

Background: SLE is an autoimmune disease of unknown origin. Pain and reduced health-related quality of life (HRQoL) are major challenges within systemic lupus erythematosus (SLE) and remain inadequately addressed by conventional immunosuppressive therapies. Transcutaneous vagus nerve stimulation (tVNS) has shown potential to modulate systemic inflammation via the cholinergic anti-inflammatory reflex and our preliminary results have indicated effects on symptoms such as fatigue (Zinglersen et al. 2024).
Objectives: To investigate if tVNS as adjuvant treatment to standard care could improve patient-reported levels of HRQoL and pain in SLE patients.
Methods: SLE patients with signs of fatigue (FACIT-F score <40) and autonomic dysfunction (Composite Autonomic Symptoms Score >12) were randomized 1:1 to active or placebo tVNS in this double-blinded parallel-group study (Zinglersen et al. 2022). In work package (WP)1 (one week), the patients performed four minutes of tVNS four times daily. After a 2-week pause, WP2 (eight weeks) entailed four minutes of tVNS, but only two times daily. The patients completed the questionnaires before and after each WP, and one week into WP2. The patients completed the Short Form-12 (SF-12) questionnaire, which includes nine items addressing various aspects of health-related quality of life (HRQoL). From their responses, two component scores were derived: one reflecting physical- (PCS) and the other reflecting mental- HRQoL (PCS). Additional average joint and muscle pain levels were assessed using a Visual Analog Scale (VAS), rating the average pain intensity experienced over the past week on a scale from 0 (no pain) to 10 (worst possible pain). Linear mixed modeling of repeated measures was performed to examine effects of tVNS in WP1 and WP2, respectively.
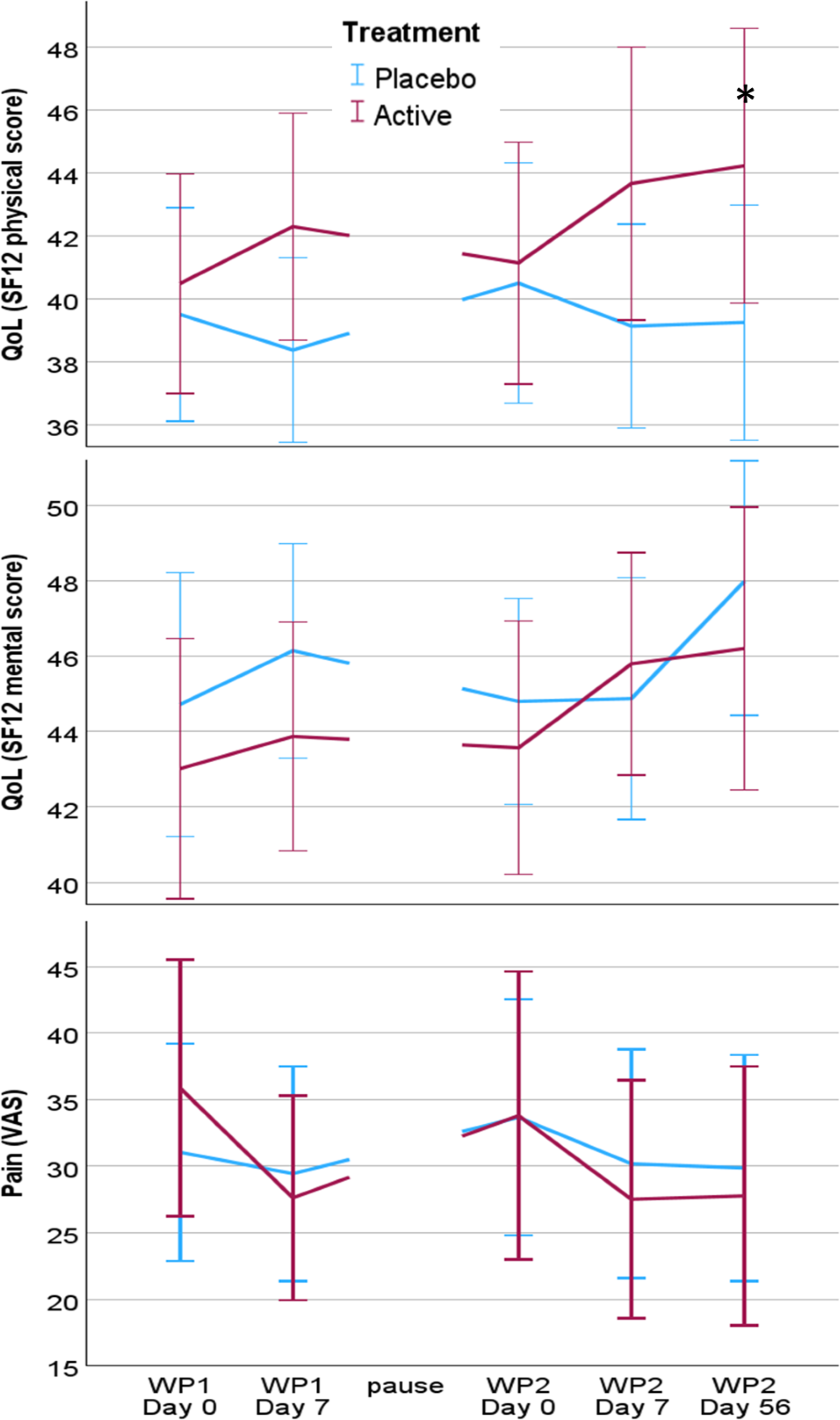
Results: Sixty-one patients were included in the analyses (30 placebo, 31 active tVNS). All the patients’ mean age was 47.6 years (±14.2 SD), and 91.8% were women. There were no differences in baseline levels of age, sex, PCS, MCS or VAS pain (data not shown, all p>0.05). Figure 1 illustrates PCS, MCS and pain during the study, separated into WP1 and WP2. PCS improved more in the active-treatment group than in the placebo-group during the study: During WP1 the active treatment-group increased their PCS with 7.3% more than the placebo group, however not statistically significant (p=0.09). In WP1 week 1 there was a similar pattern with an 8.7% relative increase in the active treatment group, p=0.07. Interestingly, during the full eight weeks of WP2, the active treatment group improved their PCS by 11.3% compared to the placebo group, which was statistically significant (p=0.03). However, neither MCS nor pain changed during the study (p>0.05).
Conclusion: Vagus nerve stimulation (VNS) shows potential as a novel therapeutic strategy for enhancing the physical components of HRQoL in SLE. Short-term stimulation protocols (two vs. four sessions per day over one week) showed no significant differences, while long-term treatment over eight weeks significantly improved PCS. However, VNS did not improve self-reported pain or mental HRQoL. These findings highlight the need for larger, randomized controlled trials to confirm efficacy and investigate underlying mechanisms.
REFERENCES: [1] Zinglersen et al. AB1036 Annals of the Rheumatic Diseases 2024;83:1839. doi: 10.1136/annrheumdis-2024-eular.1975.
[2] Zinglersen et al. BMJ Open. 2022 sep. 20;12(9):e064552. doi: 10.1136/bmjopen-2022-064552.
Acknowledgements: NIL.
Disclosure of Interests: None declared.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (