

Background: Glucocorticoids play an indispensable role in the management of SLE. Current treatment guidelines favour use of methylprednisolone in SLE with moderate-severe disease activity. However, Dexamethasone may have a similar therapeutic effect and can prove to be more cost effective. Head-to-head comparative studies between methylprednisolone and dexamethasone in SLE are scarce, particularly in real-world clinical practice settings.
Objectives: Primary-To compare the efficacy of Dexamethasone and Methylprednisolone based on the comparison of SLEDAI2K and SLEDAI-2KG scores between two groups at 60 days following the intervention. Secondary -1. To compare the achievement of SRI-50 improvement definition in the relevant parameters of SLEDAI-2K score. 2. To compare clinical SLEDAI at day 15 and 30 between the two groups.
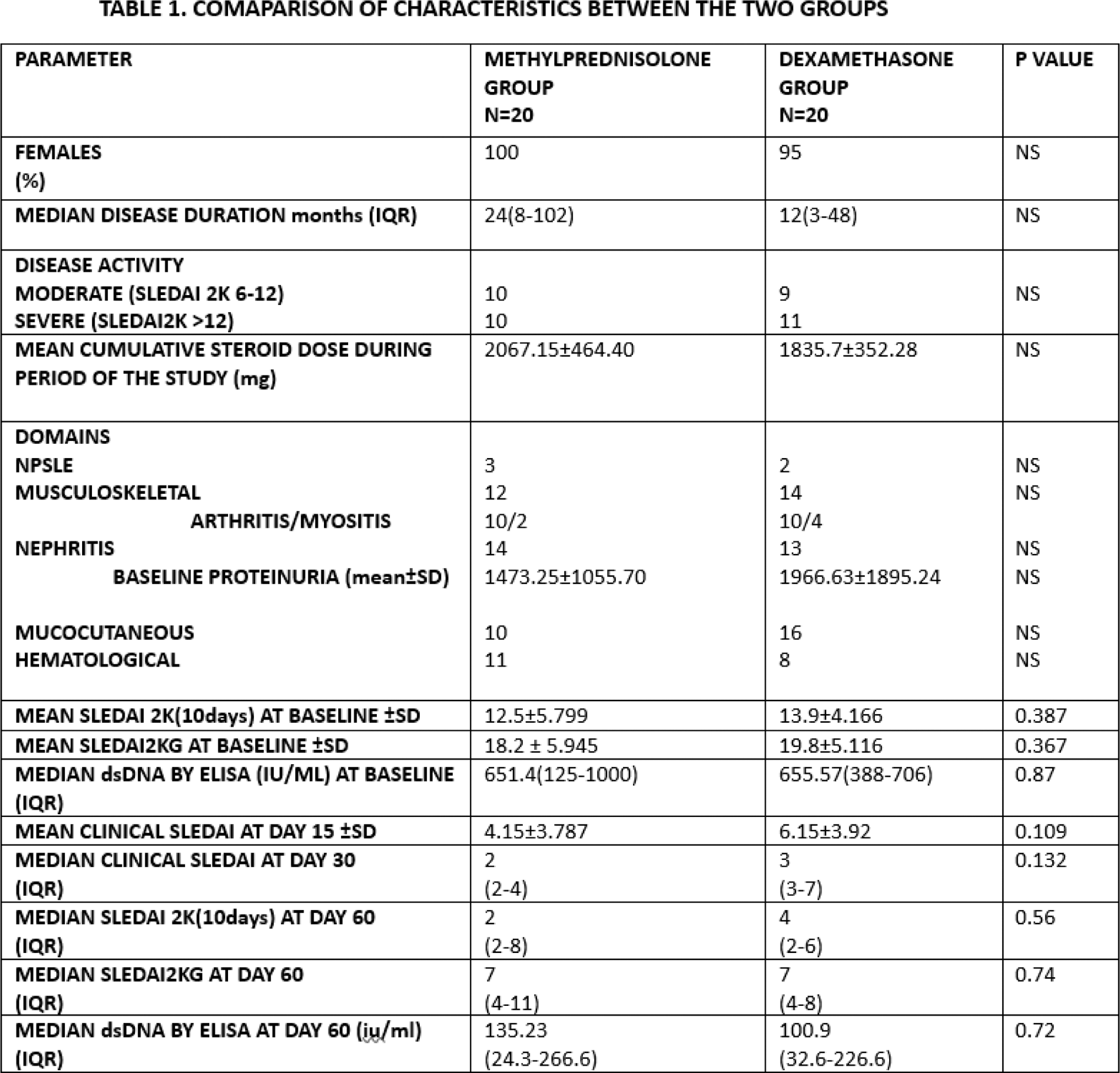
Methods: This randomized, open-label, single-center trial included 40 hospitalized adult patients with moderate to severe SLE (SLEDAI-2K ≥6). All hospitalized patients of SLE who have previously met or currently meet the 2012 SLICC and/or 2019 ACR criteria for SLE were included in the trial after obtaining written informed consent. The patients were randomized based on disease activity (i.e. SLEDAI2K score 6-12 and SLEDAI 2K>12) to receive either IV Methylprednisolone 250 mg for 3 days or IV Dexamethasone 8 mg BD for 3 consecutive days. The randomization list was generated using a computer software employing random number algorithms and stratified randomization was done. Both groups subsequently received standard of care as per physician discretion in accordance with the current international guidelines. Following the intervention, clinical SLEDAI was assessed at day 15 and day 30. SLEDAI2K (10days) and SLEDAI2KG were assessed at baseline and day 60. SLEDAI-2KG is a validated modification of the SLEDAI2K that describes disease activity while accounting for Glucocorticoid dose. SRI-50 response for each clinical domain was calculated at day 15, day 30 and day 60 for the clinical parameters and was calculated only at day 60 for the laboratory parameters. SRI 50 response is a set of validated definitions of 50% improvement in each of the SLEDAI-2K descriptors. Statistical analysis was performed using chi-square tests for categorical data and t-tests or Mann-Whitney U tests for continuous data, with a significance threshold of p < 0.05.
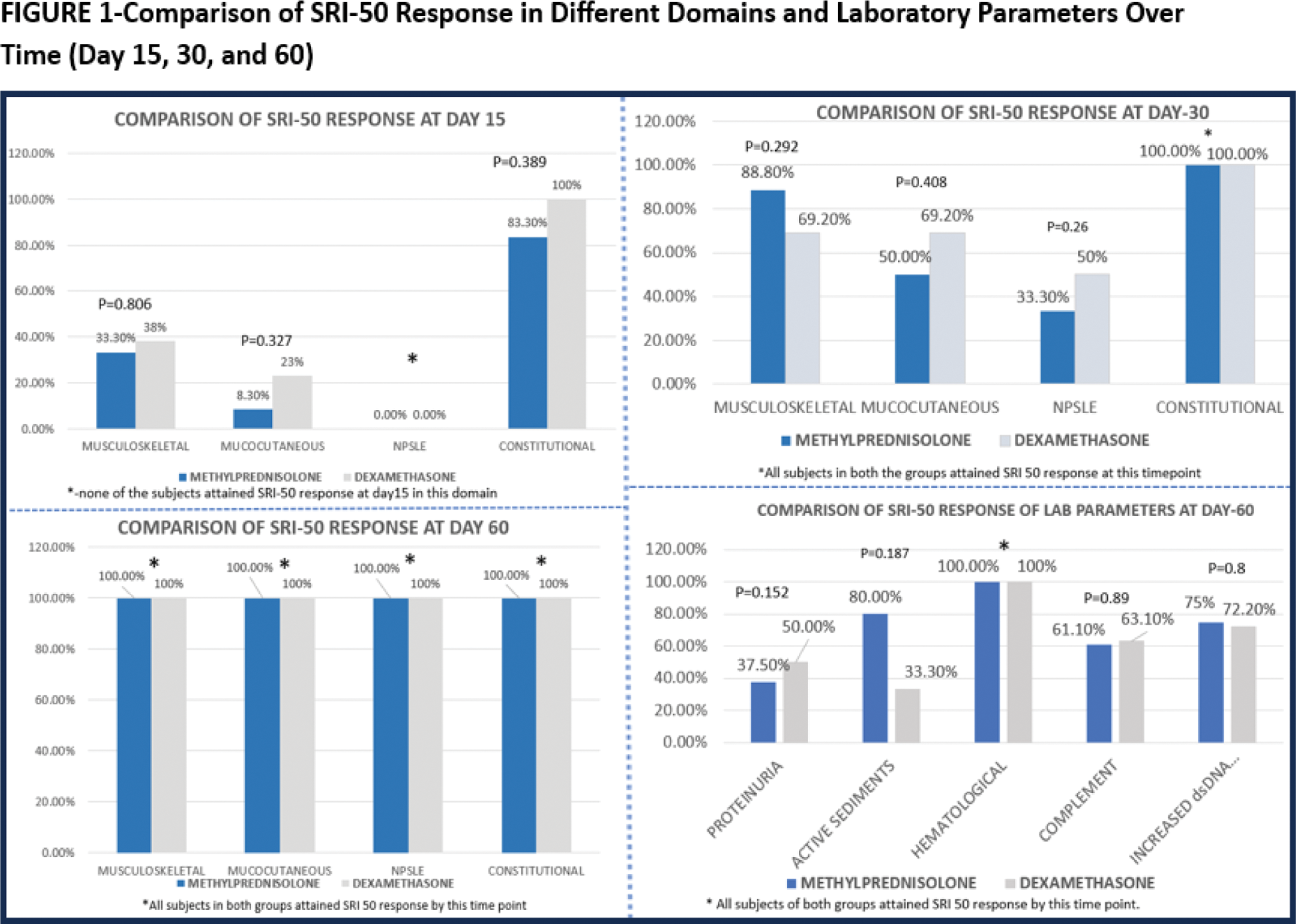
Results: We included 40 SLE patients, the baseline characteristics were well-matched between the groups, with mean SLEDAI-2K scores of 12.5 in the methylprednisolone group and 13.9 in the dexamethasone group (p = 0.386). The cumulative steroid dose at 60 days was higher in the methylprednisolone group, though the difference was not significant (Table 1). Both groups showed significant improvement in SLEDAI-2K scores from baseline to day 60, with no statistically significant difference at any time point (p > 0.05) (Table 1). SLEDAI-2KG scores followed a similar trend, indicating comparable steroid-sparing effects in both groups. Across various SLE domains, SRI-50 response rates at days 15, 30, and 60 showed no statistically significant differences between methylprednisolone and dexamethasone (Figure 1). In all the clinical domains, SRI 50 response was achieved in 100% of the patients in both the groups by day 60. Among laboratory parameters, both treatments achieved 100% response by day 60 in the hematological domain. Constitutional domain was the earliest to show SRI 50 response by day 15(83% of methylprednisolone vs 100% of dexamethasone group) (Figure 1). Cost of dexamethasone was significantly lesser than methylprednisolone ($0.77 vs $33.69 respectively).
Conclusion: This study demonstrates that dexamethasone may be as effective as methylprednisolone in reducing disease activity in SLE patients with moderate to severe disease. Both drugs resulted in comparable rates of SRI-50 improvement and similar trends in SLEDAI-2K and SLEDAI-2KG scores over the 60-day follow-up period. Dexamethasone offers a clear cost advantage, making it a valuable alternative, particularly in resource-limited settings. Future multi-center trials with larger sample sizes and longer follow-up durations are warranted to confirm these findings and explore long-term outcomes.
REFERENCES: [1] Touma Z, Gladman DD, Ibañez D, Urowitz MB. Development and initial validation of the systemic lupus erythematosus disease activity index 2000 responder index 50. J Rheumatol 2011;38:275–84.
[2] Touma Z, Gladman DD, Su J, Anderson N, Urowitz MB. A novel lupus activity index accounting for glucocorticoids: SLEDAI-2K glucocorticoid index. Rheumatology (Oxford). 2018 Aug 1;57(8):1370-1376. doi: 10.1093/rheumatology/key103. PMID: 29688532.
[3] Wei Y, Ji X-B, Wang Y-W, Wang J-X, Yang E-Q, Wang Z-C, et al. High-dose dexamethasone vs prednisone for treatment of adult immune thrombocytopenia: a prospective multicenter randomized trial. Blood 2016;127:296–302.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (