

Background: Systemic lupus erythematosus (SLE) is characterized by heterogeneous, waxing and waning clinical manifestations, leading to substantial physical and psychological burden. Active disease and repetitive flares correlate with increased fatigue, pain, and accumulation of organ damage [1–3]. Patient-reported outcome measures, such as the Systemic Lupus Activity Score (SLAQ) are increasingly recognized for their role in capturing the patient experience and could be integrated as relevant outcome in therapeutic trials.
Objectives: This study aims at assessing the current state of self-reported disease activity and understanding the factors that contribute to disease activity in Swiss SLE patients.
Methods: In this cross-sectional study, patients living in Switzerland were invited to participate in the online survey via 3 different channels: i) digital advertisements targeted to SLE patients (google search and ads), ii) newsletter of a lupus patient association (Lupus Suisse), and iii) letters to patients of a national Lupus registry (Swiss SLE Cohort Study, SSCS). Patients with self-reported physician’s diagnosis of SLE, aged ≥ 18 years, willing to provide electronic consent for the study, residing in Switzerland and able to read and complete the survey in German, French or Italian were included in the study. Disease activity was assessed using the Systemic Lupus Activity Questionnaire (SLAQ). The SLAQ, a patient-reported assessment of subjective disease activity in SLE, consists of questions on 24 specific symptoms resulting in a symptom score (ranging from 0 to 24) and a weighted total score (ranging from 0 to 47). Furthermore, SLAQ covers the presence of flares, and a patient global assessment of disease activity (ranging from 0-10) [4]. The survey also included questions regarding age, gender, region of living, treatment modalities, and treatment satisfaction (6 dimensions derived from the Lupus Europe survey [5]), providing a holistic view of the patients’ experience. Median and IQR were calculated for descriptive statistics, mean values, and standard deviations for score calculations. To build high and low disease activity strata, the respective median was applied. Comparisons between independent groups were made using the Mann-Whitney U test for continuous and the Fisher’s exact test for categorical data, without correction for multiplicity due to the exploratory nature of the survey. Multiple linear regression was used to explore associations between SLAQ total score and independent variables. The study was approved by the Cantonal Ethics Committee of Geneva (2023-01927).
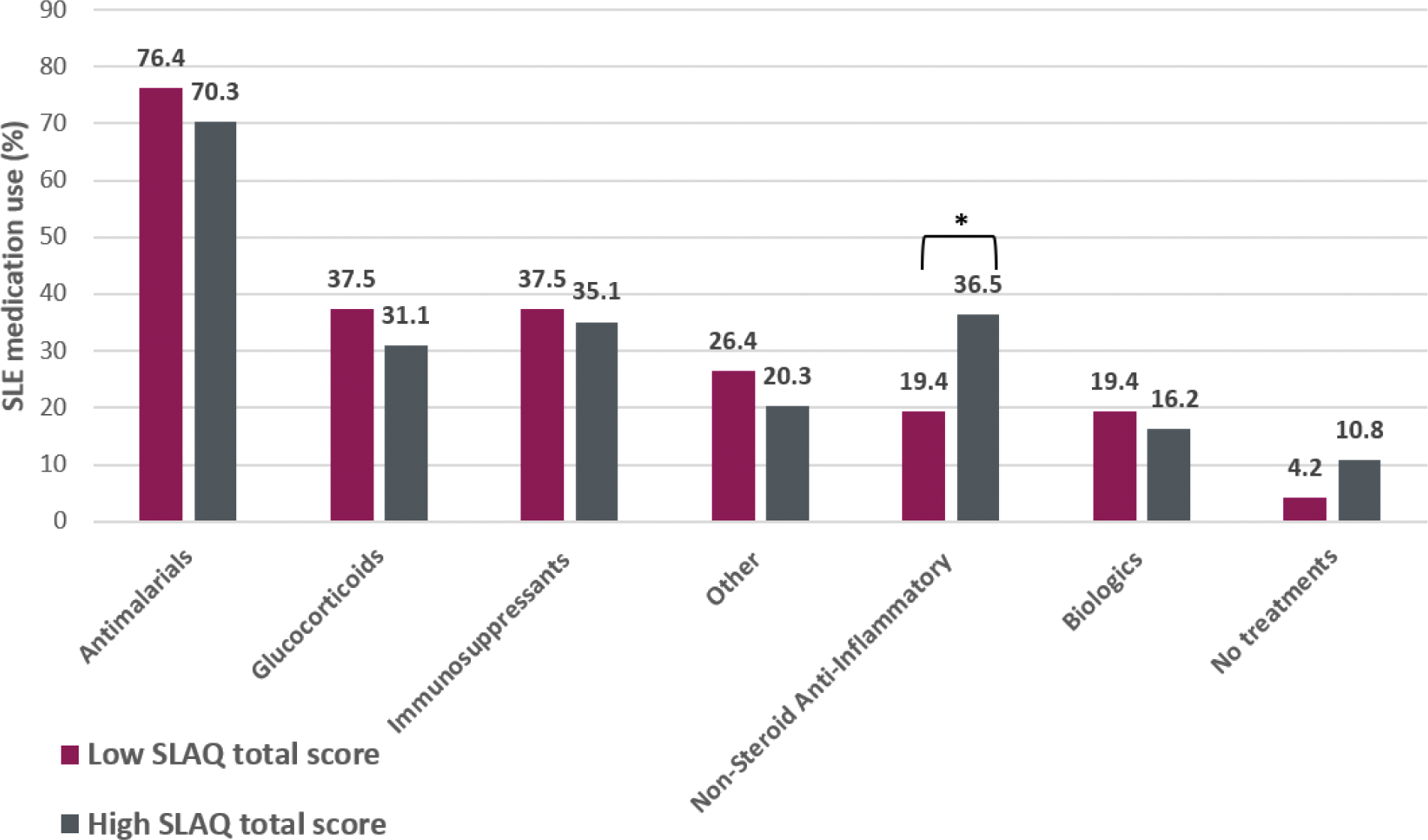
Results: Between February 14th and June 25th, 2024, 282 patients were screened, of which 27 did not meet the inclusion criteria. Out of the remaining 255 patients, 109 participants left the online survey incomplete, leaving a total of 146 patients included in the analysis. Participants had a median age of 48.5 years, were predominantly female (85.6%) and Caucasians (76.7%). Median duration of SLE was 12 years and mostly used SLE medications were antimalarials (73.3%), immunosuppressants (36.3%), and glucocorticoids (GC) (34.2%). 56% of patients on GC indicated to take more than 5mg/day. The mean SLAQ total score was 19.35 and the overall disease activity 3.86. Moderate to severe flares in the last 3 months were reported by 28.7% of patients. From the patients that did not report a flare or had a mild flare only 6.7% indicated not to be on medication (Table 1). The regression analysis (adjusted for patient demographics, medication, disease duration, and disease activity) showed a significant positive correlation between SLAQ total score and disease activity (β = 0.587, p < 0.01), having flares (β = 0.512, p < 0.01) and daily pill intake (β = 0.237, p = 0.014). Average treatment usage depending on high/low SLAQ scores shows a higher usage of NSAIDs in patients with high disease activity (36.5% vs. 19.4%, p<0.01, Figure 1). Furthermore, antidepressants or anxiolytics were used more frequently in patients with high vs. low SLAQ total scores (36.2% vs. 10.0%, p<0.01). Average treatment satisfaction across all 6 dimensions was lower in patients with high disease activity (57.2% vs 75.2%, p<0.01).
Conclusion: These findings show the persisting disease burden in patients with SLE in Switzerland underscored by substantial disease activity, existence of flares and high usage of GC. Furthermore, the study reveals an association between increased disease activity and treatment patterns as well as overall treatment satisfaction. However, prospective studies are needed to better understand the causal relationship between these outcomes and self-reported disease activity.
REFERENCES: [1] Chaigne B, Chizzolini C, Perneger T, et al. Impact of disease activity on health-related quality of life in systemic lupus erythematosus – a cross-sectional analysis of the Swiss Systemic Lupus Erythematosus Cohort Study (SSCS). BMC Immunology 2017; 18 (1): 17.
[2] Fanouriakis A, Tziolos N, Bertsias G, Boumpas DT. Update on the diagnosis and management of systemic lupus erythematosus. Annals of the Rheumatic Diseases 2021; 80 (1): 14-25.
[3] Katz P, Nelson WW, Daly RP, Topf L, Connolly-Strong E, Reed ML. Patient-Reported Lupus Flare Symptoms Are Associated with Worsened Patient Outcomes and Increased Economic Burden. J Manag Care Spec Pharm 2020; 26 (3): 275-83.
[4] Karlson E, Ma H, Rivest C, et al. Validation of a Systemic Lupus Activity Questionnaire (SLAQ) for population studies. Lupus 2003; 12 : 280-6.
[5] Cornet A, Andersen J, Myllys K, Edwards A, Arnaud L. Living with systemic lupus erythematosus in 2020: a European patient survey. Lupus Sci Med 2021; 8 (1).
Table 1. Patient and treatment characteristics.
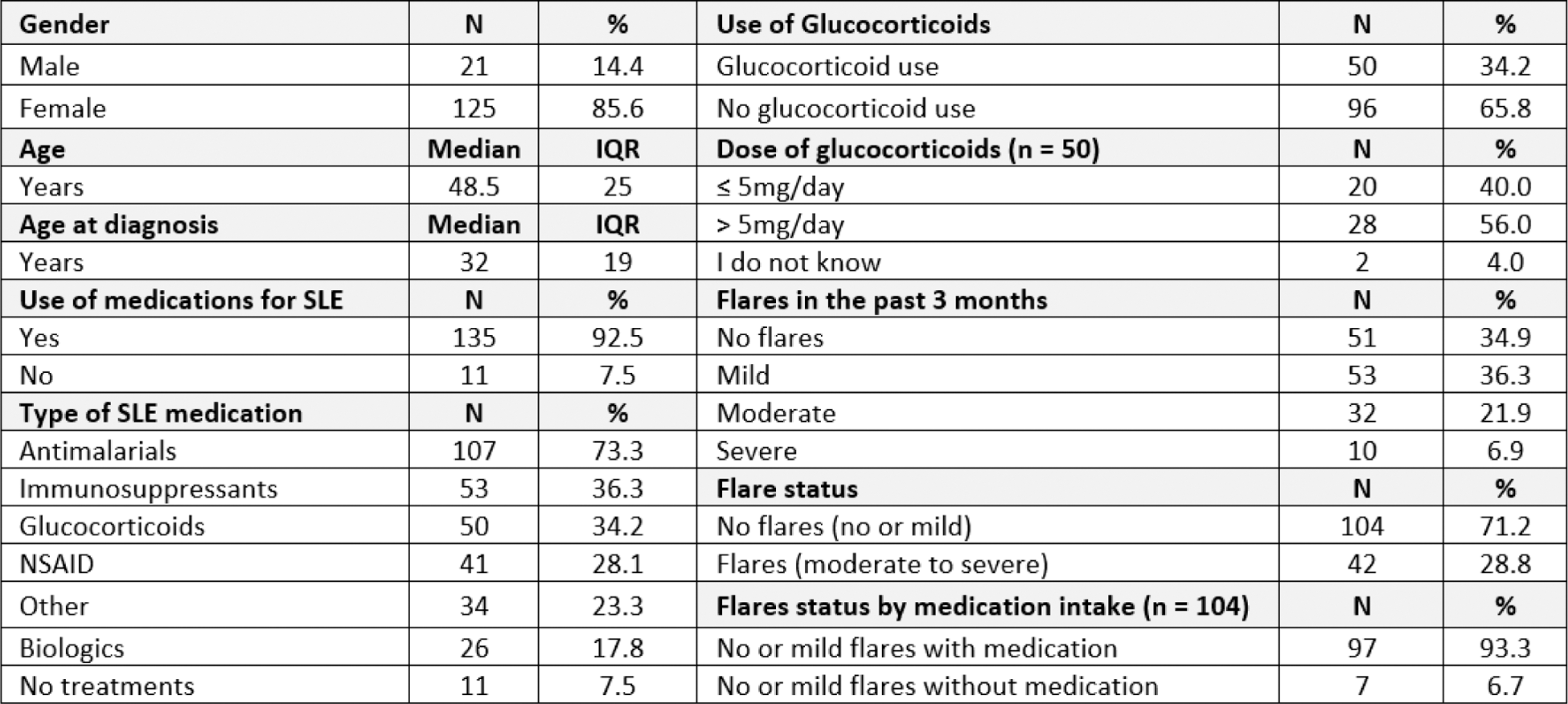
SLE medication distribution depending on low/high SLAQ total score. * = p<0.01
Acknowledgements: NIL.
Disclosure of Interests: Philipp Wörner Global ETF, AstraZeneca, Jasmin Annabelle Schäfer: None declared, Kristin Schmiedeberg: None declared, Marten Trendelenburg Research collaborations with Roche, Novartis and Idorsia (all Switzerland), Johannes von Kempis: None declared, Chiara Balbo Pogliano AstraZeneca, Peter Langer AstraZeneca, Shekoofeh Yaghmaei AstraZeneca, Carlo Chizzolini AstraZeneca, Boehringer Ingelheim (less than 10,000 Euro).
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (