

Background: Glucocorticoids (GC) are essential in managing inflammatory arthritis [1] but may also have a role in osteoarthritis [2] . GC have dose-dependent side-effects [3], but the use of intramuscular (i.m.) GC might ameliorate these issues without losing effectiveness [4, 5]. Prospective evidence on predictors for effectiveness of i.m. GC in clinical practice is limited, despite widespread use. Therefore, it is not surprising that effectiveness and safety profile of i.m. GC is part of the EULAR research agenda for RA [6].
Objectives: The present study aims to explore the course of pain after i.m. GC, and the value of routinely available parameters, such as demographics (e.g. age, gender, body mass index (BMI)), diagnosis (i.e. rheumatoid arthritis (RA), osteoarthritis (OA), polymyalgia rheumatica (PMR), psoriatic arthritis (PsA)), comedication and inflammatory parameters (C-reactive protein (CRP), erythrocyte sedimentation rate (ESR)) in predicting the 2-week response to i.m. GC administration.
Methods: A prospective cohort study was conducted. Consecutive consenting adult patients (all diagnoses) receiving one single i.m. GC (methylprednisolone 120 mg) in routine care at the rheumatology department of the Sint Maartenskliniek during a period of three months (between March 5, 2024, and June 19, 2024) were included. Primary outcome was the change in numeric rate scale (NRS) pain (0-10) between baseline (date of injection) and two weeks post-injection, and changes in pain at 4, 6 and 12 weeks after injection were evaluated as secondary outcomes. Descriptive statistics and univariate linear regression analyses were used with a p<0.05 considered to be significant. Parameters with a p<0.20 in the univariate linear regression were also analyzed in the multivariate model.
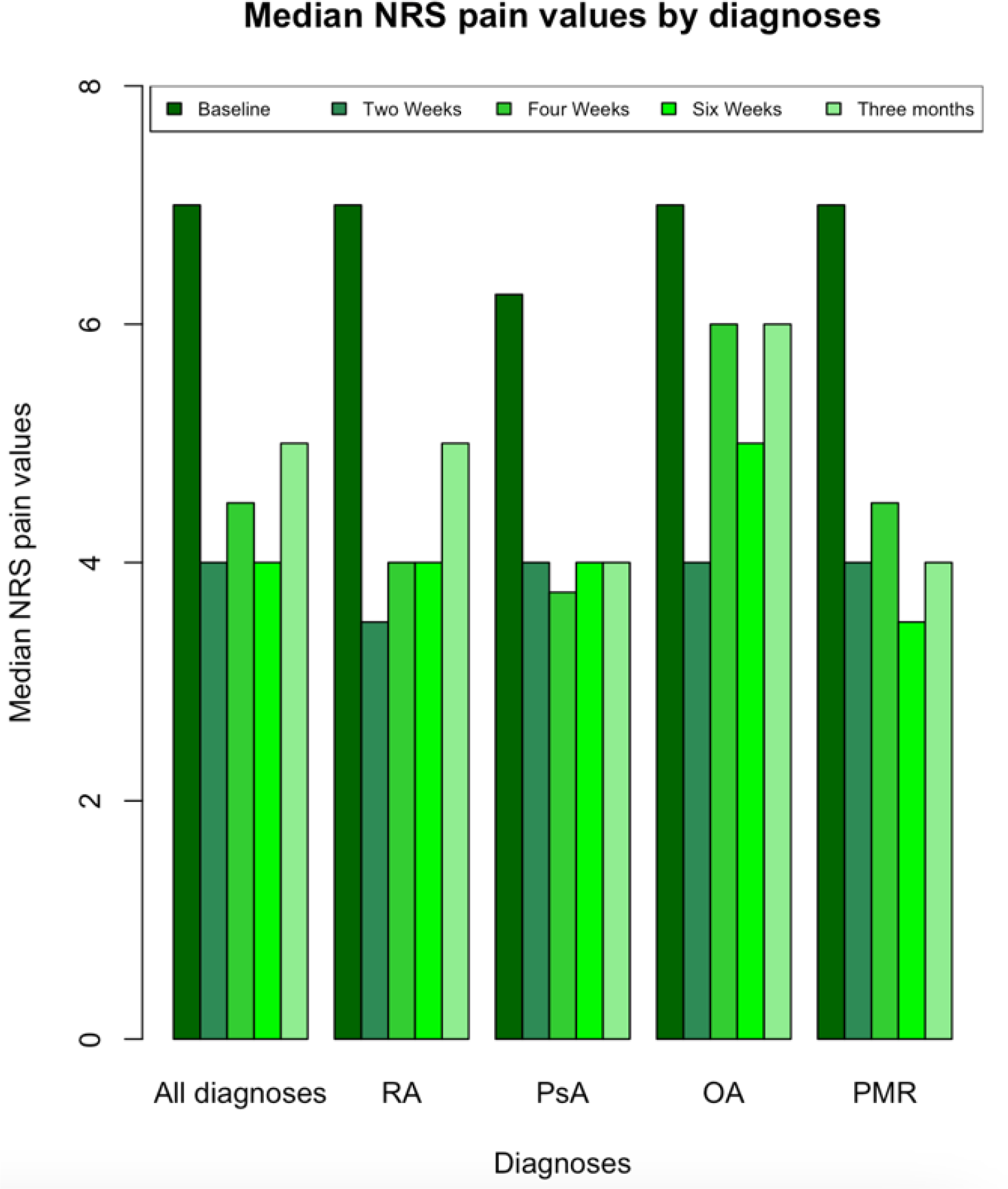
Results: 253 patients were included. The median NRS pain scores were 7 (interquartile (IQR) 3) at baseline and 4 (IQR: 4) two weeks post-injection. Univariate linear regression for the change in pain from baseline to 2 weeks revealed a significant association with seropositive RA, CRP and baseline NRS-pain. (respective beta (p-value): 1.20 (0.02), 0.04 (0.02), 0.52 (<0.001)). No other demographic (e.g., gender, BMI) or clinical factors (e.g., disease duration, comedication, ESR) were significantly associated with change in NRS-pain. In the multivariate model baseline NRS-pain was the only variable that remained statistically significant (beta 0.57 (p<0.001)).
Conclusion: In our study we observe an improvement in pain after i.m. GC, irrespective of diagnosis. This improvement is probably due to a combination of true pharmacological effect of GC, effects of concomitant disease-modifying anti-rheumatic drugs (DMARDs) (hence the persistent improvement in RA, PsA), and regression to the mean (temporary in OA, and persistent in PMR), as baseline NRS-pain remained the only predictor of response to GC. These data can inform design of future prospective studies assessing value of i.m. GC.
REFERENCES: [1] Bijlsma J. W. J. (2024). Annals of the Rheumatic Diseases collection on glucocorticoids (2020-2023): novel insights and advances in therapy. Annals of the rheumatic diseases, 83(1), 4–8.
[2] Kroon, F. P. B., et al (2019). Results of a 6-week treatment with 10 mg prednisolone in patients with hand osteoarthritis (HOPE): a double-blind, randomised, placebo-controlled trial. Lancet (London, England), 394(10213), 1993–2001.
[3] Bergstra, S. A., et al (2023). Efficacy, duration of use and safety of glucocorticoids: a systematic literature review informing the 2022 update of the EULAR recommendations for the management of rheumatoid arthritis. Annals of the rheumatic diseases, 82(1), 81–94.
[4] de Jong, P. H., et al (2014). Randomised comparison of initial triple DMARD therapy with methotrexate monotherapy in combination with low-dose glucocorticoid bridging therapy; 1-year data of the tREACH trial. Annals of the rheumatic diseases, 73(7), 1331–1339.
[5] Dasgupta, B., Dolan, A. L., Panayi, G. S., & Fernandes, L. (1998). An initially double-blind controlled 96 week trial of depot methylprednisolone against oral prednisolone in the treatment of polymyalgia rheumatica. British journal of rheumatology, 37(2), 189–195.
[6] Smolen, J. S., et al (2023). EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2022 update. Annals of the rheumatic diseases, 82(1), 3–18.
Median NRS pain values for various diagnoses at baseline and follow-up time points. The diagnoses include all the diagnoses, RA, PsA, OA and PMR. The various tinted green bars represent VAS pain values from left to right at baseline, two weeks, four weeks, six weeks and twelve weeks.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (