

Background: Chronic sleep disturbances have been found to have detrimental effects on health and functioning in both healthy individuals and SLE patients. Data on objective sleep assessment using actigraphy in rheumatic diseases is scarce let alone SLE. Actigraphy can be a useful tool to assess sleep by providing precise comprehensive data on sleep patterns, duration, and disturbances thus reducing reliance on individuals’ perceptions. It also provides the opportunity for conducting continuous assessments over extended periods in real time thus capturing variations and patterns that may not be evident in short-term. Our reason for choosing WSA was the affordability of the device, off-the-shelf availability as a consumer product, ease of use, unattended user experience, good results for apnea detection and lack of previous use in rheumatic diseases till date.
Objectives: In this study we aim to evaluate and compare the sleep quality in SLE patients and healthy controls using validated sleep questionnaires and Withing’s Sleep Analyzer (WSA). We also compared results of objective and subjective sleep assessment measures in cases.
Methods: Subjective sleep quality in enrolled SLE patients and age-sex matched controls was assessed using the Pittsburgh Sleep Quality Index (PSQI) and Epworth sleepiness scale (ESS). Data on demographics, disease severity i.e. activity and damage status were assessed during clinical examination using the SLEDAI-2K activity index and SLICC damage index respectively. Data on psychological status, physical function and overall quality of life was recorded using the Hospital anxiety and depression scale (HADS), Health assessment questionnaire (HAQ-CRD Pune version) and WHO Quality of life (QoL) scale respectively. This was followed by objective sleep assessment using the Withing’s Sleep Analyzer from the comfort of home. Cases and controls were asked to sleep on this novel non-wearable i.e. nearable device by placing it under their mattress for 5 days. Data was collected via mobile application and analysed.
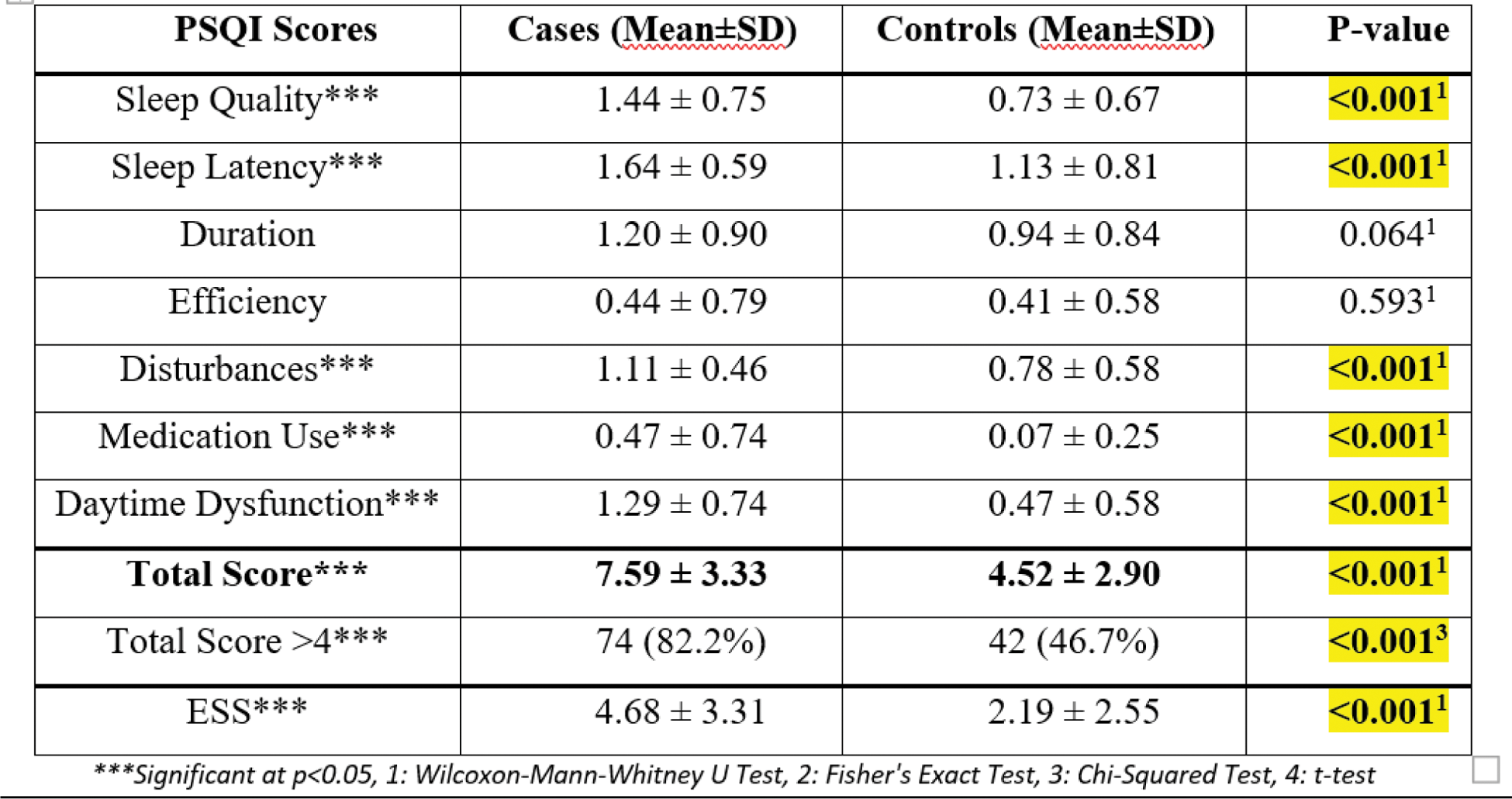
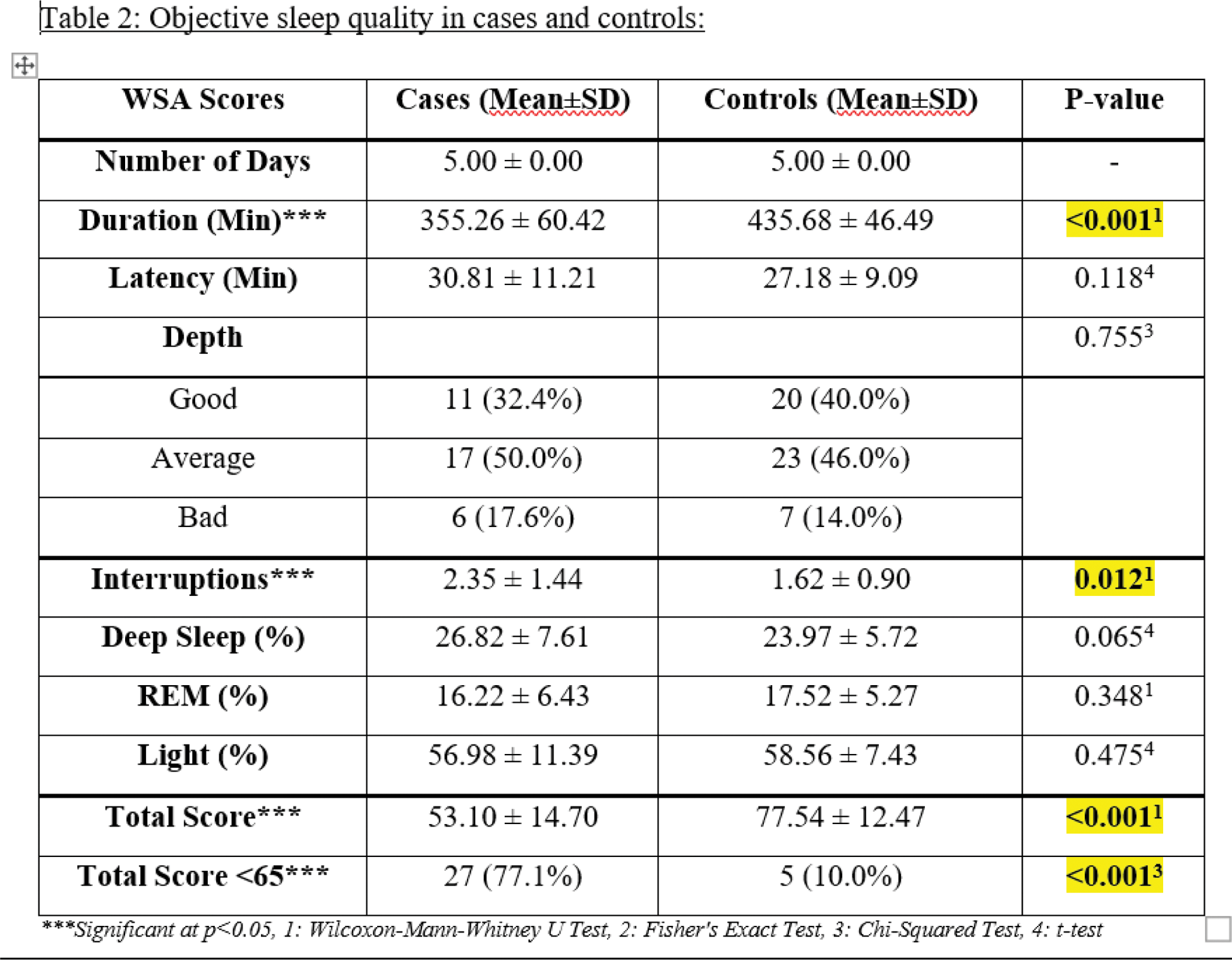
Results: 90 SLE cases and 90 healthy controls were enrolled in this study. In cases, the mean age was 37.11 ± 13.01 years with 90% females, and mean disease duration was 7.12 ± 6.65 years. Mean activity score was SLEDAI-2K of 4.01±3.72 and damage was SLICC-DI of 0.40 ± 0.76 (i.e. mild disease). Musculoskeletal involvement was seen in 72.2% cases, while 50% had mucocutaneous features, 30% had nephritis and 25.6% had haematological abnormalities as primary complaints. Cases demonstrated worse sleep quality than controls with a mean PSQI of 7.59 ± 3.33 and 4.52 ± 2.90 respectively (p<0.001). Cases also showed poorer PSQI component scores i.e. quality, latency, disturbances, medication use and daytime dysfunction (p<0.001). 82.2% cases demonstrated poor sleep (PSQI>4) compared to 46.6% controls. Mean ESS were also higher in cases than controls (4.68 ± 3.31 and 2.19 ± 2.55 respectively) (Table 1). 35 cases and 50 controls participated in the objective sleep assessment using WSA. WSA recorded overall sleep score from 0 to 100 with higher scores indicating better sleep. Cases recorded a mean sleep score of 53.10 ± 14.70 compared to 77.54 ± 12.47 in controls (p<0.005). 77.1% cases and only 10% controls demonstrated poor sleep (WSA score<65, p<0.001). Cases recorded average sleep duration of 355.2 ± 60.4 minutes, latency of 30.8 ± 11.2 minutes with 2.3 ± 1.4 nightly interruptions. Night’s sleep of a SLE patients consisted of 26.8% deep sleep, 16.2% REM sleep and 56.9% light sleep (Table 2). Cases had significantly shorter sleep duration and higher number of interruptions than controls (p<0.001 and p=0.01 respectively). WSA total score correlated well with PSQI total score (p<0.001) as well as several PSQI component scores including sleep duration (p<0.01), efficiency (p=0.02), medication use (p=0.02) and daytime dysfunction (p<0.001). Conversely, PSQI total score also correlated well with WSA sleep duration (p<0.001) and proportion of REM and light sleep (p< 0.01 and p=0.02 respectively). However, PSQI total score did not correlate with sleep latency, depth of sleep and number of interruptions recorded by WSA. PSQI score showed significant correlation with SLICC damage index in cases (p=0.02). However, no correlation could be established with total disease duration and SLEDAI-2K. WSA scores failed to show significant correlation with disease parameters. In cases, PSQI total score negatively correlated with all four WHO QoL domain scores (p<0.001). It also correlated significantly with the HAQ score (P=0.01) and Depression component of HADS score (p<0.001). WSA total score significantly correlated with WHO QoL physical domain score (p=0.02) but failed to correlate with other parameters in our cohort.
Conclusion: Objective sleep assessment using the Withing’s Sleep Analyzer revealed similar results on sleep quality in SLE patients as reported by subjective sleep measures. Both assessments recorded worse sleep quality in cases than controls and correlated well with each other. WSA failed to demonstrate association with disease parameters, psychological variables, physical function and overall QoL in this study. Sleep is multifactorial and symptoms such as fatigue, myalgia, joint pains and rashes significantly affect sleep but are missed on standard disease indices. Larger studies are needed with objective sleep assessment measures to better understand this interdependent relationship.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (