

Background: The clinical benefits of rituximab (RTX) in treating patients with autoimmune rheumatic diseases (ARDs) must be carefully balanced against its potential to increase susceptibility to infections, particularly herpes zoster (HZ). The recently developed recombinant zoster vaccine (RZV, Shingrix®) has demonstrated a favorable safety profile. However, immunogenicity and safety of the RZV in ARDs patients, specifically those undergoing RTX therapy, remains underexplored, leaving a critical gap in understanding vaccine response in this context.
Objectives: This study aims to evaluate the impact of RTX on humoral immunogenicity by comparing ARD patients treated with RTX to healthy controls and to ARD patients without RTX vaccinated with RZV (primary endpoint). Secondary objectives were assessment of the influence of demographic, clinical factors, and concomitant medications on the immune response to RZV.
Methods: This study was a prospective, controlled, randomized, phase 4 trial involving adult ARD patients treated with at least one previous RTX cycle, 22-28 weeks prior to the last infusion (ARD-RTX group). These patients were initially compared to a control group (CG) of non-immunosuppressed individuals in a 1:3 (ARD-RTX:GC) ratio. An additional comparison group of ARD patients not treated with RTX (ARD NO-RTX group) balanced with ARD-RTX group by age, sex and ARD diagnosis and randomly selected in a 1:3 ratio (ARD-RTX:ARD-NO-RTX) was assessed. ARD and CG received two intramuscular doses of the recombinant zoster vaccine (RZV) administered 6 weeks apart, on day 0 (V1) and day 42 (V2). Blood samples were obtained before the first RZV dose and 6 weeks after the second dose and humoral immunogenicity was evaluated using an in-house ELISA to quantify serum anti-gE antibody concentrations. Humoral response to the vaccine was defined as an anti-gE antibody concentration at least 4 times above the lower detection limit (0.02 mIU/mL) for initially seronegative individuals or a 4-fold increase compared to baseline levels in those who were already seropositive (>0.02 mIU/mL). Geometric mean titers (GMT) were calculated based on log-transformed antibody concentrations. Adverse events were recorded using a standardized questionnaire.
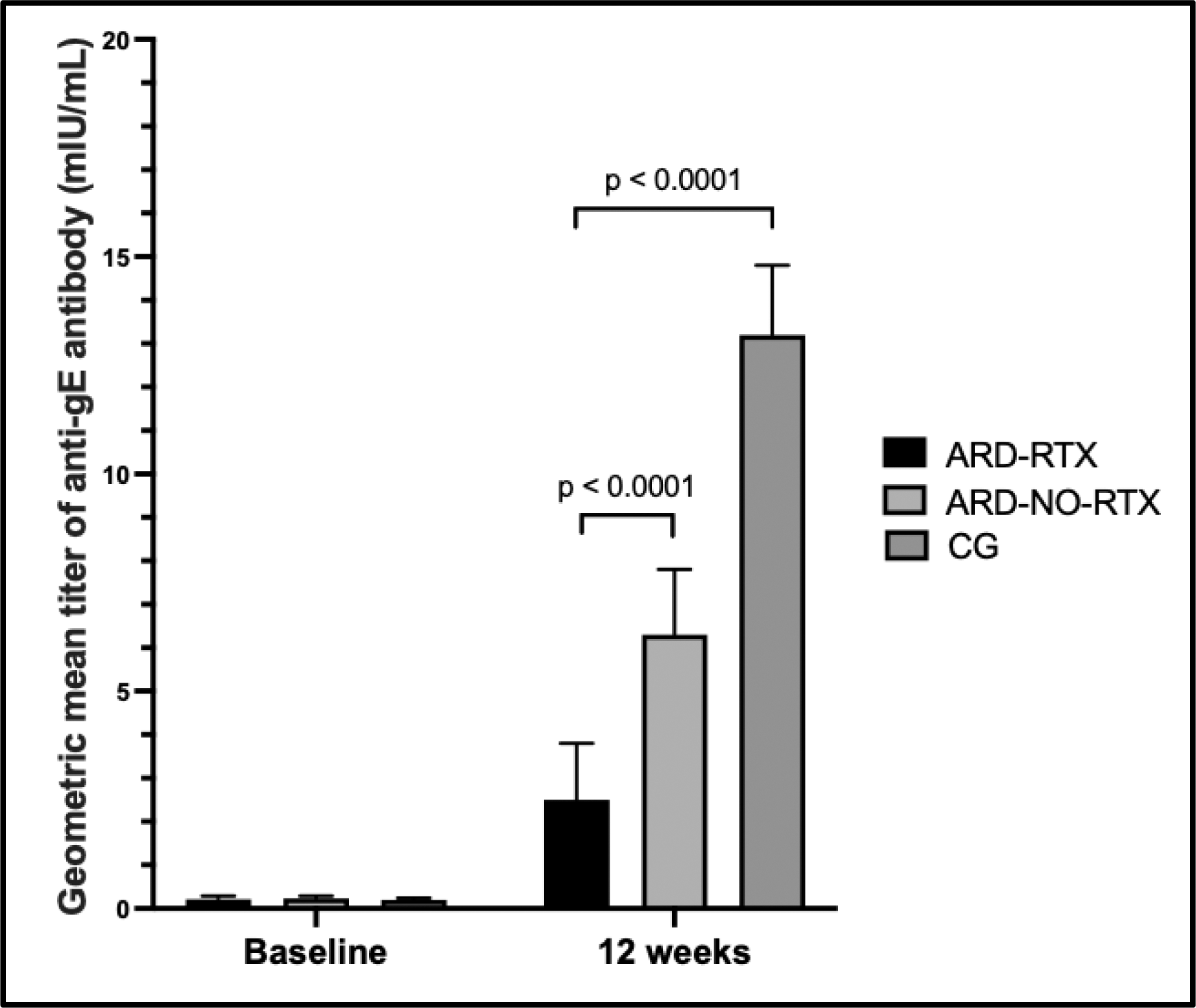
Results: A total of 67 patients in the ARD-RTX group, 201 CG participants and 201 ARD-NO-RTX group had complete data for the first 12 weeks and were included in this interim analysis. The ARD-RTX included patients with rheumatoid arthritis (39%), systemic lupus erythematosus (24%), Sjögren disease (13%), systemic sclerosis (12%), systemic autoimmune myopathy (10%), and ANCA-associated vasculitis (1%). Ongoing treatments were prednisone (49%) and immunosuppressants (81%), with the most commonly used agents being mycophenolate mofetil (37%), methotrexate (22%), azathioprine (13%), leflunomide (5%), and ciclosporin (3%). The ARD-RTX group were younger than CG (50 vs. 56 years, P<0.001), whereas the ARD-NO-RTX group had similar mean age (p=0.970) and sex distribution (p=581). Additionally, ARD diagnoses (p>0.99), glucocorticoids use (p=0.175) and immunosuppressive drugs (p>0.05) were comparable between ARD-RTX and ARD-NO-RTX groups, except for a higher leflunomide use in the later (9% vs. 23%, p=0.010). ARD-RTX patients demonstrated a lower humoral response compared to CG group (66% vs. 99%, p<0.0001) and to ARD-NO-RTX (66% vs. 86%, p<0.0001). Baseline GMTs of anti-gE antibodies were comparable between the three groups (p=487). However, following two vaccine doses, ARD-RTX group had significantly lower GMT compared to CG [2.5 (95% CI 1.7–3.8) vs. 13.2 (95% CI 11.8–14.8) mUI/mL; p <0.001] and ARD-NO-RTX group [2.5 (95% CI 1.7–3.8) vs. 6.3 (95% CI 5.1–7.8) mUI/mL; p <0.001] (Figure 1). Similarly, factor increase in GMT was lower in ARD-RTX group compared to CG group [11.9 (95% CI 7.3-19.2) vs. 74.9 (95% CI 53.9-78.1); p<0.001] and to ARD-NO-RTX group [11.9 (95% CI 7.3-19.2) vs. 26.9 (95% CI 20.9-34.6); p<0.001]. Analysis of potential factors influencing seroconversion showed no significant association with age, ARD diagnosis, glucocorticoid use, or concurrent immunosuppressive therapies (p>0.05). No cases of herpes zoster (HZ) confirmed by RT-PCR were reported up to week 12. Regarding vaccine safety, no severe adverse events were reported. However, local redness (19% vs. 32%, p=0.44), local pain (64% vs. 79%, p=0.014) and systemic reactions (40% vs. 65%, p<0.0001), including headache (21% vs. 37%, p=0.016) and fatigue (13% vs. 26%, p=0.035) following the first vaccine dose were significantly lower in ARD-RTX group compared to CG. Conversely, chills were more frequently reported in ARD-RTX group following both the first dose (9% vs. 2%, p=0.009) and pruritus after the second dose (8% vs. 2%, p=0.025).
Conclusion: This study provides novel data showing that RTX use significantly impairs the humoral response to RZV, independently of age, disease and other concomitant immunosuppressive therapies. These findings highlight the unique challenges ARD patients face in achieving adequate vaccine-induced immune response and emphasize the potential need for booster doses in this population (NCT05879419).
Comparison of humoral immunogenicity after RZV between autoimmune rheumatic diseases (ARD) patients under rituximab (ARD-RTX) with ARD without RTX (ARD-NO-RTX) and healthy control group (CG)
REFERENCES: NIL.
Acknowledgements: “Funding for this study was provided by GSK [NCT05879419]. GSK was provided the opportunity to review a preliminary version of this publication for factual accuracy, but the authors are solely responsible for final content and interpretation.”.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (