

Background: EULAR recommendations informing the management of Polymyalgia Rheumatica (PMR) and Large Vessel Vasculitis (LVV) were previously published separately in 2015 [1] and 2020 [2] respectively. Newly-emerging evidence has necessitated a comprehensive update.
Objectives: To conduct a systematic review to inform the 2025 EULAR recommendations for PMR and LVV.
Methods: This systematic review (PROSPERO registration: CRD42024560403), performed on 19th June 2024 on MEDLINE, EMBASE and CENTRAL searched for literature on adult patients with PMR, Giant Cell Arteritis (GCA) or Takayasu arteritis (TAK), using MeSH terms for pharmacological and non-pharmacological management. Eligible studies included randomized controlled trials (RCTs) and observational studies, published since 1st April 2014 (for PMR) or 31st December 2017 (for LVV), without language limitations. Abstracts presented at EULAR 2024 and ACR 2024 were hand-searched specifically for any RCT data, also looking to identify related full-manuscript publications following the end-date of the systematic review search. Studies were assessed for their qulity using relevant Risk of Bias (RoB) tools for RCTs/observational studies (Cochrane Risk of Bias 2/Newcastle Ottawa Scale).
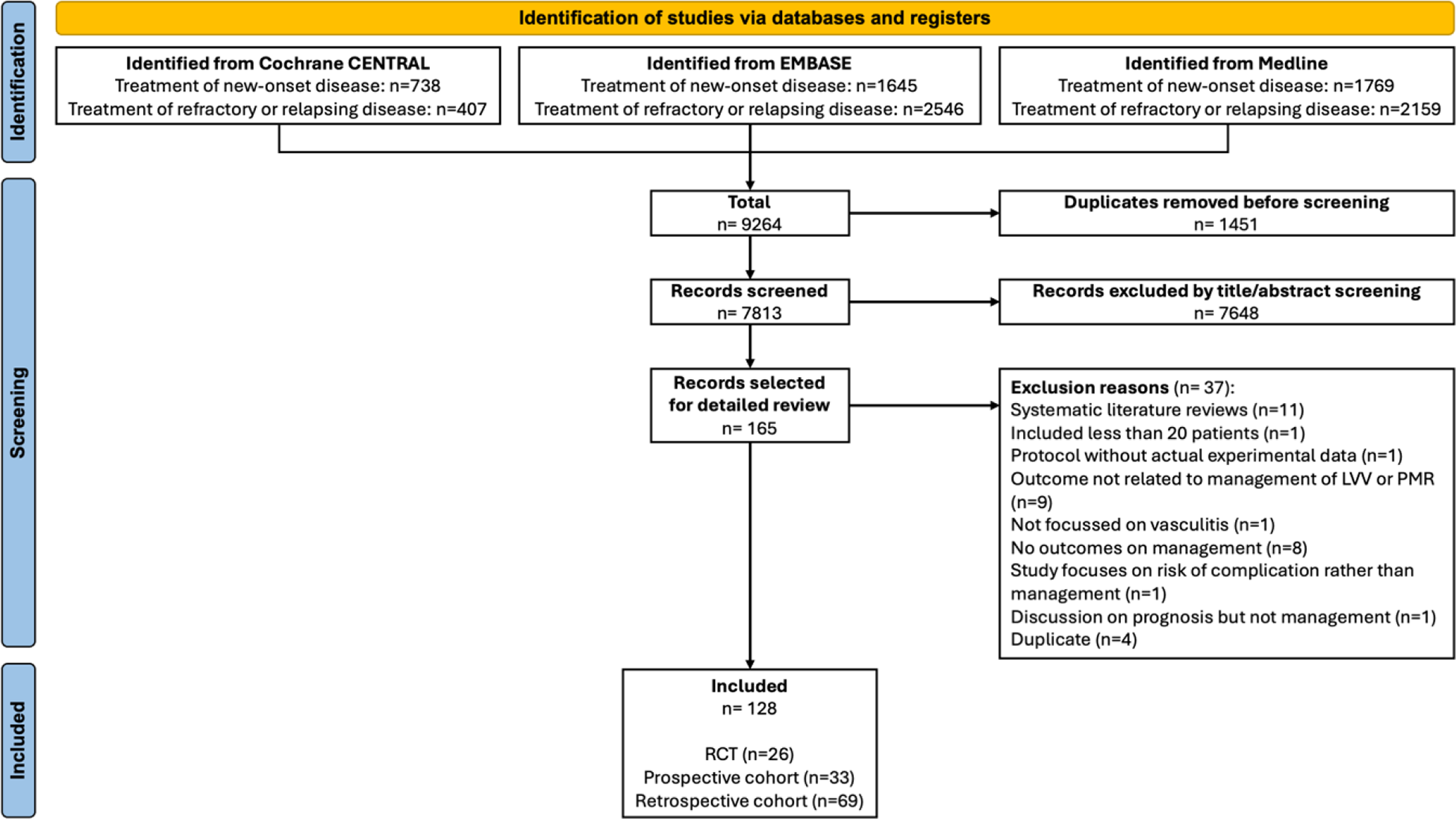
Results: After deduplication and full-text screening, 128 articles were included (26 RCTs) (Figure 1), along with 4 RCTs from ACR & EULAR 2024 abstracts. RCTs of GCA (low RoB) reported superiority of secukinumab (Phase-2, n=1), mavrilimumab (Phase-2, n=1) and upadacitinib (Phase-3, n=1) vs placebo; Phase-2 trials of sarilumab (n=1) and sirukumab (n=1) were prematurely terminated (high RoB). The two RCTs of sariliumab and sirukumab in GCA were at high risk of bias due to their premature termination due to which no inferences could be drawn about their effectiveness. Secondary analyses from the GiACTA trial reported similar efficacy of tocilizumab versus placebo in those with cranial symptoms, PMR symptoms, or both cranial & PMR symptoms, and better improvements in SF-36 and fatigue scores with tocilizumab than placebo. RCTs of TAK reported similar effectiveness of methotrexate or mycophenolate (unclear RoB), and superiority of adalimumab vs tocilizumab (high RoB). Phase-3 RCTs in PMR revealed superiority of tocilizumab and sarilumab (low RoB) vs. placebo while smaller phase-2 studies provided initial evidence for efficacy of rituximab (low RoB) and abatacept (unclear RoB) vs placebo (Table 1). A RCT showed similar efficacy of tofacitinib or glucocorticoids in PMR although this study was at high risk of bias.
Conclusion: The systematic review identified several new Phase-2 and Phase-3 RCTs about the efficacy and safety of IL-6i, IL-17i, Janus kinase inhibitors and B-cell depletion therapies in GCA and PMR. High quality adequately sized RCTs in TAK are still lacking. This evidence was used to inform the 2025 EULAR recommendations for management of Polymyalgia Rheumatica and Large Vessel Vasculitis.
REFERENCES: [1] Dejaco C, et al. Ann Rheum Dis. 2015 Oct;74(10):1799-807.
[2] Hellmich B, et al. Ann Rheum Dis. 2020 Jan;79(1):19-30.
PRISMA flowchart
Low Risk of Bias RCTs* of patients with GCA or PMR
| Disease,
| Author Year | Intervention (n)
| Primary outcome | Results | Conclusion |
|---|---|---|---|---|---|
| GCA: treatment-naïve and relapsing | Cid 2022 | MAV (n=42)
| Time to flare (ITT) | HR 0.38 (0.15-0.92) | MAV superior to PBO |
| Venhoff 2023 | SEC(n=27)
| Sustained remission (28w, ITT) | RD 0.50 (0.29 - 0.67)
| SEC superior to PBO | |
| PMR: treatment-naive | Cutolo 2017 | MR PRED (n=32)
| Complete response rate (4w, PP) | RD 12.22 (95%CI -15.82% to 40.25%) did not met non-inferiority criteria (-15%). | MR PRED similar to IR PRED. |
| Bonelli 2022 | TOC (n=19)
| GC-free remission (16w) | OR 12.9 (2.2-73.6) | TOC superior to PBO | |
| PMR: treatment-naïve and relapsing | Marsman 2021 | RTX
| GC-free remission (21w) | RD (one sided 95%CI): 27 (4) %
| RTX superior to PBO |
| Devauchelle-Pensec 2022 | TOC (n=49)
| CRP PMR- AS <10 with PRED≤5 mg/day/ reduction in PRED dose ≥10 mg/day (w24, ITT) |
| TOC superior to PBO | |
| PMR: Relapsing | Spiera 2023 | SAR (n=59)
| Sustained remission (52w) | SAR 28%, PBO 10% | SAR superior to PBO |
*From full published manuscripts
CRP. -C-reactive protein, GC – Glucocorticoid, HR – Hazard ratio, IR – Immediate release, ITT – Intention to treat, MAV – Mavrilimumab, OR – Odds ratio, PBO – Placebo, PP – Per protocol, PRED – Prednisone, MPRED – Methylprednisolone, MR – Modified release, PMR-AS - PMR activity score, RD - Risk difference, RR – Risk ratio, RTX – Rituximab, SAR – Sarilumab, SEC – Secukinumab, TOC – Tocilizumab
Acknowledgements: Funding was received from EULAR.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (