

Background: Psoriatic arthritis (PsA) occurs in males and females at similar rates. Females participating in randomized controlled trials (RCTs) have shown lower treatment efficacy based on composite scores, especially pertaining to biologic disease-modifying anti-rheumatic drug (DMARD) treatments [1]. Important sex differences in baseline patient (pt) characteristics were identified in a large, pooled cohort of PsA pts from 3 Phase 3 RCTs of guselkumab (GUS), where females exhibited longer PsA duration; higher body mass index (BMI), tender joint count (TJC) and Leeds Enthesitis Index (LEI) score; and more severe PsA impact on quality of life [2].
Objectives: Given the importance of achieving low levels of disease activity across PsA domains, we assessed sex-disaggregated GUS efficacy across multiple PsA signs and symptoms at Week (W) 24 in this same pooled cohort.
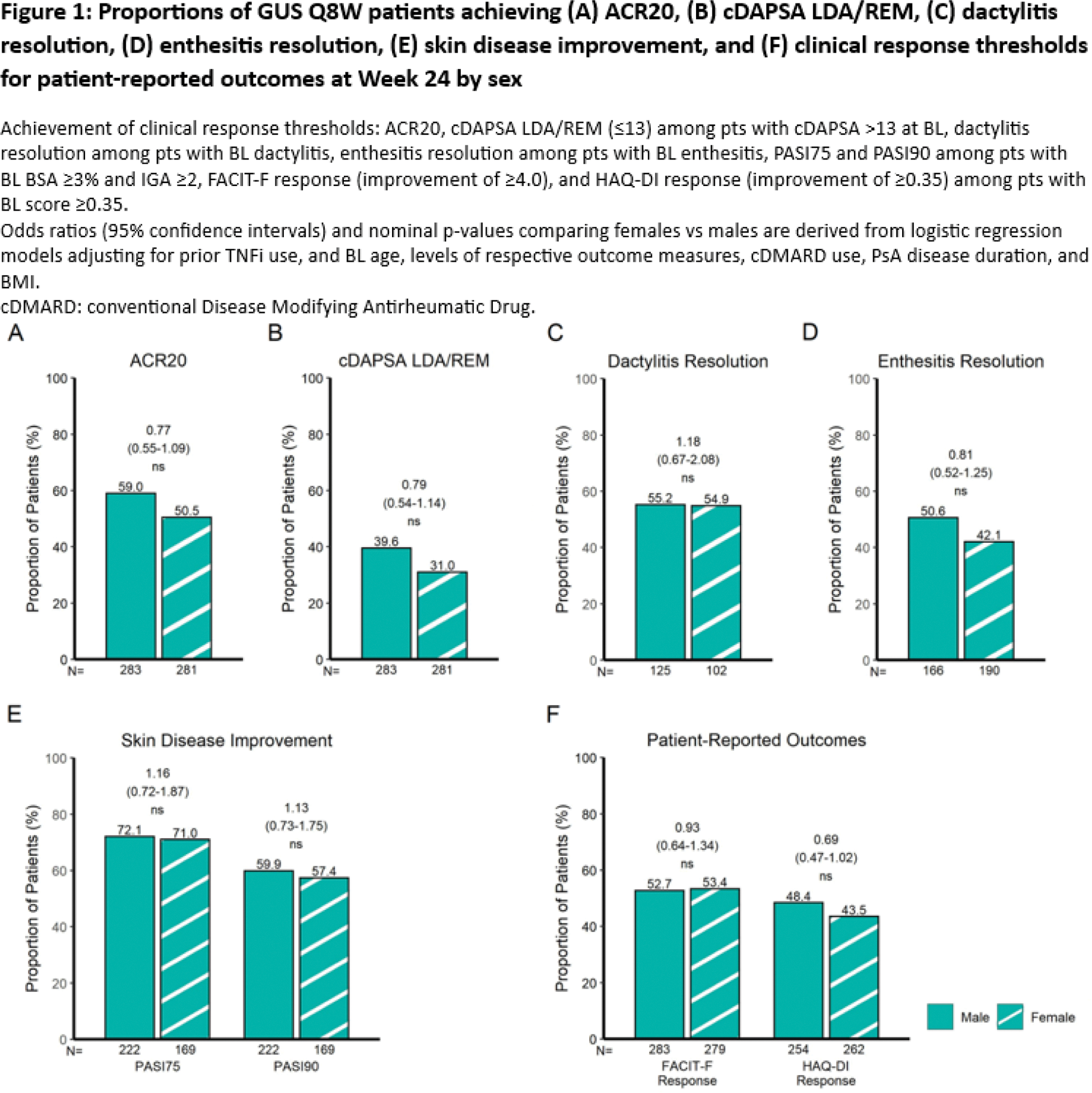
Methods: Post hoc analyses included 381 PsA pts from DISCOVER-1 (NCT03162796), 739 from DISCOVER-2 (NCT03158285), and 285 from COSMOS (NCT03796858). Rates of achievement of ACR20, Clinical Disease Activity Index for PsA (cDAPSA) low disease activity (LDA)/remission (REM), dactylitis and enthesitis resolution, Psoriasis Area Severity Index improvement from baseline of at least 75% (PASI75), PASI90, as well as Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F) and Health Assessment Questionnaire-Disability Index (HAQ-DI) response at W24 were evaluated by sex in pts receiving GUS at W0, W4 and every 8 weeks (Q8W). Non-responder imputation was applied for missing data. Achievement of clinical response at W24 was compared between GUS Q8W-treated male and female pts via adjusted logistic regression (Figure 1).
Results: Among 564 PsA pts treated with GUS Q8W, no significant difference in GUS efficacy was observed between female and male pts as measured by the primary efficacy outcome common to the 3 RCTs (ACR20) and across PsA domains at W24 (Figure 1). After adjusting for baseline characteristics with sex-specific differences in the pooled cohort, including age (female vs male; mean: 48.2 vs 46.1 years; p=0.0007), PsA duration (6.9 vs 6.0 years; p=0.0086), and BMI (29.9 vs 28.8 kg/m 2 ; p=0.0007) 2 , GUS efficacy was found to be comparable between sexes in terms of achieving ACR20 response (female vs male; 50.5 vs 59.0%; odds ratio [OR] [95% confidence interval (CI)]: 0.77 [0.55-1.09]; not significant [ns]; Figure 1A); composite measure indicating low levels of joint disease activity (cDAPSA LDA/REM: 31.0 vs 39.6%; OR [95% CI]: 0.79 [0.54-1.14]; ns; Figure 1B); dactylitis resolution (54.9 vs 55.2%; OR [95% CI]: 1.18 [0.67-2.08]; ns; Figure 1C); enthesitis resolution (42.1 vs 50.6%; OR [95% CI]: 0.81 [0.52-1.25]; ns; Figure 1D); skin disease improvement (PASI75: 71.0 vs 72.1%; OR [95% CI]: 1.16 [0.72-1.87]; ns and PASI90: 57.4 vs 59.9%; OR [95% CI]: 1.13 [0.73-1.75]; ns; Figure 1E); and clinically meaningful improvements in pt-reported outcomes (FACIT-F: 53.4 vs 52.7%; OR [95% CI]: 0.93 [0.64-1.34]; ns and HAQ-DI: 43.5 vs 48.4%; OR [95% CI]: 0.69 [0.47-1.02]; ns; Figure 1F).
Conclusion: In a large, pooled cohort of PsA pts from 3 RCTs, sex has no significant impact on GUS efficacy as measured by ACR20 and across PsA domains, after adjusting for the sex differences in baseline characteristics.
REFERENCES: [1] Eder. Lancet Rheum 2023;5:e716-27.
[2] Coates et al, ACR 2024; AB1832254.
Acknowledgements: NIL.
Disclosure of Interests: Lihi Eder Consultant: AbbVie, Eli Lilly, Janssen, Novartis, Pfizer, and UCB, Research grants: AbbVie, Eli Lilly, Janssen, Novartis, Pfizer, Sandoz, and UCB, Carlo Selmi Consulting/speaker fees: AbbVie, Alfa-Wassermann, Amgen, Biogen, Eli Lilly, EUSA, Galapagos, Janssen, Novartis, and SOBI, Grant/research support: AbbVie, Amgen, and Pfizer, Philip J. Mease Grants: AbbVie, Amgen, Eli Lilly, Janssen, Novartis, Pfizer, and UCB, Consulting fees: AbbVie, Acelyrin, Amgen, Bristol Myers Squibb, Eli Lilly, Immagene, Janssen, Novartis, Pfizer, UCB, and Ventyx, Grants: AbbVie, Acelyrin, Amgen, Bristol Myers Squibb, Eli Lilly, Janssen, Novartis, and UCB, Alexis Ogdie Consulting/Advisory Boards: AbbVie, Amgen, Bristol Myers Squibb, Celgene, Corrona, Eli Lilly, Gilead, GlaxoSmithKline, Janssen, Novartis, Pfizer, and UCB, Grants: AbbVie to Penn, Amgen to Forward/NDB, Novartis to Penn, Pfizer to Penn; Other Funding: National Psoriasis Foundation, NIAMS, Rheumatology Research Foundation, University of Pennsylvania, Francois Nantel Shareholder: Johnson & Johnson, consultant: Janssen, Frederic Lavie Employee of Immunology Global Medical Affairs, Janssen Pharmaceutical Companies and owns stock in Johnson & Johnson, Mohamed Sharaf Employee of EMEA Medical Affairs, Johnson & Johnson Middle East FZ LLC, Dubai, United Arab Emirates and owns stocks in Johnson & Johnson, Emmanouil Rampakakis Employee of JSS Medical Research, Consultant: Janssen, Laura Pina Vegas Support for attending meeting: Novartis, Laura C. Coates Speaker for AbbVie, Amgen, Biogen, Celgene, Eli Lilly, Galapagos, Gilead, GlaxoSmithKline, Janssen, Medac, Novartis, Pfizer, and UCB, Consultant: AbbVie, Amgen, Bristol Myers Squibb, Celgene, Eli Lilly, Gilead, Galapagos, Janssen, Moonlake, Novartis, Pfizer, and UCB, Grants/research support from AbbVie, Amgen, Celgene, Eli Lilly, Janssen, Novartis, Pfizer, and UCB, Oyediran Adelakun Johnson & Johnson, Janssen Scientific Affairs, LLC, a Johnson & Johnson company.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (