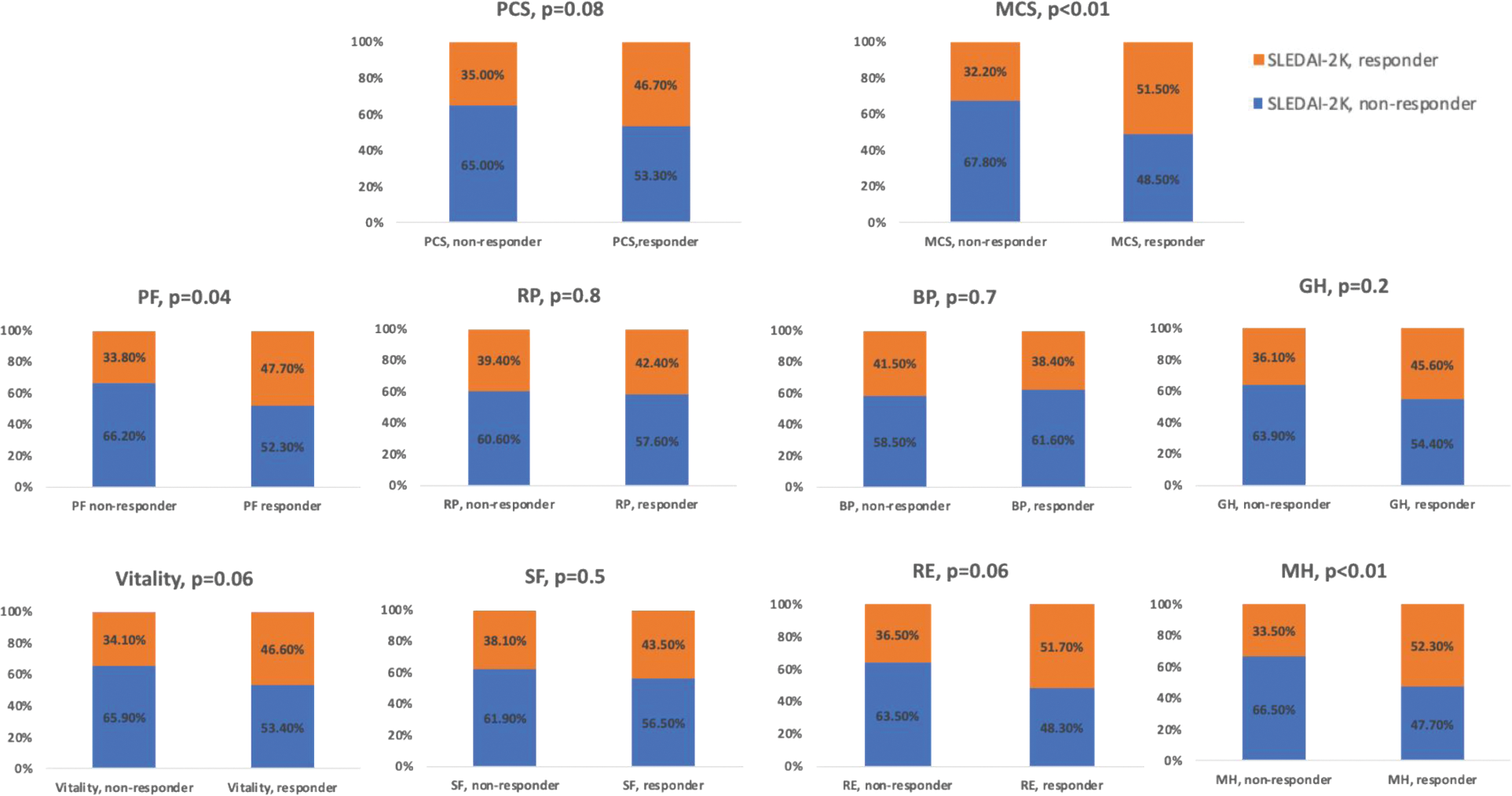
Results: A total of 247 patients were included with a median age of 37.1 years (IQR 28.5–46.5) at the study visit, a female-to-male ratio of 8.8:1, and a median SLE duration from diagnosis of 9.25 years (IQR 4.39–16.07). The median SLEDAI-2K score was 8 (IQR 6–12), with common organ involvements being mucocutaneous (46.6%), renal (44.9%), and musculoskeletal (23.5%). Most patients had active serology (79.4%) and a median SDI of 1 (IQR 0–2) at the study visit. Most patients received hydroxychloroquine (83%), with mycophenolate mofetil (49.4%) being the most commonly prescribed immunosuppressant, followed by azathioprine (44.9%). Among MCS score responders, a significantly greater proportion also achieved SLEDAI-2K responses compared to MCS non-responders (52 of 101, 51.5% vs 47 of 146, 32.2%, p<0.01). This association was not observed between PCS score responders and non-responders. For individual SF-36 domains, significantly more patients who reported clinically meaningful improvements in physical function (53 of 111, 47.7 vs. 46 of 136, 33.8%, p=0.04) and mental health (45 of 86, 52.3 vs. 69 of 189, 33.5, p<0.01) domains also achieved SLEDAI-2K responses. No significant differences were reported for other domains, although there was a trend in vitality and role emotional responders to achieve SLEDAI-2K responses (Figure 1). When absolute change in SLEDAI-2K was studied, a significant difference in LSM change in SLEDAI-2K scores from baseline to one year was observed between PCS responders vs. non-responders (-5.13 and -3.09, p<0.01) as well as MCS responders vs. non-responders (-5.19 and -3.14, p<0.01). When the impact of PCS and MCS score response on SLEDAI-2K over 5 years was studied, PCS score responders had a lower SLEDAI-2K at years two [b -0.94, 95% confidence intervals (-1.45,-0.38)], three [-1.66 (-2.22,-1.10)], and five [-1.62 (-2.23,-1.00)] but not at year one as compared to PCS score non responders. In another LMM model for impact of MCS score response on SLEDAI-2K, MCS score responders had a lower SLEDAI-2K at years one [-1.1 (-1.98,-0.21)], two [-0.93 (-1.48,-0.37)], three [-1.65 (-2.22,-1.08)] and five [-1.65 (-2.21,-1.08)] compared to MCS score non responders.
Conclusion: In patients with active SLE, clinically important improvements in disease activity were particularly notable among those who reported clinically meaningful improvements in MCS scores, physical function, and mental health domains of the SF-36 at one year. Patients demonstrating meaningful improvements in both MCS and PCS scores also experienced greater reductions in SLEDAI-2K scores at one year. PCS and MCS score responders consistently showed lower SLEDAI-2K values over five years as compared to non-responders. These findings indicate that improvements in some aspects of HRQoL are associated with significant reductions in disease activity in SLE patients.
Proportion of SLEDAI-2K responders and non-responders in patients reporting improvements ≥ MCID versus non-responders in SF-36 summary and domain scores.
PCS- Physical Component Summary Score, MCS- Mental Component Summary Score, GH- General Health, MH-Mental Health, PF- Physical Function, BP- Bodily Pain, RP- Role Physical, SF- Social Function, RE- Role Emotional.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Pankti Mehta: None declared, Patricia Katz: None declared, Vibeke Strand Abbvie, Alumis, Amgen Corporation, Arthrosi, Artiva, AstraZeneca, Bayer, Blackrock, BMS, Boehringer Ingelheim, Catalys, Citryll, Contura, Cullinan, Fate Therapeutics, Fortress Biotech, Gate Biosciences, Genasence, Genentech/ Roche, GSK, Inmedix, Kiniksa, Lipum, Longitude Capital, MED Institute, Novartis, R-Pharm, RAPT, Royalty Pharma, Sanofi, Scipher, Setpoint, Sobi, Spherix, Synact, Takeda, Topography, Zoe, Fadi Kharouf: None declared, Laura Whitall Garcia: None declared, Qixuan Li: None declared, Anca Askanase: None declared, Christopher D Saffore AbbVie, full-time employee of AbbVie, Denise Kruzikas AbbVie, full-time employee of AbbVie, Dafna D. Gladman AbbVie, AstraZeneca, Amgen, Eli Lilly, Janssen, GSK, Novartis, Pfizer, UCB, AbbVie, Amgen, Eli Lilly, Janssen, Novartis, Pfizer, UCB, Zahi Touma: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (