

Background: Lupus nephritis (LN) is a common complication of systemic lupus erythematosus (SLE), with kidney failure and cardiovascular events being the primary causes of death in SLE patients. Nearly all SLE patients show renal pathological abnormalities upon electron microscopy and immunofluorescence examination [1]. LN patients often slowly progress to chronic kidney disease (CKD), with significant risk factor for cardiovascular events. CKD in SLE patients is typically insidious, with persistent proteinuria and gradual deterioration of renal function. Without intervention, some patients eventually require dialysis, while many die from cardiovascular complications [2]. Thus, controlling proteinuria and protecting renal and cardiovascular health is crucial for LN patients. Albuminuria is an independent risk factor for worsened renal function and elevated cardiovascular risk in CKD associated with SLE [3]. Despite the use of immunosuppressants combined with RAASi and/or sodium-glucose cotransporter 2 inhibitors (SGLT2i), many patients do not achieve optimal outcomes. These patients, with urinary protein >0.5 g/d after 6–12 months of treatment, remain at high cardiorenal risk, highlighting the need for more effective management. Finerenone, a novel nonsteroidal mineralocorticoid receptor antagonist (MRA), has been shown in global RCTs to have significant renal and cardiovascular protective effects, particularly in diabetic patients with CKD, where it reduces the urinary albumin/creatinine ratio (UACR) [4]. The FINEARTS-HF study further demonstrated Finerenone’s ability to reduce heart failure events and cardiovascular deaths in HFmrEF/HFpEF patients [5]. Overactivation of the mineralocorticoid receptor (MR) contributes to proteinuria, CKD progression, and cardiovascular risk, making it a crucial therapeutic target. Finerenone’s nonsteroidal structure provides greater selectivity and reduced side effects, and its distributed equally in kidney and heart tissues, simultaneously blocking inflammation and fibrosis in both organs [6].
Objectives: Given the complex interplay between SLE, CKD, and cardiovascular disease, we hypothesize that Finerenone reduces proteinuria and improves renal and cardiac outcomes in LN patients by blocking MR-mediated biological effects, including inflammation and fibrosis. This study aimed to evaluate the impact of Finerenone on residual proteinuria in LN patients receiving stable immunosuppressants and maximized doses of RAASi and/or SGLT2i.
Methods: Ten patients with histologically confirmed LN, stable on glucocorticoids, immunosuppressants, and RAASi for at least six months, were enrolled (Table 1).
Results: Four patients had steroid-induced diabetes and were on SGLT2i. The median proteinuria was 1.18 (0.31–5.14) g/day, and the median UACR was 1388.75 (477.77–7941.56) mg/g. Finerenone 20 mg/day was added to the treatment regimen. Within 12 weeks, proteinuria decreased to 0.46 (0.19–3.34) g/day (a 61.4% reduction), and UACR decreased to 531.19 (192.44–3727.08) mg/g (a 61.8% reduction). Serum albumin levels increased slightly, and eGFR remained stable. Meanwhile, no adverse events occurred.
Conclusion: This pilot trial demonstrates that Finerenone effectively reduces residual proteinuria in LN patients and offers additional renal protection when combined with standard therapies. Despite the small sample size, our findings suggest that Finerenone can be safely and effectively used in LN patients. We believe that the combination of Finerenone, RAAS inhibitors, and SGLT2 inhibitors may provide additive renal benefits in LN due to their complementary mechanisms, potentially enabling comprehensive ‘lupus-cardio-kidney’ co-management. However, larger RCTs are needed to confirm these results and evaluate the cardio-renal benefits of Finerenone in LN patients.
Table 1. Clinical characteristics of the patients.
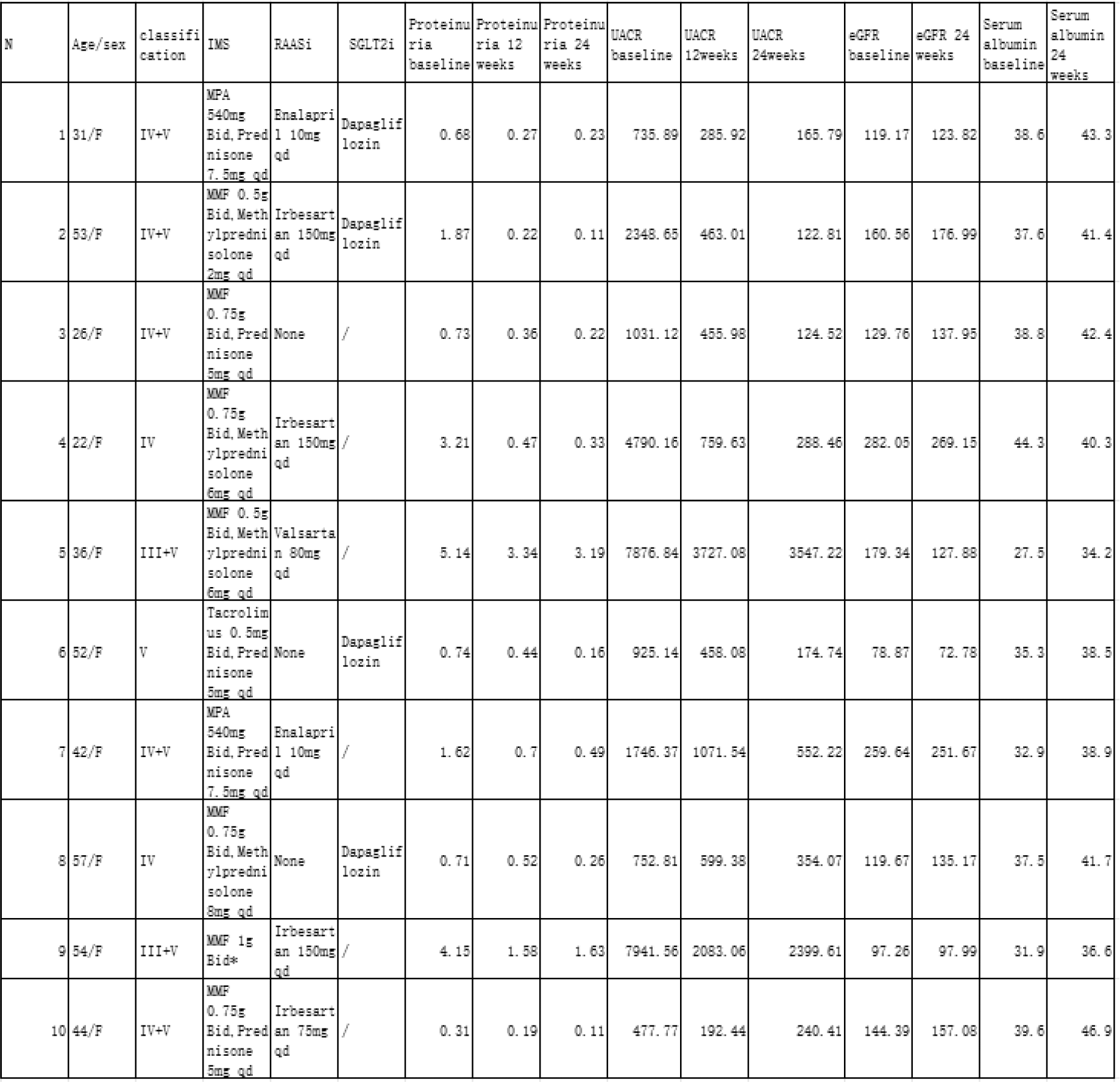
MPA: Mycophenolic acid, MMF: Mycophenolate mofetil.
*: The patient refused glucocorticoid treatment due to the development of osteonecrosis of the femoral head.
REFERENCES: [1] Siegel, C.H. and L.R. Sammaritano, Systemic Lupus Erythematosus: A Review. JAMA, 2024. 331(17): p. 1480-1491.
[2] Bello, N., et al., Cardiovascular events and risk in patients with systemic lupus erythematosus: Systematic literature review and meta-analysis. Lupus, 2023. 32(3): p. 325-341.
[3] Lichtnekert, J. and H.J. Anders, Lupus nephritis-related chronic kidney disease. Nat Rev Rheumatol, 2024.
[4] Pitt, B., et al., Cardiovascular Events with Finerenone in Kidney Disease and Type 2 Diabetes. N Engl J Med, 2021. 385(24): p. 2252-2263.
[5] Solomon, S.D., et al., Finerenone in Heart Failure with Mildly Reduced or Preserved Ejection Fraction. N Engl J Med, 2024.
[6] Nishiyama, A., Pathophysiological mechanisms of mineralocorticoid receptor-dependent cardiovascular and chronic kidney disease. Hypertens Res, 2019. 42(3): p. 293-300.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (