

Background: Xeligekimab (GR1501), the first self-innovated IL-17A inhibitor in China, has demonstrated its safety in healthy people in phase I studies.
Objectives: This multi-center, randomized, placebo-controlled,double-blind phase 2 study (NCT05162937) was conducted to further assess the efficacy and safety of Xeligekimab (GR1501) in patients with Moderate to severe axial spondyloarthritis.
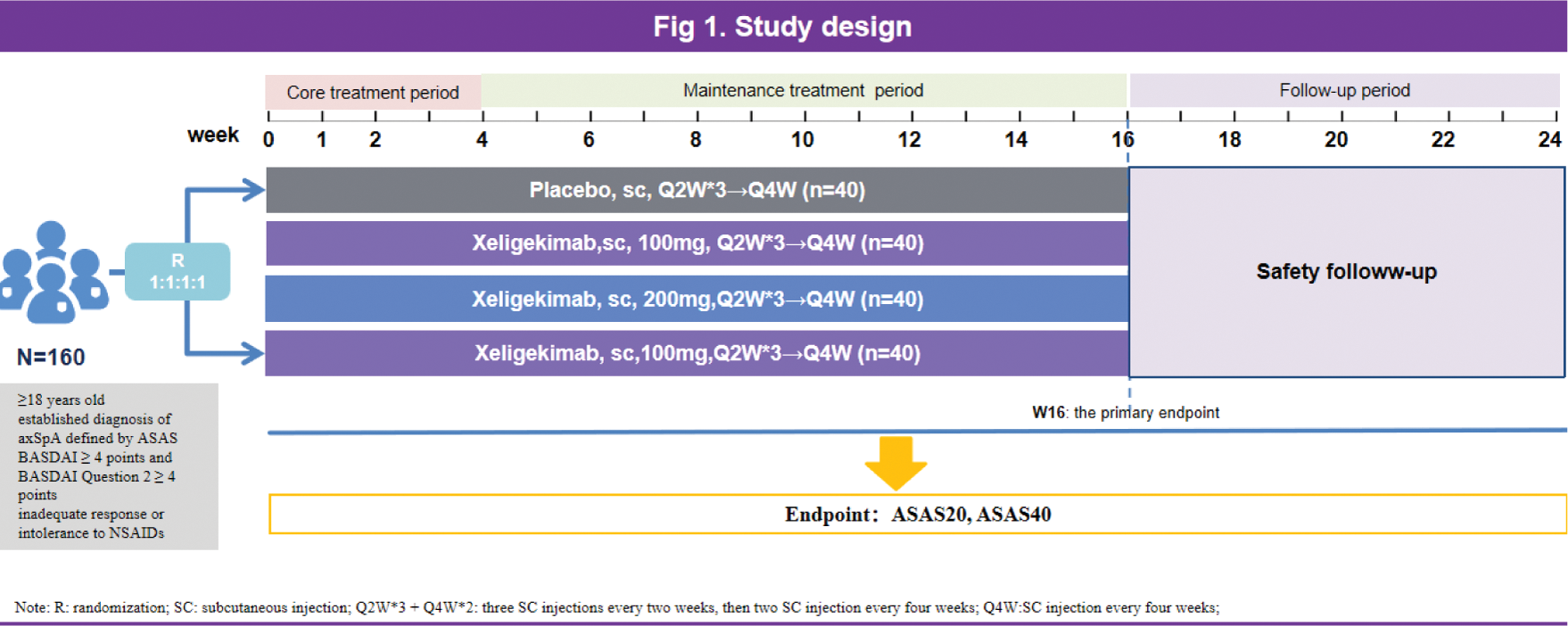
Methods: Eligible patients, aged 18-60 years,diagnosed with axial spondyloarthritis according to ASsessment in Ankylosing Spondylitis intermational Society (ASAS), exhibited moderately to severely active disease, were randomized (1:1:1:1) to receive either a placebo, 100mg, 200mg or 300mg of Xeligekimab (GR1501), administered Q2W for early 4 weeks, then Q4W until week 16 (last dose at week 12), following 8 weeks follow-up until week 24 (Figure 1). The primary endpoint was the proportion of patients who achieving ASAS 20 at week 16.
Schematic overview of the study cohort.
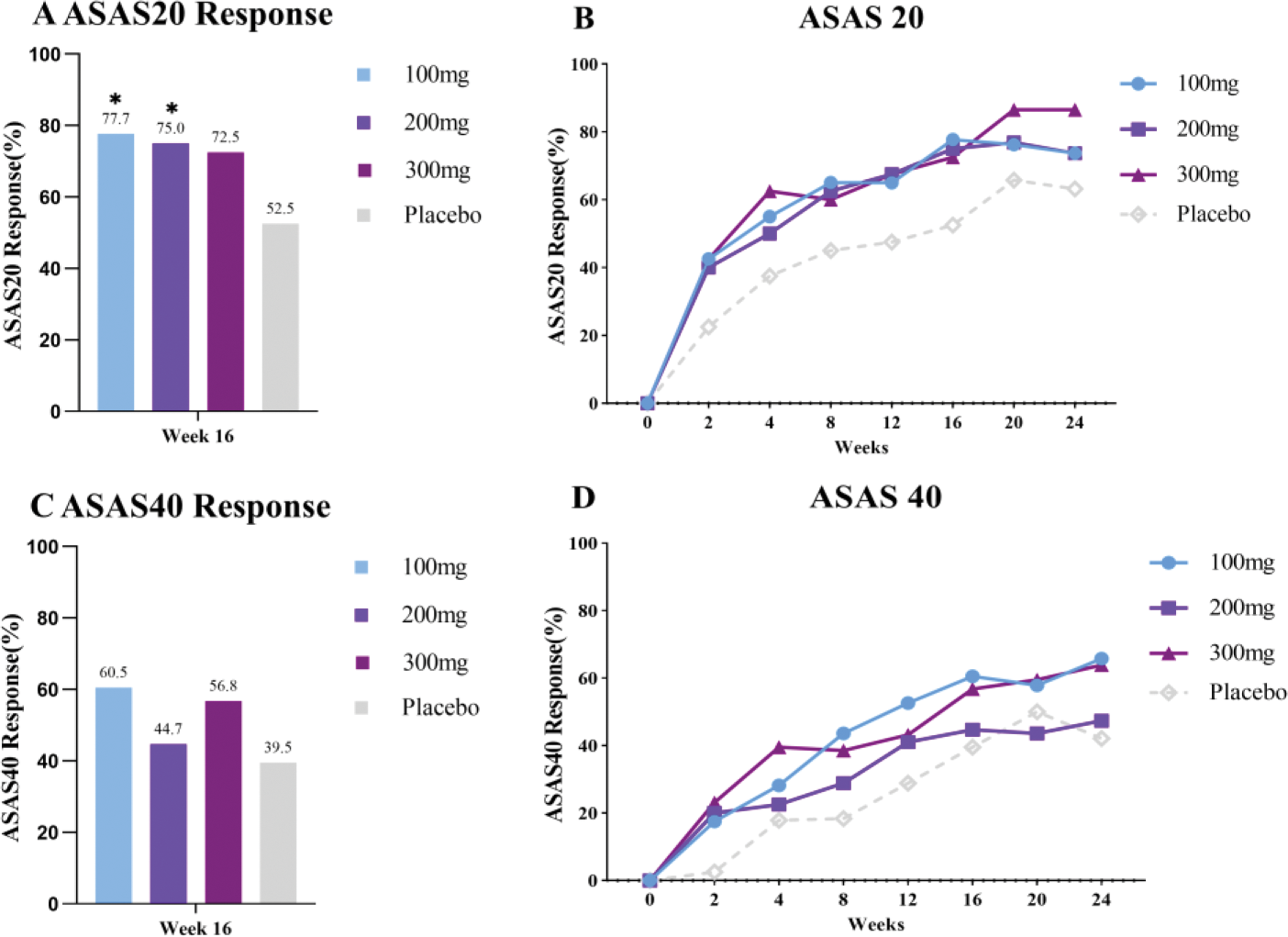
Results: A total of 160 patients were randomized, and 150(93.8%) of them completed the 16-week treatment. The proportion of patients who achieved ASAS 20 at 16 weeks after treatment in the placebo group (n=40), 100 mg group (n=40), 200 mg group (n=40), and 300 mg group (n=40) were 52.5%, 77.5%, 75.0%, and 72.5%, respectively (Figure 2A). There were statistically significant differences between the 100 mg group and the placebo group and the 200 mg group and the placebo group (P<0.05)。Figure 2A and Figure 2C show the effects of different doses of medication on ASAS20 and ASAS40 response rates at week 16. Figure 2B and Figure 2D illustrate the effects of different doses of medication on ASAS20 and ASAS40 response rates over the treatment period. It can be observed that response rates increase over time in all dose groups, but there are differences in response rates among different dose groups. Throughout the study period, 87.5% of treatment-related adverse events (TEAEs) occurred in the 100 mg group, 77.5% in the 200 mg group, 90.0% in the 300 mg group, and 77.5% in the placebo group. However, there were no adverse events leading to death or withdrawal from the trial in either the experimental group or the control group.
Effects of different Doses of Xeligekimab (GR1501) on ASAS20 and ASAS40.A ASAS20 Response at Week 16;B. ASAS20 Response 0-24Week;C ASAS40 Response at Week 16; D. ASAS20 Response 0-24Week.*P<0.05 vs. the Placebo group.
Conclusion: Xeligekimab (GR1501) 100~300mg group showed great efficacy and safety in patients with moderate to severe axial spondyloarthritis, but compared with the 100mg group and the 200mg group, the overall incidence of adverse events in the 300mg dose group was higher, so 100mg and 200mg were to be chosen for Xeligekimab (GR1501) phase III study.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (