

Background: Quality of life (QoL) is a multidimensional concept that encompasses an individual’s overall well-being, including physical, mental, and social aspects. In the context of systemic lupus erythematosus (SLE), a chronic autoimmune disease, assessing QoL is particularly crucial due to the diverse and often debilitating manifestations of the disease. Although a recent study did not find an association between adherence to the EULAR quality indicators and patient reported outcomes, disease remission appears to be linked to an improvement in physical but not mental QoL. This suggests a potential discordance between patients and physician’s perceptions.
Objectives: This study aims at describing the burden of disease in terms of quality of life and to explore its determinants in Swiss SLE patients.
Methods: In this cross-sectional study, patients living in Switzerland were invited to participate in the online survey via 3 different channels: i) digital advertisements targeted to SLE patients (google search and ads), ii) newsletter of a lupus patient association (Lupus Suisse), and iii) letters to patients of a national Lupus registry (Swiss SLE Cohort Study, SSCS). Patients with self-reported physician’s diagnosis of SLE, aged ≥ 18 years, willing to provide electronic consent for the study, residing in Switzerland and able to read and complete the survey in German, French or Italian were included in the study. Self-reported burden of disease was assessed using the Lupus Patient-Reported Outcome scale LupusPRO™ (©, 2007, Rush University Medical Center and the Board of Trustees at the University of Illinois, Chicago) which was designed and validated to assess the impact of SLE on patients’ health-related quality of life (HRQol). The derived HRQoL total score as well as its 7 domain scores for symptoms, cognition, medications, procreation, physical health, pain, emotional health, and body image are ranging from 0 (worst situation) to 100 (best situation). The survey also included questions regarding age, gender, region of living, treatment modalities, treatment satisfaction (6 dimensions derived from the Lupus Europe survey), and disease activity (assessed using the Systemic Lupus Activity Questionnaire (SLAQ)) providing a holistic view of the patients’ experience. Median and IQR were calculated for descriptive statistics, mean values, and standard deviations for score calculations. To build high and low HRQoL and disease activity strata, the respective median was applied. Multiple linear regression was used to explore associations between HRQoL total score and independent variables. Comparisons between independent groups were made using the Mann-Whitney U test for continuous and the Fisher’s exact test for categorical data, without correction for multiplicity due to the exploratory nature of the survey. The study was approved by the Cantonal Ethics Committee of Geneva (2023-01927).
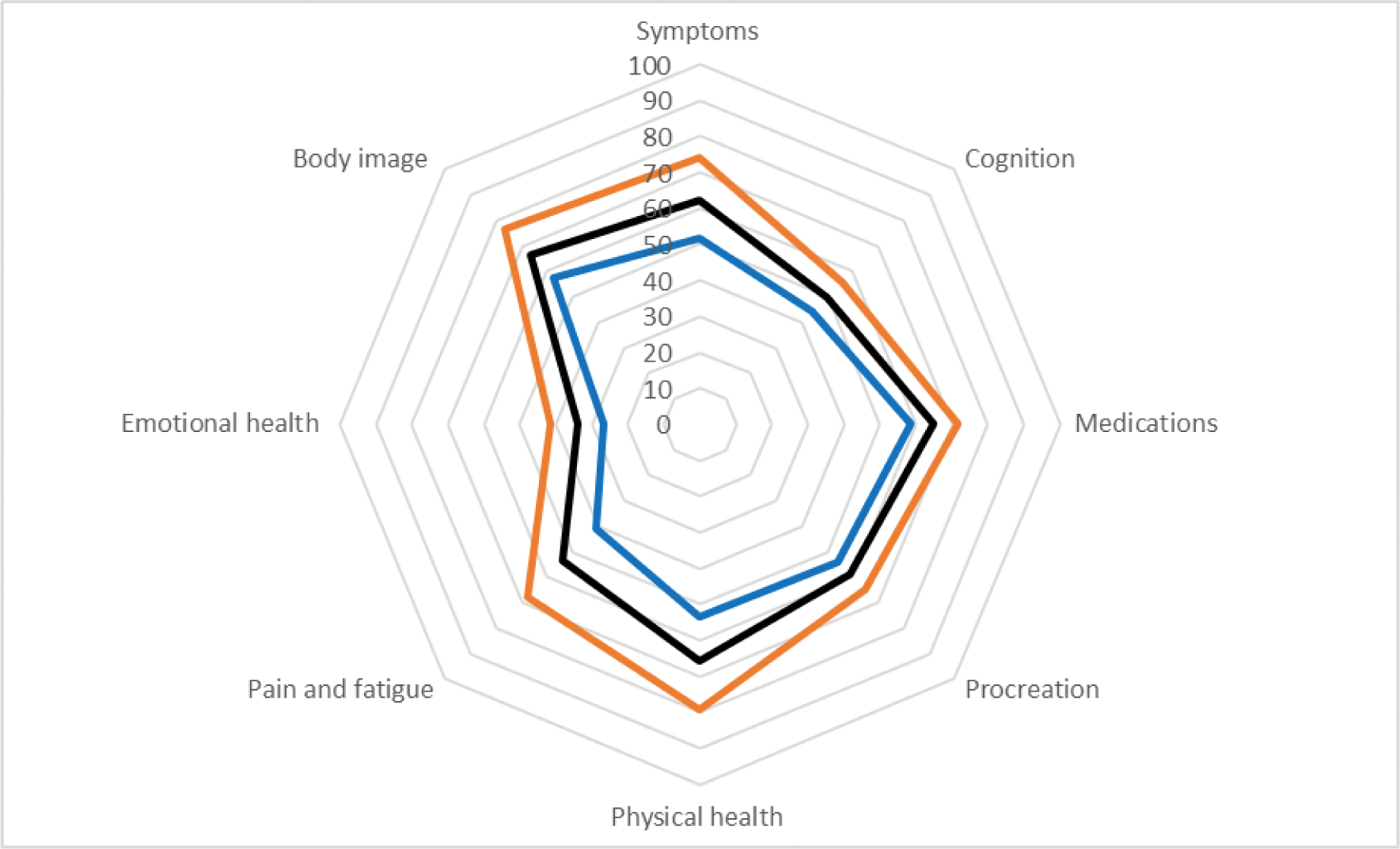
Results: Between February 14 th and June 25 th , 2024, 282 patients were screened, of which 27 did not meet the inclusion criteria. Out of the remaining 255 patients, 109 participants left the online survey incomplete, leaving a total of 146 patients included in the analysis. Participants had a median age of 48.5 years, were predominantly female (85.6%) and Caucasians (76.7%). The median duration of SLE was 12 years, and the most commonly used SLE medications were antimalarials (73.3%), immunosuppressants (36.3%), and glucocorticoids (34.2%). Among patients taking other medications besides SLE treatments (n=108), those with a low HRQoL were more likely to use painkillers, antidepressants or anxiolytics compared to those with high HRQoL (P<0.01 for both types of medications). Further treatment characteristics stratified by patients with high and low HRQoL can be found in Table 1. Overall LupusPRO™ Health Related QoL score was 55.9 with particularly low scores in emotional health (score 33.9). Figure 1 shows overall results as well as results stratified by low and high disease activity. Regression analysis (adjusted for age, gender, time since diagnosis, ethnicity, language and geographic region, medication, social and psychological support) revealed a significant negative correlation between HRQoL and disease activity (β = -0.608, p < 0.01), avoidant coping strategies (β = 0.420, p < 0.05), daily pill intake (β = -0.253, p < 0.01) and the occurrence of flares in the last 3 months (β = -0.498, p < 0.01). Average treatment satisfaction across all 6 dimensions varied significantly by recruitment method with the lowest percentage reported among digitally recruited participants with 50.0% vs. 69.4% in patients recruited via the patient association and 75.3% in patients recruited via the SLE cohort study (p < 0.01 both vs digital recruitment).
Conclusion: Compared to previous studies, our cohort of SLE patients showed a relatively low health related QoL, particularly driven by the emotional health and cognition domains. Furthermore, we could identify an increase in disease activity, avoidant coping, daily pill intake, and the presence of flares in the last 3 months as predictors for a decreased HRQoL. Further investigations will be necessary to gain a deeper understanding of the cause-and-effect relationship between QoL and these contributing factors.
Table 1. Treatment characteristics in patients with high and low HRQoL based on Lupus PROTM total scores .
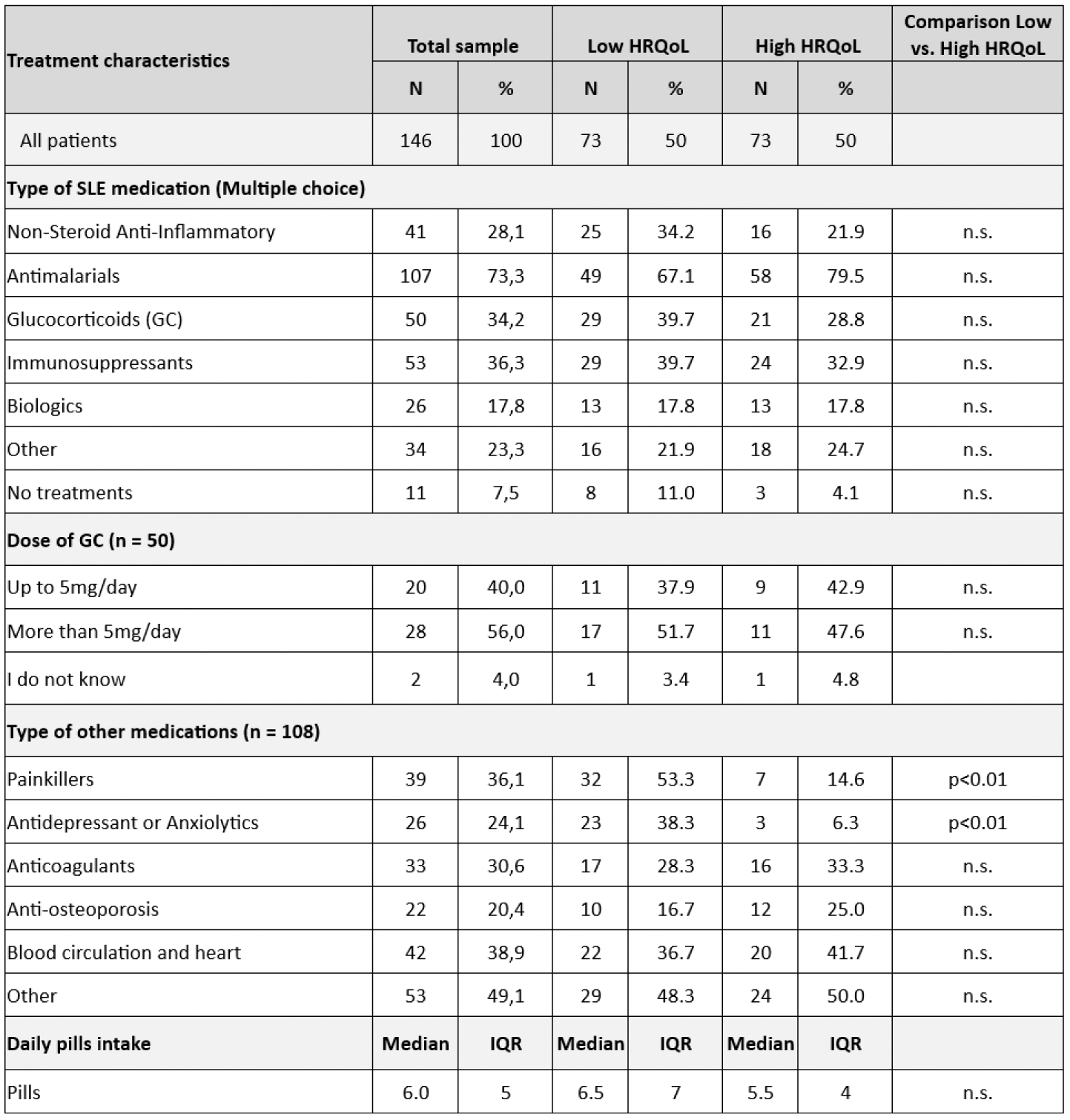
Radar diagram of LupusPRO TM domains scores for the overall study population (black line) and in patients with high (blue line) and low (orange line) disease activity.
REFERENCES: NIL.
Acknowledgements: NIL.
Disclosure of Interests: Chiara Balbo Pogliano AstraZeneca, Jasmin Annabelle Schäfer: None declared, Kristin Schmiedeberg: None declared, Marten Trendelenburg Research collaborations with Roche, Novartis and Idorsia (all Switzerland), Johannes von Kempis: None declared, Peter Langer AstraZeneca, Philipp Wörner AstraZeneca ETF, AstraZeneca, Shekoofeh Yaghmaei AstraZeneca, Carlo Chizzolini Astra Zeneca, Boehringer Ingelheim (less than 10,000 Euro).
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (