

Background: Systemic lupus erythematosus (SLE) is a chronic inflammatory autoimmune disease associated with debilitating symptoms that substantially impact patients’ health-related quality of life (HRQOL). Patients with SLE often experience considerable pain, fatigue, impaired physical and social activities, and psychological distress; thus, optimal treatments for SLE should aim to reduce disease activity and improve HRQOL-related symptoms. Upadacitinib (UPA), an oral selective Janus kinase inhibitor, demonstrated significantly reduced disease activity vs placebo in a phase 2 trial of patients with moderately to severely active SLE. Here, we evaluated associations between patient-reported outcomes (PROs; including fatigue, pain, and physical and mental function) and disease activity in patients treated with UPA.
Objectives: To describe associations between clinically important improvements in PROs and reduced disease activity from the phase 2 SLEek trial evaluating UPA 30 mg monotherapy vs placebo in patients with SLE.
Methods: SLEek was a phase 2, randomized, double-blind, placebo-controlled trial evaluating the efficacy and safety of UPA and elsubrutinib (a Bruton’s tyrosine kinase inhibitor) when administered as monotherapy or combination therapy. Patients with moderately to severely active SLE were randomized 1:1:1:1:1 to receive once-daily UPA 30 mg and elsubrutinib 60 mg, UPA 15 mg and elsubrutinib 60 mg, UPA 30 mg monotherapy, elsubrutinib 60 mg monotherapy, or placebo for 48 weeks. Here, we report results from the UPA 30 mg monotherapy and placebo arms. Assessments included achievement of British Isles Lupus Assessment Group-Based Composite Lupus Assessment (BICLA), Systemic Lupus Erythematosus Responder Index 4 (SRI-4), and Lupus Low Disease Activity State (LLDAS) responses among patients who reported or did not report minimum clinically important differences (MCID) in PROs at weeks 24 and 48. MCIDs were based on previously published thresholds of ≥ 4-point increase in Functional Assessment of Chronic Illness Therapy (FACIT)-Fatigue (in patients with FACIT-Fatigue ≤ 48 at baseline), ≥ 2.5-point increase in 36-Item Short-Form Health Survey Physical and Mental Component Summary scores (SF-36 PCS and MCS scores; in patients with SF-36 PCS and MCS scores ≤ 97.5 at baseline), and ≥ 1-point reduction from baseline in Patient’s Assessment of Pain Numerical Rating Scale (Pain NRS, 0–10 scale; in patients with baseline Pain NRS ≥ 1). Missing data were handled using nonresponder imputation. Between-group differences and P values (UPA vs placebo) were estimated using the Cochran-Mantel-Haenszel test, adjusted for baseline corticosteroid dose, Systemic Lupus Erythematosus Disease Activity Index 2000 score at screening, baseline interferon score, and baseline immunosuppressant use.
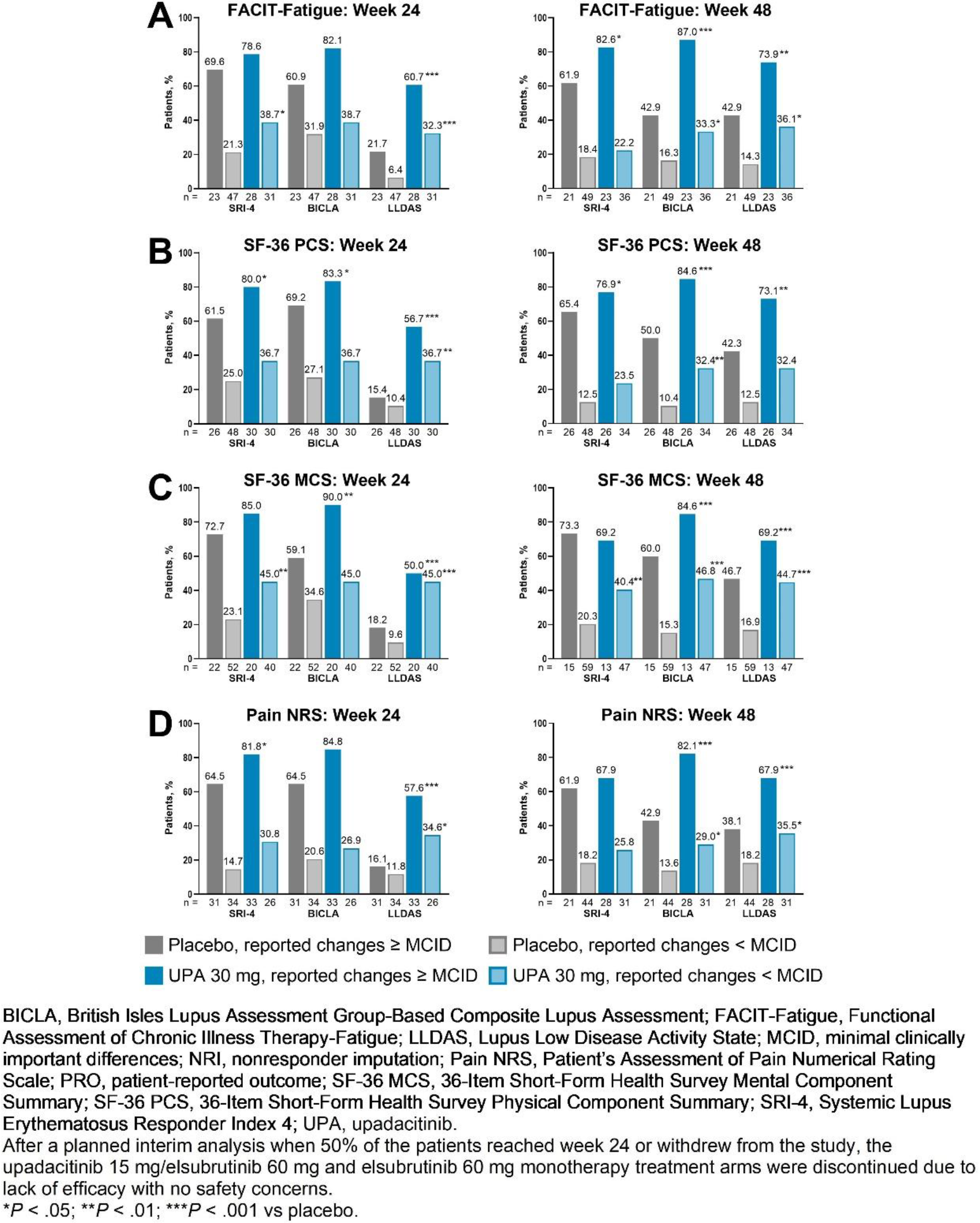
Results: A total of 137 patients were included in the analysis (UPA 30 mg, n = 62; placebo, n = 75). Over 90% of patients were female, consistent with the general population of patients with SLE, and most patients were White. Mean baseline FACIT-Fatigue, SF-36 PCS, SF-36 MCS, and Pain NRS scores were 29.3, 40.3, 42.4, and 5.3, respectively, in the placebo group and 27.1, 38.2, 43.7, and 7.1 in the UPA group. Overall, among patients who reported changes ≥ MCID for fatigue, physical or mental function, or pain at week 48, a significantly higher proportion achieved SRI-4, BICLA, and LLDAS responses with UPA vs placebo (all P <.05 except for SRI-4 among patients who received UPA and reported changes ≥ MCID for SF-36 MCS scores or Pain NRS; Figure 1). Similar trends were observed at week 24: responses were significantly higher with UPA vs placebo for LLDAS in patients who reported changes ≥ MCID for FACIT-Fatigue; for SRI-4, BICLA, and LLDAS among patients who reported changes ≥ MCID for SF-36 PCS scores; for BICLA and LLDAS in patients who reported changes ≥ MCID for SF-36 MCS scores; and for SRI-4 and LLDAS in patients who reported changes ≥ MCID for Pain NRS. SRI-4, BICLA, and LLDAS responses at weeks 24 and 48 were lower among patients who did not report clinically meaningful improvements in FACIT-Fatigue, SF-36 PCS or MCS scores, or Pain NRS; however, a higher proportion of these patients achieved SRI-4, BICLA, and LLDAS responses with UPA vs placebo. Of patients who received UPA and reported changes ≥ MCID for FACIT-Fatigue at week 48, over 80% achieved SRI-4 and BICLA responses, and approximately three-quarters achieved LLDAS (Figure 1A). In patients who received UPA and reported clinically meaningful changes in SF-36 PCS or MCS scores or Pain NRS at week 48, over 80% achieved BICLA, and SRI-4 and LLDAS were achieved by approximately three-quarters of patients who reported changes ≥ MCID for SF-36 PCS scores (Figure 1B) and approximately two-thirds of patients who reported changes ≥ MCID for SF-36 MCS scores (Figure 1C) and Pain NRS (Figure 1D).
Conclusion: Patients with moderately to severely active SLE who reported changes ≥ MCID in PROs for fatigue, physical and mental function, and pain generally demonstrated higher SRI-4, BICLA, and LLDAS responses than those who did not. These findings demonstrate the strong association of PRO improvements with disease activity improvements in SLE. Associating HRQOL improvements from the patient’s perspective with reduced disease activity highlights the potential multifaceted benefits of UPA for patients with SLE.
REFERENCES: NIL.
Acknowledgements: AbbVie funded this study and participated in the study design, research, analysis, data collection, interpretation of data, reviewing, and approval of the abstract. All authors had access to relevant data and participated in the drafting, review, and approval of this abstract. No honoraria or payments were made for authorship. AbbVie and the authors thank all the trial investigators and the patients who participated in this clinical trial. Medical writing support was provided by Callie A S Corsa, PhD, ISMPP CMPP, of JB Ashtin, and funded by AbbVie.
Disclosure of Interests: Vibeke Strand VS has served as a consultant for AbbVie, Alpine Immune Sciences, Alumis, Amgen, AstraZeneca, Bayer, Blackrock, Boehringer Ingelheim, Bristol Myers Squibb, Celltrion, Citryll, Contura, Cullinan, Fortress, Genentech/Roche, GlaxoSmithKline, Inmedix, Janssen, Kiniksa, Lilly, Novartis, Omeros, Pfizer, R-Pharma, Rapt, Regeneron, Samsung, Sandoz, Sanofi, Scipher, Setpoint, Sobi, Spherix, Synact, and Takeda, Zahi Touma ZT has served as a consultant for AstraZeneca, Biogen IDEC, EMD Serono, GlaxoSmithKline, Janssen, Lilly, Pfizer, Sarkana, and UCB, Anca Askanase ADA has received consulting fees from AbbVie, Amgen, Aurinia, AstraZeneca, Bristol Myers Squibb, and GlaxoSmithKline. She has also served as an investigator for AstraZeneca, GlaxoSmithKline, Janssen, Lilly, Pfizer, UCB, and Vielo, Christopher D Saffore CDS is a full-time employee of AbbVie, and may hold AbbVie stock or stock options, CDS is a full-time employee of AbbVie, and may hold AbbVie stock or stock options, Denise Kruzikas DTK is a full-time employee of AbbVie, and may hold AbbVie stock or stock options, DTK is a full-time employee of AbbVie, and may hold AbbVie stock or stock options, Karim Masri KRM is a full-time employee of AbbVie, and may hold AbbVie stock or stock options, KRM is a full-time employee of AbbVie, and may hold AbbVie stock or stock options, Siran Fang SF is a full-time employee of AbbVie, and may hold AbbVie stock or stock options, SF is a full-time employee of AbbVie, and may hold AbbVie stock or stock options, Yi Peng YP is a full-time employee of AbbVie, and may hold AbbVie stock or stock options, YP is a full-time employee of AbbVie, and may hold AbbVie stock or stock options, Patricia Katz: None declared, Marta Mosca MM has received consulting fees from AstraZeneca, Bristol Myers Squibb, GlaxoSmithKline, Novartis, UCB, Idorsia, Otsuka, Abbvie, Biogen.
© The Authors 2025. This abstract is an open access article published in Annals of Rheumatic Diseases under the CC BY-NC-ND license (